Abstract
Recently, we found, using a cigarette carcinogen-induced lung cancer model, that soy protein isolate (SPI) was superior to casein in preventing lung cancer. In this study, we have attempted to identify the component(s) within SPI responsible for this chemopreventive effect. We fractionated the SPI using ethanol to separate the ethanol-soluble fraction (ESF) and the washed SPI and compared their efficacy to diets made with amino acids that comprise soy protein or casein, in preventing lung tumor formation in A/J mice. Only the ethanol-soluble fraction was as effective as SPI in preventing lung tumor formation. Since isoflavones and saponins are known ethanol-soluble bioactives from soy, we added isoflavones, or saponins or both to casein and found that isoflavones or saponins alone did not reduce lung nodule formation. However, when we combined soy saponins and isoflavones, we saw a significant (P < 0.05) reduction in NNK-induced lung nodules, and an increase in plasma isoflavone levels, suggesting that the saponins may enhance the bioavailability of the isoflavones in these mice. Taken together, we suggest that the superior efficacy of SPI over casein could be attributed, at least in part, to the synergistic effect of the soy saponins and isoflavones.
Similar content being viewed by others
Introduction
The first hint that soy foods might lower the incidence of cancer came from population studies showing breast cancer rates were low in places where there was high consumption of soy1,2. Subsequent animal studies confirmed soy was indeed more effective than casein, a milk protein commonly used in rodent chows, at preventing mammary, prostate, liver, ovarian, colon, and cervical cancers3,4,5,6,7,8,9,10,11.
However, despite decades of research, the active component(s) in soy responsible for its anti-cancer activity has not been completely elucidated. Historically, Bowman-Birk inhibitors (BBIs) were one of the first components of soy to be investigated for potential anti-carcinogenic activity. These small proteins (6–9 kDa), are potent serine protease inhibitors that bind and inactivate the digestive enzymes trypsin, chymotrypsin, and elastase, thus allowing the BBIs to enter the circulation12. Early reports in the 1980s and 90 s suggested that BBIs may be effective in both cancer prevention13 and treatment, at least for prostate cancer14. Although BBIs subsequently received Investigational New Drug status from the FDA13, they proved ineffective in decreasing oral lesions in a 2013 randomized, placebo-controlled trial with oral leukoplakia patients, perhaps due to stability issues15.
In addition to BBIs, a number of other soy peptides and proteins have been studied. For example, soy peptides or soy protein hydrolysates, obtained using digestive enzymes, have been shown to suppress the growth of multiple cancer cell lines in vitro16,17. Of all the components within these hydrolysates, the most investigated has been lunasin, a 43 amino acid polypeptide18 that has been shown to inhibit the carcinogen-induced transformation of cells and the proliferation of several cancer cell lines19,20,21,22,23,24,25. Lunasin has also been shown to inhibit tumor growth in vivo26,27. Mechanistically, it has been suggested that lunasin does this, at least in part, by preventing histone acetylation28,29.
It should also be mentioned that plant proteins in general differ from animal proteins in essential amino acid content and this is especially true for methionine, which is significantly lower in soy proteins than in casein30. This could be important, given that methionine has been shown to promote cancer cell growth and metabolism31.
In addition to peptides and proteins, soybeans are a rich source of isoflavones. Because of their high levels in soy, it was originally hypothesized that they were the components that reduced the risk of hormone-sensitive cancers such as breast and prostate cancers. This is because some of these ethanol-soluble compounds have been shown to bind estrogen receptors, potentially eliciting estrogenic activity at low concentrations and anti-estrogenic activities at high concentrations32,33. The three most abundant isoflavones in soy are genistin, daidzin, and glycetin, which are the glycosylated forms of the aglycones genistein, daidzein, and glycetein, respectively34. As well, daidzein can be metabolized by the gut microbiome to generate equol, which also has estrogenic activity35. A number of other components in soy with potential anti-cancer activity have been investigated, including soy saponins, which comprise approximately 5% of the dry weight of soybeans36. These saponins have been shown in vitro to reduce the growth of breast and colon cancer cell lines37,38,39. Tested in vivo, soy saponins have been shown to repress human MDA-MB-231 mammary tumor growth in immunocompromised mice37 and reduce the number of carcinogen-induced aberrant crypt foci in rats40.
We recently found that soy protein isolate (SPI), a protein-enriched (> 90%) product from soybeans, devoid of fat and carbohydrate (CHO), was superior to casein in reducing the induction of lung cancer nodules by the cigarette smoke carcinogen, nicotine-derived nitrosamine ketone (NNK)41. This observation has given us the opportunity to investigate which component(s) within SPI is responsible for this effect. Our results are presented herein.
Methods
Mice
Female A/J mice were purchased from the Jackson Laboratory (Bar Harbor, ME), and acclimatized to our specific pathogen-free facility in the British Columbia Cancer Research Centre. They were fed our standard Envigo code 2920 chow, ad libitum, until the mice reached 12 weeks of age, at which time they were randomly assigned to the various diets indicated below. The mice (10–15/cage) were housed in double-decker rat cages containing one exercise wheel and two huts per level instead of in standard mouse cages, so they could exercise freely as described previously41. Mice that reached humane endpoints before the experimental endpoints due to reasons unrelated to the study are excluded from the analysis. All animal experiments complied with the ARRIVE guidelines. All experiments were performed following guidelines and regulations and was approved by the University of British Columbia Animal Care Committee (A14-075/A18-0138/A22-0072).
Diets
All diets were custom-made by Envigo (Madison, WI), vacuum-packed in 1 kg bags, irradiated, and stored at − 20 °C before and after opening. All diets used in this study were isocaloric low CHO diets and contained 15% (of total calories) as the resistant starch, Amylose, 35% as protein and 50% as fat, with the fat component being a mixture of fats normally consumed by humans on a Western diet42. Isoflavones (Novasoy400) was a gift from Archer Daniels Midland (Decatur, IL). The isoflavone levels within Novasoy and SPI were determined by N.P. Analytical Laboratories (St. Louis, MO). Isoflavone-free (< 0.3%) saponins (Saponin B-50), which contain a minimum of 59.9% soyasaponin group B and 12.3% soyasaponin group A, were a gift from J-Oil Mills (Japan).
Study #1
To investigate the role of soy proteins and isoflavones in preventing NNK-induced lung nodule formation, 12-week old mice (9–12 mice/group) were put on low CHO, (15% Amylose) diets containing either casein, SPI, or casein supplemented with isoflavones (Table 1). The isoflavones were added to casein at 3.6 g/kg, so that the level of genistein, the main isoflavone in SPI, was comparable to that present in SPI (Table 1).
Animal experiments
After acclimatizing to their diets for two weeks, mice were then intraperitoneally (IP) injected with NNK (50 mg/kg body weight) twice, one week apart, to initiate lung nodule formation. The mice were kept on their respective diets for 20 weeks and then anesthetized with isoflurane and euthanized by CO2 asphyxiation, followed by cervical dislocation. Blood was obtained by cardiac puncture into heparin-coated tubes and centrifuged at 2500×g for 10 min at 4℃. Plasma was immediately frozen in dry ice and stored at − 80 °C for future biochemical analyses. Mouse lungs were rinsed with PBS and tumor nodules were counted by two blinded counters using a Leica MZ9.5 microscope (Meyer Instruments, Houston TX). Lungs were either immediately frozen or fixed in 10% formalin after inflating the lung with 1 mL of PBS. The fixed lungs were then stored in 70% ethanol, prior to paraffin embedding, sectioning (4 micron thickness) and H&E staining.
Study #2
To determine if non-isoflavone components in SPI were contributing to the anti-cancer activity of the SPI, the SPI was ethanol washed to remove the isoflavones and other ethanol soluble compounds as described in Supplementary Fig S1, using an extraction method adapted from Fukui et al.43. Briefly, SPI powder obtained from Envigo (Solae Supro® 661) was mixed with 70% ethanol (10 × v/w) for 16 h at 23℃. After vacuum filtration using a Whatman no. 1 filter, the filter cake was re-suspended in 70% ethanol (3 × v/w) and mixed for 15 min. The soy protein was allowed to settle for 30 min and then vacuum-filtered to obtain ethanol extract #2. The settled soy protein was extracted a third and final time with 99.5% ethanol (2 × v/w), stirred for 15 min before subjecting the mixture to a final vacuum filtration. The washed SPI was then spread out on a pre-weighed baking tray and air-dried for 16 h. Once the weight was stable, the washed SPI was stored at -20℃ until shipment to Envigo for incorporation into mouse chows. The ethanol solutions from the three extractions were pooled and evaporated to complete dryness using a Labconco lyophilizer (Kansas City, MO). This ethanol-soluble fraction (ESF) constituted approximately 8.1% of the SPI. This ESF of SPI was incorporated into the low CHO diet at a level comparable to that found in SPI. Since we did not find that isoflavones alone were effective in lowering lung nodule formation, we added the ESF to 15% Amylose/Isoflavones to explore the possibility that non-isoflavone components within the ESF may work synergistically with isoflavones.
To determine if the superiority of SPI over casein at reducing NNK-induced lung nodules was due to differences in their amino acid composition, we formulated two diets containing amino acid mixtures that reflected the amino acid compositions of casein and soy protein. The diets used in this Study #2 are described in Table 2. The amino acid profile of the diets containing amino acids instead of intact proteins is described in Table 3. The casein containing 15% Amylose diet supplemented with isoflavones were used to confirm our findings in Study #1. The 15% Amylose diet served as controls. Animal experiments were performed as described in Study #1.
Study #3
Based on our findings from Study #2, we concluded that the tumor lowering activity was in the ESF of the SPI. Since soy saponins are ethanol soluble compounds previously reported to have anti-cancer effects44, we tested in Study #3 the effect of 15% Amylose diets containing soy saponins at a level comparable to that in SPI, in the presence and absence of soy isoflavones. We describe the composition of the diets used in Study #3 in Table 4. The 15% Amylose diet served as controls. Animal experiments were performed as described in Study #1.
Quantitation of saponins
Total saponins were quantified using a modified vanillin-sulfuric acid method, as described by Le et al.45. Briefly, ground rodent chows, the SPI, ethanol-washed SPI, ethanol-extract from SPI and Novasoy were reconstituted at 50 mg/mL in 70% ethanol for the extraction of saponins. Genistein was reconstituted at 0.1 mg/mL in 70% ethanol. The mixtures were sonicated for 2 min (40 Amp) twice and chilled on ice between rounds and after. The samples were centrifuged at 16,060×g for 10 min at 4℃, and the supernatants filtered using a 0.45 µm membrane filter. Then, 0.25 mL of each extract was combined with 0.25 mL of 8% vanillin made in ethanol and 2.5 mL of 72% sulfuric acid in a screw-capped glass tube. After mixing, the tubes were incubated in a 65℃ water bath for 15 min, immediately chilled on ice and the absorption read at 560 nm. Soyasaponin I (Cat# S9951, Sigma-Aldrich, St. Louis, MO) reconstituted in 70% ethanol at 1 mg/mL was used to generate a standard curve, and results were expressed as total Soyasaponin I equivalent.
Quantification of isoflavones in plasma
The genistein in mouse plasma was quantified using LC/MS, based on a method described by Twaddle et al.46. Briefly, 65 µL of plasma was mixed with an equal volume of d4-genistein (300 ng/mL in acetonitrile), vortexed and sonicated for 10 min. After centrifugation at 16,060×g for 5 min at 23℃, 100 µL of the supernatant was mixed with 900 µL of β-glucuronidase from Helix pomatia solution (G1512, 100 µg in 25 mM citrate buffer pH 5.0). After brief vortexing, the samples were placed on a heating block at 37℃ for 1 h. Upon completion, three rounds of solvent extraction were performed using 1 mL ethyl acetate. The ethyl acetate top layer was pooled, vacuum-evaporated and reconstituted in 50% methanol for LC/MS analysis.
Blank plasma samples (60 µl) were spiked with genistein and d4-genistein (Cayman Chemical, Ann Arbor, MI) internal standard (IS) and precipitated with an equal volume of acetonitrile, centrifuged at 20,000 g for 2 min and 100 µL of supernatant added to 900 µL of enzyme stock. Following incubation they were extracted with ethyl acetate, the extract dried and the residue taken up in 100 µL of 67% acetonitrile. The resulting seven calibration samples had a final IS of 150 ng/mL with genistein ranging from 0–1500 ng/mL. Test samples were similarly prepared, omitting the genistein spike. All samples were analysed with a Waters Acquity LC coupled to a Waters Quattro Premier using a 2.1 × 50 mm BEH C18, 1.7µ column (Waters). Water (0.1% formic acid (FA)) and ACN (0.1% FA) were LC solvents A and B with 5% B, 0–0.2 min; 5–98% B, 0.2–2.5 min; 95% B, 2.5–3.0 min; 98–5% B, 3–3.1 min; with total run length 4 min at 0.33 mL/min. All MS data was collected in ES + at unit resolution with the following parameters: capillary, 3 kV; extractor and RF lens, 3 V and 0.1 V; source and desolvation temperatures, 120℃ and 350℃; desolvation and cone (N2) flow, 1000 L/h and 50 L/h; collision gas (Ar) flow, 0.13 mL/min (7.8e-3 m bar). Detection was by multiple reaction monitoring with m/z 271.1 > 153 and 271.1 > 91 for genistein and m/z 275.1 > 154.1 and 271.1 > 94.1 for d4-genistein (45 V/45 V and 45 V/40 V cone/collision respectively for both pairs) with 0.1 s dwell each and 1.9 min retention time (RT) for both. Quantitation was via the AUC ratio of genistein/d4-genistain using a linear fit with 1/X weighting with R2 > 0.99 and accuracy within ± 15% across the entire range (5-1500 ng/ml) using Quanlynx (Waters) and exported to Excel for further analysis.
Statistics
Significant differences between treatment groups were performed using one-way ANOVA, using post-hoc Tukey’s or Dunnett test to identify treatment groups that are significantly (P < 0.05) different when compared to the 15% Amylose group. Assessment for normality was performed with Shapiro–Wilk test. All statistical analyses were performed using Graphpad Prism 10.2.3.
Results
SPI but not isoflavone-supplemented casein reduces lung nodule numbers
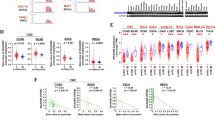
In an earlier study, we found using a low CHO, fish oil-containing diet, that 35% soy protein isolate (SPI) was more effective than 35% casein, at reducing NNK-induced lung nodules41. To confirm the potential chemopreventive effect of soy protein, we investigated the effect of SPI versus casein in the absence of fish oil, since fish oil is a potent inhibitor of lung nodule formation, and obscures to some extent the effect of protein source. As shown in Fig. 1, replacing 35% casein with 35% SPI in a low CHO (15% Amylose) diet significantly lowered NNK-induced lung nodule numbers from an average of ~ 10 ± 1 to ~ 5 ± 1 (mean ± SEM). Since soy isoflavones, present in both soy and its protein-rich product, SPI, may be the bioactive component of soy responsible for this effect, we assessed the role of isoflavones in the prevention of NNK-induced lung nodule formation. Specifically, we compared the 15% Amylose diet containing casein supplemented with soy isoflavones at levels comparable to that found in SPI, the 15% Amylose with casein only and the 15% Amylose diet containing SPI only. Interestingly, the low CHO diet containing isoflavone-supplemented casein was not effective in lowering lung nodule numbers compared to the 15% Amylose diet with casein alone (Fig. 1).
The efficacy of SPI in reducing NNK-induced lung nodule formation in A/J mice is not due to soy isoflavones. Female A/J mice (n = 12) were acclimatized to either a 15% Amylose/30% casein/50% fat diet, a 15% Amylose/30% SPI/50% fat diet or a 15% Amylose/30% casein/50% fat + isoflavone diet for 2 weeks before IP injection of NNK (50 mg/kg body weight) twice, one week apart. The mice were then kept on their respective diets for 20 weeks, euthanized and lung tumor nodules counted by two blinded counters using a Leica MZ9.5 microscope. (A) Data shown are the biological replicates of lung nodule counts per mouse as mean ± SEM. Significant differences between treatment groups were identified using one-way ANOVA followed with Tukey’s multiple comparison test. Different letters denote significant (P < 0.05) differences between treatments. (B) Representative H&E images of the tumors in the lung. Images were taken using Olympus BX46 microscope at × 100 magnification.
The efficacy of soy protein isolate is not due to the protein component of the SPI
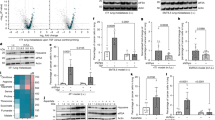
To determine if the superiority of SPI over casein was due to the proteins themselves, we carried out two studies. First, we asked if the difference in amino acid composition between soy and casein could be responsible for the superior efficacy of SPI in reducing NNK-induced lung nodules. This is possible, since certain amino acids such as methionine, which is at a significantly higher level in casein than in soy proteins30, have been shown to promote cancer cell growth and metabolism31. To test this, we replaced SPI and casein in the diets with cocktails of amino acids at levels and compositions similar to that found in casein and soy protein. As shown in Fig. 2, the cocktail of soy amino acids was not superior to casein amino acids in reducing NNK-induced lung nodules. Secondly, we treated the SPI with ethanol to separate the proteins from the ethanol-soluble components, such as soy isoflavones, saponins, and other ethanol-soluble compounds. Ethanol washing of SPI produced an ethanol-insoluble SPI protein preparation and an ethanol-soluble fraction, the latter being approximately 8.1% (w/w) of the SPI. We then tested both the pure protein preparation (ethanol-washed SPI) and the ESF and found that the protein component, i.e., the ethanol-washed SPI, did not contribute to the chemopreventive effect of the SPI (Fig. 2). Since bioactive peptides, such as lunasin, reported to have anti-cancer properties18, will be in the ethanol-washed protein fraction, we conclude that the chemopreventive effect of SPI is also unlikely due to bioactive peptides.
The efficacy of SPI in reducing NNK-induced lung nodule formation in A/J mice (n = 8–9) is not due to the difference in amino acid composition between SPI and casein, nor to the soy proteins but to the ESF within the SPI. (A) Values are biological replicates and the data are shown as the mean ± SEM. Significant differences between treatment groups and the 15% Amylose group were identified using one-way ANOVA followed with Dunnett’s multiple comparison test. Asterisks denote a significant difference between mice fed the different diets, *P ≤ 0.05, ***P ≤ 0.001. (B) Representative H&E images of the tumors in the lung from groups that were significantly impacted by the diet, relative to the 15%Amylose. Images were taken using Olympus BX46 microscope at × 100 magnification.
The efficacy of soy protein isolate to prevent lung nodules may be attributable to saponins
Given that the protein fraction of SPI was not responsible for the superiority of SPI over casein, we formulated a 15% Amylose, 35% casein + ESF diet that contained a level of ESF equal to that present in unfractionated SPI. This diet composed of casein supplemented with the ESF significantly lowered lung nodule formation (Fig. 2). Since we showed that isoflavones did not contribute to the prevention of lung nodule formation, we hypothesized that other ethanol-soluble components, such as the saponins, might be responsible for the lower lung nodule numbers in SPI-fed mice.
To test this hypothesis, we quantified the total saponin content of the low CHO diets containing casein, SPI, isoflavones only, the ethanol-washed soy protein, and the ESF. As shown in Fig. 3, ethanol washing of the SPI efficiently transferred the saponins from the SPI to the ESF. We also tested Novasoy, which is the isoflavone mixture used to formulate the isoflavone-supplemented diet, for its saponin content. It is clear that while Novasoy does contain saponins, the level identified in the product was relatively minor compared to that found in SPI.
Saponin content of the chows. The 15% Amylose/ESF chow was supplemented with ESF at saponin levels equal to that in unfractionated SPI. Values are technical replicates and results are expressed as soyasaponin I equivalents. The data (n = 3) are shown as mean ± SD. Significant differences between treatment groups were identified using one-way ANOVA followed with Tukey’s multiple comparison test. Different letters denote significant (P < 0.05) difference between treatments.
When the saponin content in mouse chows was compared, the saponin level in the diets corresponded closely with the efficacy of the diet to reduce lung nodule counts, in that the diets with SPI and ESF had comparable, high levels of saponins (Fig. 3). It is thus possible that soy saponins, rather than isoflavones, may be the bioactive compounds contributing to the chemopreventive activity of the SPI.
Soy saponins reduce NNK-induced lung cancer by increasing plasma isoflavone levels
To confirm that saponins were responsible for the superior effects of SPI, we compared diets containing SPI, casein + saponins, casein + isoflavones, and casein + saponins + isoflavones for their ability to lower the number of NNK-induced lung nodules. As shown in Fig. 4A, B, only the casein supplemented with both saponins and isoflavones significantly reduced the number of lung nodules, compared to casein alone. This suggested these two ethanol-soluble components worked together to lower lung cancer nodules.
(A) Soy saponins plus soy isoflavones significantly reduce NNK-induced lung nodules. The data (n = 5–12) are biological replicates and shown as the mean ± SEM. Significant differences between treatment groups and the 15% Amylose group were identified using one-way ANOVA followed with Dunnett’s multiple comparison test. Asterisks denote a significant difference between mice fed the different diets, *P ≤ 0.05, ** P ≤ 0.01. (B) Representative H&E images of the tumors in the lung from groups that were significantly impacted by the diet, relative to the 15%Amylose. Images were taken using Olympus BX46 microscope at 100 × magnification. (C) Soy saponins increase plasma levels of genistein in mice. The data (n = 5–12) are biological replicates and shown as the mean ± SEM. Significant differences between treatment groups were identified using one-way ANOVA followed with Tukey multiple comparison test. Groups not sharing the same letter denote significant (P < 0.05) differences between treatments. (D) Representative mass spectra of genistein (m/z 271.13, top panel) and its fragments (bottom panel).
To determine how these two agents worked together to lower NNK-induced lung nodules, we postulated that perhaps the saponins, given their amphipathic “soapy” nature, might be loosening the tight junctions between colonocytes and/or damaging the colonocytes47, allowing more isoflavones to enter the circulation. To test this, we quantified the level of the isoflavone, genistein, in mice fed 15% Amylose/SPI, 15% Amylose/isoflavones and 15% Amylose/saponins + isoflavones, and found that the mice on the combination of saponins and isoflavones had significantly higher genistein levels than the mice on the other two diets (Fig. 4C). We also measured the levels of genistein in the plasma of mice fed either casein (15% Amylose) or casein + saponins (15% Amylose/saponins) as negative controls and, as expected, found no detectable genistein in their plasmas (Fig. 4C). Representative mass spectra of genistein and its fragments are depicted in Fig. 4D.
Discussion
Lung cancer continues to be the world’s leading cause of cancer deaths48. Observational studies, however, suggest that soy foods may be helpful in preventing lung cancer since its consumption is inversely correlated with lung cancer incidence and mortality49,50. The anti-cancer properties of soy have also been demonstrated in several animal models3,9,10, which show that soy is superior to other protein sources in slowing cancer growth3,4,5,6,7,8. We recently confirmed the anti-cancer properties of soy by showing that low CHO diets containing SPI were significantly more effective than the same low CHO diets containing casein, a milk protein commonly formulated in standard rodent chow, in preventing lung tumors in cigarette carcinogen-injected A/J mice41. Despite the numerous reports of the anti-cancer properties of soy, the component(s) responsible for this activity has not been completely elucidated. In the current study, we used the NNK-induced lung cancer mouse model to try and identify these components.
One of the soy components thought to contribute to its anti-cancer properties is the isoflavones, which is a family of phytoestrogenic compounds naturally occurring in soy. While genistin, daidzin and glycetin, which are the glycosylated versions of genistein, daidzein, and glycetein, respectively, are the most commonly found isoflavones in soy foods, they typically are hydrolyzed to their aglycones prior to absorption34,51. These aglycones are typically comparable or more anti-proliferative than their glycosylated counterparts, likely due to differences in their water solubility52,53. Genistein, typically the most abundant isoflavone, for example, has been shown to suppress non-small-cell lung cancer progression in a xenograft model54. There are also multiple reports suggesting the efficacy of isoflavones in slowing the growth of breast55, colon56, and bladder57 cancers in mice. In a meta-analysis that found a reduced risk of cancer incidence with higher consumption of soy foods, the intake of isoflavones was inversely associated with cancer incidence58. We were thus surprised when we found that isoflavones, added to a standard low CHO diet containing casein, at a comparable level to that found in SPI, had no impact on lung tumorigenesis.
There have also been reports suggesting that the protein component, and not the isoflavones, is the primary bioactive agent within soy foods59. Lunasin, for example, is a bioactive peptide derived from the digestion of soy protein that has been shown to have anti-proliferative effects against lung cancer cell lines in vitro and in vivo23,60. BBIs are another family of proteins present in soy that has been reported to have anti-cancer effects61. We show here that isoflavone-poor-soy protein was ineffective at preventing NNK-induced lung tumor formation. This rules out the possibility that soy proteins or their breakdown products such as lunasin, BBIs, or other bioactive peptides are significantly chemopreventive, at least in this model system.
Since the amino acid profile of soy protein is distinct from casein, and different amino acids could play different roles in cancer development, we decided to test if this difference in amino acid profile could contribute to the anti-cancer effects of soy. We, however, found that the low CHO diets made with a set of amino acids corresponding to that in casein or soy protein made no difference in lung tumor numbers. This further confirmed that the chemopreventive activity of soy is unlikely attributable to the soy protein within the SPI.
Saponins are co-extracted with the isoflavones during the ethanol washing of SPI. Soyasaponins are naturally occurring triterpenoid glycosides with an oleanane-type aglycone that are structurally diverse depending on the number, position and type of polysaccharides attached to the aglycone backbone62. The two major types of saponins are the group A soyasaponins, which are glycosylated at C-3 and C-22 positions, and group B soyasaponins, which are glycosylated or conjugated to a 2,3-dihydro-2,5-dihydroxy-6-methyl-4-pyrone (DDMP) moiety at the C-3 position 44. Saponins are amphiphilic due to the presence of the lipid-soluble aglycone and the water-soluble polysaccharide chain, giving them a soap-like property63. These soy saponins have recently been reported to have anti-proliferative effects37,39,44. When saponins were added to our low CHO diet, however, we saw no evidence of it being effective in reducing the development of lung nodules in our A/J mice. However, when the saponins were combined with isoflavones at levels present in SPI, they were almost as effective as SPI. This is consistent with saponins + isoflavones being the major contributors responsible for the ability of SPI to reduce NNK-induced lung nodules. The fact that the combination of isoflavones and saponins are effective at reducing lung nodule numbers, but not when they are present independently, could also explain a number of publications that reported a greater efficacy of SPI or concentrate when compared to pure isoflavones57,64,65. Our finding that this combination was not quite as effective as unfractionated SPI, could suggest that there are as of yet unidentified components within SPI that contribute to its efficacy in reducing NNK-induced lung nodules.
Since saponins are detergent-like molecules that can permeabilize cell membranes, we hypothesized during this study that the soy saponins might be enhancing the absorption of isoflavones through the gastrointestinal barrier, which leads to higher circulating isoflavone levels. We showed that this was indeed the case, suggesting that these soy saponins appear to act, at least in part, by increasing plasma levels of isoflavones. It is also possible that the saponins and the isoflavones work to prevent lung tumour formation via a synergistic mechanism that we have not currently identified.
Since genistein has previously been shown to be oxidized by CYP4501A2, a cytochrome P450 reported to be involved in converting the pro-carcinogen NNK to its active metabolites, it is conceivable that genistein may help reduce the formation of lung nodule formation by interfering with the biotransformation of NNK66,67. However, future work is needed to confirm this potential mechanism of action. To conclude, we have identified the combination of isoflavones and saponins as the primary components of SPI that contribute to the efficacy of soy in preventing cigarette carcinogen-induced lung tumor formation. However, we cannot completely rule out that other components within the SPI contribute to the lowering of NNK-induced lung nodules in our mouse model of lung cancer.
Data availability
Reasonable requests for data can be made to the corresponding author.
References
Trock, B. J., Hilakivi-Clarke, L. & Clarke, R. Meta-analysis of soy intake and breast cancer risk. J. Natl. Cancer Inst. 98, 459–471. https://doi.org/10.1093/jnci/djj102 (2006).
Wei, Y. et al. Soy intake and breast cancer risk: a prospective study of 300,000 Chinese women and a dose-response meta-analysis. Eur. J. Epidemiol. 35, 567–578. https://doi.org/10.1007/s10654-019-00585-4 (2020).
Mercer, K. E., Pulliam, C. F., Pedersen, K. B., Hennings, L. & Ronis, M. J. Soy protein isolate inhibits hepatic tumor promotion in mice fed a high-fat liquid diet. Exp. Biol. Med. 242, 635–644. https://doi.org/10.1177/1535370216685436 (2017).
Taha, A. A. A. et al. The effect of the type of dietary protein on the development of ovarian cancer. Oncotarget 9, 23987–23999. https://doi.org/10.18632/oncotarget.25253 (2018).
Robles-Ramirez Mdel, C., Ramon-Gallegos, E., Reyes-Duarte, F. J. & Mora-Escobedo, R. Effect of germinated soy protein on the growth of HeLa cervical cancer cells in female athymic mice. Nutr. Cancer 64, 1261–1268 (2012).
Hakkak, R., Korourian, S., Ronis, M. J., Johnston, J. M. & Badger, T. M. Soy protein isolate consumption protects against azoxymethane-induced colon tumors in male rats. Cancer Lett. 166, 27–32. https://doi.org/10.1016/s0304-3835(01)00441-4 (2001).
Simmen, R. C. et al. Inhibition of NMU-induced mammary tumorigenesis by dietary soy. Cancer Lett. 224, 45–52. https://doi.org/10.1016/j.canlet.2004.11.009 (2005).
Zhou, J. R. et al. Inhibition of orthotopic growth and metastasis of androgen-sensitive human prostate tumors in mice by bioactive soybean components. Prostate 53, 143–153. https://doi.org/10.1002/pros.10141 (2002).
Hakkak, R. et al. Diets containing whey proteins or soy protein isolate protect against 7,12-dimethylbenz(a)anthracene-induced mammary tumors in female rats. Cancer Epidemiol. Biomark. Prev. 9, 113–117 (2000).
Appelt, L. C. & Reicks, M. M. Soy induces phase II enzymes but does not inhibit dimethylbenz[a]anthracene-induced carcinogenesis in female rats. J. Nutr. 129, 1820–1826. https://doi.org/10.1093/jn/129.10.1820 (1999).
Aronson, W. J. et al. Decreased growth of human prostate LNCaP tumors in SCID mice fed a low-fat, soy protein diet with isoflavones. Nutr. Cancer 35, 130–136. https://doi.org/10.1207/S15327914NC352_6 (1999).
Cid-Gallegos, M. S., Corzo-Rios, L. J., Jimenez-Martinez, C. & Sanchez-Chino, X. M. Protease inhibitors from plants as therapeutic agents- a review. Plant Foods Hum. Nutr. 77, 20–29. https://doi.org/10.1007/s11130-022-00949-4 (2022).
Kennedy, A. R. The Bowman-Birk inhibitor from soybeans as an anticarcinogenic agent. Am. J. Clin. Nutr. 68, 1406S-1412S. https://doi.org/10.1093/ajcn/68.6.1406S (1998).
Wan, X. S. et al. Treatment with soybean-derived Bowman Birk inhibitor increases serum prostate-specific antigen concentration while suppressing growth of human prostate cancer xenografts in nude mice. Prostate 41, 243–252 (1999).
Armstrong, W. B. et al. Bowman Birk inhibitor concentrate and oral leukoplakia: a randomized phase IIb trial. Cancer Prev. Res. (Phila) 6, 410–418. https://doi.org/10.1158/1940-6207.CAPR-13-0004 (2013).
Robles-Ramirez Mdel, C., Ramón-Gallegos, E., Mora-Escobedo, R. & Torres-Torres, N. A peptide fraction from germinated soybean protein down-regulates PTTG1 and TOP2A mRNA expression, inducing apoptosis in cervical cancer cells. J. Exp. Ther. Oncol. 9, 255–263 (2012).
Gonzalez-Montoya, M., Hernandez-Ledesma, B., Silvan, J. M., Mora-Escobedo, R. & Martinez-Villaluenga, C. Peptides derived from in vitro gastrointestinal digestion of germinated soybean proteins inhibit human colon cancer cells proliferation and inflammation. Food Chem. 242, 75–82. https://doi.org/10.1016/j.foodchem.2017.09.035 (2018).
Wan, X. et al. Lunasin: a promising polypeptide for the prevention and treatment of cancer. Oncol. Lett. 13, 3997–4001. https://doi.org/10.3892/ol.2017.6017 (2017).
Pabona, J. M. et al. The soybean peptide lunasin promotes apoptosis of mammary epithelial cells via induction of tumor suppressor PTEN: similarities and distinct actions from soy isoflavone genistein. Genes Nutr. 8, 79–90 (2013).
Hsieh, C. C., Hernandez-Ledesma, B. & de Lumen, B. O. Lunasin, a novel seed peptide, sensitizes human breast cancer MDA-MB-231 cells to aspirin-arrested cell cycle and induced apoptosis. Chem. Biol. Interact. 186, 127–134. https://doi.org/10.1016/j.cbi.2010.04.027 (2010).
Dia, V. P. & Mejia, E. G. Lunasin promotes apoptosis in human colon cancer cells by mitochondrial pathway activation and induction of nuclear clusterin expression. Cancer Lett. 295, 44–53. https://doi.org/10.1016/j.canlet.2010.02.010 (2010).
Dia, V. P. & Gonzalez de Mejia, E. Lunasin induces apoptosis and modifies the expression of genes associated with extracellular matrix and cell adhesion in human metastatic colon cancer cells. Mol. Nutr. Food Res. 55, 623–634. https://doi.org/10.1002/mnfr.201000419 (2011).
McConnell, E. J., Devapatla, B., Yaddanapudi, K. & Davis, K. R. The soybean-derived peptide lunasin inhibits non-small cell lung cancer cell proliferation by suppressing phosphorylation of the retinoblastoma protein. Oncotarget 6, 4649–4662. https://doi.org/10.18632/oncotarget.3080 (2015).
Inaba, J., McConnell, E. J. & Davis, K. R. Lunasin sensitivity in non-small cell lung cancer cells is linked to suppression of integrin signaling and changes in histone acetylation. Int. J. Mol. Sci. 15, 23705–23724. https://doi.org/10.3390/ijms151223705 (2014).
de Mejia, E. G., Wang, W. & Dia, V. P. Lunasin, with an arginine-glycine-aspartic acid motif, causes apoptosis to L1210 leukemia cells by activation of caspase-3. Mol. Nutr. Food Res. 54, 406–414. https://doi.org/10.1002/mnfr.200900073 (2010).
Galvez, A. F., Chen, N., Macasieb, J. & de Lumen, B. O. Chemopreventive property of a soybean peptide (lunasin) that binds to deacetylated histones and inhibits acetylation. Cancer Res. 61, 7473–7478 (2001).
Hsieh, C. C., Hernandez-Ledesma, B., Jeong, H. J., Park, J. H. & de Lumen, B. O. Complementary roles in cancer prevention: protease inhibitor makes the cancer preventive peptide lunasin bioavailable. PLoS ONE 5, e8890. https://doi.org/10.1371/journal.pone.0008890 (2010).
Jeong, H. J., Jeong, J. B., Kim, D. S. & de Lumen, B. O. Inhibition of core histone acetylation by the cancer preventive peptide lunasin. J. Agric. Food Chem. 55, 632–637. https://doi.org/10.1021/jf062405u (2007).
Hernández-Ledesma, B., Hsieh, C.-C. & de Lumen, B. O. Relationship between lunasin’s sequence and its inhibitory activity of histones H3 and H4 acetylation. Mol. Nutr. Food Res. 55, 989–998. https://doi.org/10.1002/mnfr.201000632 (2011).
Gorissen, S. H. M. et al. Protein content and amino acid composition of commercially available plant-based protein isolates. Amino Acids 50, 1685–1695. https://doi.org/10.1007/s00726-018-2640-5 (2018).
Wanders, D., Hobson, K. & Ji, X. Methionine restriction and cancer biology. Nutrients 12, 684. https://doi.org/10.3390/nu12030684 (2020).
Messina, M. J. & Wood, C. E. Soy isoflavones, estrogen therapy, and breast cancer risk: analysis and commentary. Nutr. J. 7, 17. https://doi.org/10.1186/1475-2891-7-17 (2008).
Pabich, M. & Materska, M. Biological effect of soy isoflavones in the prevention of civilization diseases. Nutrients 11, 1660. https://doi.org/10.3390/nu11071660 (2019).
Peñalvo, J. L., Nurmi, T. & Adlercreutz, H. A simplified HPLC method for total isoflavones in soy products. Food Chem. 87, 297–305. https://doi.org/10.1016/j.foodchem.2003.12.028 (2004).
Setchell, K. D. & Clerici, C. Equol: history, chemistry, and formation. J. Nutr. 140, 1355S-1362S. https://doi.org/10.3945/jn.109.119776 (2010).
Shi, J. et al. Saponins from edible legumes: chemistry, processing, and health benefits. J. Med. Food 7, 67–78. https://doi.org/10.1089/109662004322984734 (2004).
Huang, S., Huang, P., Wu, H., Wang, S. & Liu, G. Soyasaponin Ag inhibits triple-negative breast cancer progression via targeting the DUSP6/MAPK signaling. Folia Histochem. Cytobiol. 59, 291–301. https://doi.org/10.5603/FHC.a2021.0029 (2021).
Rowlands, J. C., Berhow, M. A. & Badger, T. M. Estrogenic and antiproliferative properties of soy sapogenols in human breast cancer cells in vitro. Food Chem. Toxicol. 40, 1767–1774. https://doi.org/10.1016/s0278-6915(02)00181-3 (2002).
Zhang, W. & Popovich, D. G. Effect of soyasapogenol A and soyasapogenol B concentrated extracts on HEP-G2 cell proliferation and apoptosis. J. Agric. Food Chem. 56, 2603–2608. https://doi.org/10.1021/jf0731550 (2008).
Guo, Y. W., Chen, Y. H., Chiu, W. C., Liao, H. & Lin, S. H. Soy saponins meditate the progression of colon cancer in rats by inhibiting the activity of beta -glucuronidase and the number of aberrant crypt foci but not cyclooxygenase-2 activity. ISRN Oncol. 2013, 645817. https://doi.org/10.1155/2013/645817 (2013).
Elisia, I. et al. The effect of diet and exercise on tobacco carcinogen-induced lung cancer. Carcinogenesis 40, 448–460. https://doi.org/10.1093/carcin/bgz060 (2019).
Hintze, K. J., Benninghoff, A. D. & Ward, R. E. Formulation of the total Western diet (TWD) as a basal diet for rodent cancer studies. J. Agric. Food Chem. 60, 6736–6742. https://doi.org/10.1021/jf204509a (2012).
Fukui, K., Tachibana, N., Fukuda, Y., Takamatsu, K. & Sugano, M. Ethanol washing does not attenuate the hypocholesterolemic potential of soy protein. Nutrition 20, 984–990. https://doi.org/10.1016/j.nut.2004.08.011 (2004).
Zhang, W. & Popovich, D. G. Chemical and biological characterization of oleanane triterpenoids from soy. Molecules 14, 2959–2975. https://doi.org/10.3390/molecules14082959 (2009).
Le, A. V., Parks, S. E., Nguyen, M. H. & Roach, P. D. Improving the vanillin-sulphuric acid method for quantifying total saponins. Technologies 6, 84. https://doi.org/10.3390/technologies6030084 (2018).
Twaddle, N. C., Churchwell, M. I. & Doerge, D. R. High-throughput quantification of soy isoflavones in human and rodent blood using liquid chromatography with electrospray mass spectrometry and tandem mass spectrometry detection. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 777, 139–145. https://doi.org/10.1016/s1570-0232(02)00275-1 (2002).
Tsai, C. Y., Chen, Y. H., Chien, Y. W., Huang, W. H. & Lin, S. H. Effect of soy saponin on the growth of human colon cancer cells. World J. Gastroenterol. 16, 3371–3376. https://doi.org/10.3748/wjg.v16.i27.3371 (2010).
Sung, H. et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 71, 209–249. https://doi.org/10.3322/caac.21660 (2021).
Nachvak, S. M. et al. Soy, soy isoflavones, and protein intake in relation to mortality from all causes, cancers, and cardiovascular diseases: a systematic review and dose-response meta-analysis of prospective cohort studies. J. Acad. Nutr. Diet 119, 1483-1500.e17. https://doi.org/10.1016/j.jand.2019.04.011 (2019).
Yang, G. et al. Soy food intake and risk of lung cancer: evidence from the Shanghai Women’s Health Study and a meta-analysis. Am. J. Epidemiol. 176, 846–855. https://doi.org/10.1093/aje/kws168 (2012).
Křížová, L., Dadáková, K., Kašparovská, J. & Kašparovský, T. Isoflavones. Molecules 24, 1076. https://doi.org/10.3390/molecules24061076 (2019).
Zhu, Y., Yao, Y., Shi, Z., Everaert, N. & Ren, G. Synergistic effect of bioactive anticarcinogens from soybean on anti-proliferative activity in MDA-MB-231 and MCF-7 human breast cancer cells in vitro. Molecules 23, 1557. https://doi.org/10.3390/molecules23071557 (2018).
Yanagihara, K., Ito, A., Toge, T. & Numoto, M. Antiproliferative effects of isoflavones on human cancer cell lines established from the gastrointestinal tract. Cancer Res. 53, 5815–5821 (1993).
Yu, Y. et al. Soy isoflavone genistein inhibits hsa_circ_0031250/miR-873-5p/FOXM1 axis to suppress non-small-cell lung cancer progression. IUBMB Life 73, 92–107. https://doi.org/10.1002/iub.2404 (2021).
Sharma, M. et al. Therapeutic effects of dietary soybean genistein on triple-negative breast cancer via regulation of epigenetic mechanisms. Nutrients 13, 3944. https://doi.org/10.3390/nu13113944 (2021).
Xiao, X. et al. Genistein suppresses FLT4 and inhibits human colorectal cancer metastasis. Oncotarget 6, 3225–3239. https://doi.org/10.18632/oncotarget.3064 (2015).
Singh, A. V., Franke, A. A., Blackburn, G. L. & Zhou, J. R. Soy phytochemicals prevent orthotopic growth and metastasis of bladder cancer in mice by alterations of cancer cell proliferation and apoptosis and tumor angiogenesis. Cancer Res. 66, 1851–1858. https://doi.org/10.1158/0008-5472.Can-05-1332 (2006).
Fan, Y. et al. Intake of soy, soy isoflavones and soy protein and risk of cancer incidence and mortality. Front. Nutr. 9, 847421. https://doi.org/10.3389/fnut.2022.847421 (2022).
Chiesa, G. et al. Reduced mammary tumor progression in a transgenic mouse model fed an isoflavone-poor soy protein concentrate. Mol. Nutr. Food Res. 52, 1121–1129. https://doi.org/10.1002/mnfr.200700296 (2008).
Devapatla, B., Shidal, C., Yaddanapudi, K. & Davis, K. R. Validation of syngeneic mouse models of melanoma and non-small cell lung cancer for investigating the anticancer effects of the soy-derived peptide lunasin. F1000Research 5, 2432. https://doi.org/10.12688/f1000research.9661.2 (2016).
Witschi, H. & Espiritu, I. Development of tobacco smoke-induced lung tumors in mice fed Bowman-Birk protease inhibitor concentrate (BBIC). Cancer Lett. 183, 141–146. https://doi.org/10.1016/s0304-3835(02)00156-8 (2002).
Kamo, S., Suzuki, S. & Sato, T. The content of soyasaponin and soyasapogenol in soy foods and their estimated intake in the Japanese. Food Sci. Nutr. 2, 289–297. https://doi.org/10.1002/fsn3.107 (2014).
Timilsena, Y. P., Phosanam, A. & Stockmann, R. Perspectives on saponins: food functionality and applications. Int. J. Mol. Sci. https://doi.org/10.3390/ijms241713538 (2023).
Mercer, K. E. et al. Diet supplementation with soy protein isolate, but not the isoflavone genistein, protects against alcohol-induced tumor progression in DEN-treated male mice. Adv. Exp. Med. Biol. 1032, 115–126. https://doi.org/10.1007/978-3-319-98788-0_9 (2018).
Hewitt, A. L. & Singletary, K. W. Soy extract inhibits mammary adenocarcinoma growth in a syngeneic mouse model. Cancer Lett. 192, 133–143. https://doi.org/10.1016/S0304-3835(02)00712-7 (2003).
Smith, T. J., Guo, Z., Guengerich, F. P. & Yang, C. S. Metabolism of 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone (NNK) by human cytochrome P450 1A2 and its inhibition by phenethyl isothiocyanate. Carcinogenesis 17, 809–813. https://doi.org/10.1093/carcin/17.4.809 (1996).
Nagayoshi, H. et al. Oxidation of naringenin, apigenin, and genistein by human family 1 cytochrome P450 enzymes and comparison of interaction of apigenin with human P450 1B1.1 and Scutellaria P450 82D.1. Chem. Res. Toxicol. 36, 1778–1788. https://doi.org/10.1021/acs.chemrestox.3c00229 (2023).
Acknowledgements
This work was supported by the Lotte and John Hecht Memorial Foundation [Grant ID #4340 to GK].
Author information
Authors and Affiliations
Contributions
IE and GK conceived and designed the study, interpreted data and wrote the manuscript. IE, MY, SK, AW, CD, SW, VC, HA, acquired and analyzed data, and revised the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Elisia, I., Yeung, M., Kowalski, S. et al. Saponins in soy reduce NNK-induced lung cancer by increasing plasma isoflavone levels. Sci Rep 15, 12714 (2025). https://doi.org/10.1038/s41598-025-97687-9
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-97687-9