Abstract
This study aimed to investigate the association between serum uric acid level before gestational age of 20 weeks and preeclampsia. This study included pregnant women of three hospitals from January 2018 to June 2024. Clinical data were extracted using electronic medical record systems. The exposure factor was serum uric acid measured before gestational age of 20 weeks, with the primary outcome being preeclampsia, and secondary outcomes being preterm birth. Smooth curve fitting, ROC analysis, threshold effects, multivariate logistic regression, and subgroup analysis were employed to examine the relationship between uric acid and preeclampsia. The Kaplan-Meier method and log-rank test were used to evaluate the impact of serum uric acid on the gestational age at delivery. A total of 44,609 singleton pregnancies were included. There was a nonlinear relationship between serum uric acid level and the risk of preeclampsia, with a turning point at a uric acid level of 240 µmol/L. After adjusting for confounders, compared to non-preeclampsia cases, the risk of preeclampsia increased 1.38 times (95% CI: 1.28-1.48) for uric acid level between 240~360 µmol/L and 2.14 times (95% CI: 1.61-2.85) for UA levels ≥ 360 µmol/L. Similar positive associations were observed between uric acid level and preterm birth. Subgroup analysis maintained this positive correlation. Interaction tests indicated that BMI might influence the strength of the association between uric acid and preeclampsia (P < 0.05). Elevated serum uric acid level before gestational age of 20 weeks increase the risk of preeclampsia. Elevated uric acid levels in early pregnancy are associated with an increased risk of preeclampsia, suggesting that monitoring uric acid may help identify women at higher risk.
Similar content being viewed by others
Introduction
Preeclampsia (PE) is a severe complication of pregnancy, accounting for 10% to 15% of maternal deaths globally, posing a significant threat to maternal and neonatal health1. The hallmark symptoms of PE include new-onset hypertension, proteinuria, or multi-organ dysfunction occurring after the 20th week of gestation2. While the exact etiology of PE remains unclear, inadequate trophoblast invasion, impaired remodeling of placental spiral arteries, and placental dysfunction are considered primary causes3. Currently, there is no effective cure for PE; once diagnosed, the only definitive treatment is termination of the pregnancy. Therefore, identifying serum markers that enhance the predictive power for PE risk is clinically significant for its prediction and management.
Uric acid (UA) is not only the end product of exogenous purine metabolism but also its serum concentration can be increased by diets high in purines or other factors that induce purine nucleotide degradation, such as alcohol and fructose intake4. Our previous single-center cohort study indicated that elevated serum UA at 20 weeks of gestation may be a causative factor for PE, with particularly strong evidence for measurements taken between 8 and 12 weeks of pregnancy5. Several other studies have reported similar findings. One study demonstrated that increased UA levels before 10 weeks of gestation were linked to a higher risk of PE6. Another two studies found that elevated UA levels before 18 weeks7 and before 20 weeks8 of gestation were independent risk factors for PE. However, inconsistent results have also been reported. One prospective cohort study showed no significant difference in UA levels between women who developed PE and those who did not during early and mid-pregnancy. Serum UA levels rose significantly only after clinical symptoms of PE appeared9. The inconsistency across studies may stem from limited sample sizes or insufficient adjustment for confounding variables. Another important possible explanation is that most prior studies focused on the timing of UA elevation during pregnancy, without adequately considering that the degree of elevation itself might be a key driver of the observed associations with PE. To date, few studies have systematically examined the relationship between stratified UA levels and the risk of PE. Therefore, further investigation into how varying concentrations of UA before gestational age of 20 weeks influence PE risk may help clarify its potential predictive value and provide new insights for early identification and intervention.
To address these gaps, this study categorized pregnant women based on UA levels to explore the association between different concentrations of UA before gestational age of 20 weeks and the risk of PE. Using multicenter clinical data with a large sample size, we applied smoothed curve fitting and threshold effect analysis to assess the dose-response relationship between UA and PE. Multivariable logistic regression was conducted to evaluate the independent association between UA levels and PE risk. Subgroup analyses were also performed to examine whether age, body mass index (BMI), or parity modified the observed association. Our findings may support risk stratification for PE based on UA levels before gestational age of 20 weeks. Interventions targeting UA reduction could potentially serve as preventive strategies against PE and related adverse pregnancy outcomes, offering valuable reference for clinical practice.
Methods
Design and participants
This multicenter retrospective cohort study included 44,609 singleton pregnancies from three hospitals—Obstetrics and Gynecology Hospital of Fudan University, the First People’s Hospital of Chenzhou, and Wuxi Maternal and Child Health Care Hospital—between January 2018 and June 2024. Of these, 2,444 women were diagnosed with PE, and 42,165 were non-cases. All pregnant women established their prenatal records and delivered at one of the above three hospitals. Clinical data were obtained from the hospital information system (HIS). The study included women meeting the following criteria: (1) age of 18 years or older, (2) singleton pregnancy, and (3) receipt of prenatal care and delivery at one of the participating hospitals. Exclusion criteria were: (1) initial UA measurement obtained after 20 weeks of gestation, (2) multiple pregnancies, (3) pre-existing hypertension, (4) pre-pregnancy diagnosis of conditions such as type 1 or type 2 diabetes, hyperlipidemia, or metabolic syndrome, as well as known kidney disease, heart disease, or other chronic medical or surgical conditions, and (5) incomplete clinical or laboratory records. Complete maternal and infant records should include basic demographic characteristics of the mother (such as height, weight, blood pressure, education level, and past medical history), laboratory test results, as well as delivery data covering gestational age at birth and newborn weight. This study was approved by the ethics committees of three above Hospital. All participants provided broad informed consent at the time of registration. This study adheres to the principles outlined in the Declaration of Helsinki.
Variables and measurements
In this study, the first serum UA measured before gestational age of 20 weeks was used as the exposure factor. Venous blood samples were collected during the early pregnancy visits and UA levels were analyzed using an automated biochemical analyzer. In the nonpregnant population, hyperuricemia is usually defined as a serum UA level higher than 7.0 mg/dl in men and 6.0 mg/dl in women. In pregnant women, UA levels are 25%~35% lower than in nonpregnant women, and serum UA levels usually fall below 4 mg/dl (1 mg/dl = 60 µmol/l)10,11.
Smooth curve fitting indicated that 240 µmol/L was the inflection point in the association between uric acid levels and the risk of PE. Although Receiver Operating Characteristic (ROC) curve analysis suggested an optimal cutoff of 216 µmol/L(Supplementary Table S1), threshold effect analysis further supported the appropriateness of 240 µmol/L as the inflection point. In conjunction with previous studies showing that hyperuricemia in non-pregnant women is typically defined as serum uric acid > 360 µmol/L and that uric acid levels during pregnancy are generally below 240 µmol/L10,12, we categorized uric acid levels into three groups: < 240 µmol/L, 240~360 µmol/L, and ≥ 360 µmol/L. Additional stratified analyses were conducted per 50 µmol/L and per 100 µmol/L increase in uric acid levels to comprehensively evaluate its association with PE.
Based on previous research and clinical experience13,14, we summarized the following covariates that may influence the relationship between UA and PE. The covariates included in this study are: age (years), BMI (kg/m2), systolic pressure, diastolic pressure, use of aspirin, use of antihypertensives, family history of hypertension, tobacco, alcohol consumption, in vitro fertilization (IVF), parity, alanine transaminase (U/L), total cholesterol (mmol/L), triglycerides (mmol/L), fasting blood glucose (mmol/L), test week, and adverse pregnancy history. All covariates were collected during the first prenatal visit, with the exact gestational week of blood collection recorded as 11.42 ± 3.84 weeks. Age was categorized into normal (< 35 years) and advanced maternal age (≥35 years). BMI was classified as underweight/normal (< 24 kg/m2) and overweight (≥ 24 kg/m2). Supplementary Table S2 showed the definition of variables.
Outcomes and measurements
The primary outcome measure was PE. According to the 2020 edition of the American College of Obstetricians and Gynecologists (ACOG) guidelines15, the diagnostic criteria for PE include a normal pre-pregnancy blood pressure, followed by the onset of systolic blood pressure ≥ 140 mmHg or diastolic blood pressure ≥ 90 mmHg after 20 weeks of gestation, accompanied by a urine dipstick protein level of 2+ or greater, or a 24-hour urinary protein excretion exceeding 300 mg. Alternatively, the diagnosis can be made in the absence of proteinuria if any of the following conditions are present: platelet count < 100 × 109/L, liver function impairment (serum transaminases more than twice the upper limit of normal), renal impairment (serum creatinine > 1.1 mg/dL or more than twice the upper limit of normal), pulmonary edema, new-onset headache not attributable to another condition, or visual disturbances.
The secondary outcome was preterm birth (delivery before 37 weeks of gestation), with gestational age determined by ultrasound before gestational age of 20 weeks.
Statistical analysis
Continuous variables are expressed as means and standard deviations (SD), while categorical variables are presented as percentages (%). The statistical analysis includes four main steps to comprehensively analyze the relationship between UA levels and the primary outcome of PE, as well as the secondary outcome of preterm birth. Firstly, participants were divided into PE and non-PE groups according to clinical guidelines. Baseline covariate differences between the two groups were tested using chi-square tests for categorical variables and Student’ s t-tests for continuous variables. Secondly, smooth curve fitting plots were utilized to explore the shape of the relationship between UA and PE. ROC analysis was performed to determine the optimal cutoff value for the association between uric acid levels and PE. Threshold effect analysis was conducted to confirm the nonlinear relationship between UA and PE. Log-likelihood ratio tests were performed on single-segment linear regression models and double-segment linear models, comparing segmented regression model I and model II. A P-value < 0.05 indicates a significant non-linear relationship. Thirdly, multivariate logistic regression models were used to test the association between UA and PE in both models. Model I did not adjust for any covariates. Model II adjusted for age, BMI, systolic pressure, diastolic pressure, aspirin use, depressor use, hypertension history, tobacco, alcohol, IVF, parity, alanine transaminase, total cholesterol, triglycerides, fasting blood glucose, gestational week of the test, and adverse pregnancy history. UA was categorized into three levels: < 240 µmol/L, 240~360 µmol/L, and ≥ 360 µmol/L. Stratified analyses were conducted for each 50 µmol/L and 100 µmol/L increase in UA levels. Fourthly, the impact of serum UA on gestational age at delivery was assessed using the Kaplan-Meier method and log-rank test. To evaluate the robustness of the relationship between UA and PE, subgroup analyses were performed based on age, BMI, and parity to test the effect of different subgroups on the outcomes, as these are well-established risk factors for PE according to previous studies16. Interaction tests were used to detect potential heterogeneity among these subgroups, with a p-value > 0.05 indicating no significant heterogeneity.
All reported p-values are two-tailed. Software IBM SPSS (version 21.0. IBM; Armonk, NY) and the R statistical packages (R Foundation; https://www.r.project.org; version 4.4.1) were used for statistical analysis.
Results
Baseline characteristics
As shown in Table 1, the study included a total of 44,609 singleton pregnant women, consisting of 2,444 PE and 42,165 non-PE. Significant differences (P < 0.05) were observed between the PE group and the non-PE group regarding age, BMI, UA levels, systolic pressure, diastolic pressure, alanine transaminase, total cholesterol, triglycerides, fasting blood glucose, use of aspirin, use of antihypertensives, family history of hypertension, alcohol consumption, IVF, and parity. No significant differences (P > 0.05) were found in terms of the gestational week of the test, smoking, and adverse pregnancy history.
Identification of non-linear relationship
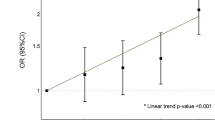
As shown in Figure 1 by the results of the generalized additive model (GAM) and smooth curve fitting, there exists a non-linear association between UA and PE. The segmented regression model results in Table 2 indicate that the turning point in the relationship between UA levels and PE is 240 μmol/L. When UA is < 240 μmol/L, every increase of 100 μmol/L in UA is associated with an 81% increase in the risk of PE (OR = 1.81, 95% CI: 1.55–2.11.55.11). When UA is ≥ 240 μmol/L, every increase of 100 μmol/L in UA is associated with a 154% increase in the risk of PE (OR = 2.54, 95% CI: 2.24–2.88.24.88). The log-likelihood ratio test showed P = 0.006, indicating a non-linear relationship between UA and the risk of developing PE.
Smoothed curve fitting reflected the dose-response relationship between UA and PE. The red line represents the fitted curve of UA and PE, and the blue line represents the 95% confidence interval of the curve. UA, uric acid; PE, preeclampsia; BMI, body mass index.
Association between UA and PE
As illustrated in Figure 2, among pregnant women with serum UA levels greater than 240 μmol/L before gestational age of 20 weeks, the incidence of PE reached 8.05%, which is 1.8 times the incidence observed in those with serum UA levels less than 240 μmol/L before gestational age of 20 weeks. Among all cases of PE, 41.04% of the pregnant women had serum UA levels greater than 240 μmol/L before gestational age of 20 weeks.
(A) The incidence of PE in groups defined by serum UA levels ≥240 and < 240 µmol/L, respectively. The y-axis represents the percentage of participants who developed PE in each group. (B) The distribution of serum UA levels (≥240 vs. <240 µmol/L) among all women who developed PE, showing the number and proportion in each category. UA, uric acid; PE, preeclampsia.
As shown in Table 3, UA levels exhibit a significant positive correlation with the primary outcome of PE and the secondary outcome of preterm birth. In Model I, compared to UA levels < 240 μmol/L, the OR for the development of PE was 1.75 (95% CI: 1.61–1.91.61.91) for UA levels in the range of 240~360 μmol/L, and the OR was 6.03 (95% CI: 4.73–7.69.73.69) for UA levels ≥ 360 μmol/L. Similar positive correlations were observed between UA levels and preterm birth. Model II adjusted for age, BMI, systolic pressure, diastolic pressure, aspirin use, depressor use, family history of hypertension, tobacco, alcohol, IVF, parity, alanine transaminase, total cholesterol, triglycerides, fasting blood glucose, gestational week of the test, and adverse pregnancy history. Even after these adjustments, the ORs for PE and preterm birth remained positively correlated with UA levels. Specifically, when UA levels were ≥360 μmol/L, the ORs for PE and preterm birth were 1.74 (95% CI: 1.23–2.48.23.48) and 2.15 (95% CI: 1.46–3.17.46.17), respectively.
Subgroup analysis
As shown in Table 4, a consistent positive correlation between UA and PE was observed across different subgroups, confirming the robustness of the association. No interaction was observed between age, parity, and UA (interaction P > 0.05), suggesting that the correlation between UA and PE is not influenced by age or parity. However, BMI did impact the association between UA and PE (interaction P < 0.05), with pregnant women having a BMI < 24 kg/m2 showing a higher risk of PE at equivalent UA levels compared to those with a BMI ≥ 24 kg/m2. This indicates that BMI may modify the correlation between UA and PE, with underweight or normal-weight pregnant women exhibiting a stronger association.
Kaplan-Meier curves
UA was closely associated with delivery time, and the gestational age at delivery in the higher UA group, especially in the group of UA ≥ 360 μmol/L, was generally earlier (P<0.0001) (Figure 3). The result suggest that elevated serum UA before gestational age of 20 weeks in PE patients significantly increases the cumulative risk of preterm birth.
Kaplan-Meier curve analysis of cumulative risk of preterm birth with different UA levels. The gestational age at delivery in the higher UA group was generally earlier compared to the lower UA group (P<0.0001). UA, uric acid.
Discussion
This study aimed to investigate the dose-response relationship between serum UA level before gestational age of 20 weeks and PE. The results show a significant positive correlation between UA and PE, with a nonlinear association where a UA level of 240 μmol/L acts as a turning point. Subgroup analysis indicates that this association is consistent across different subgroups. Interaction tests reveal that this association is independent of age and parity. Interestingly, compared to pregnant women with a BMI ≥ 24 kg/m2, those with a BMI < 24 kg/m2 had a higher risk of PE at equivalent UA levels.
The relationship between UA and PE has been extensively studied in previous research. Nair et al. reported that the mean serum UA levels were significantly higher in women with PE compared to normotensive pregnant women17. A recent systematic review has concluded that UA levels are elevated in PE patients during early, mid, and late pregnancy, and that UA can serve as a predictor of the severity of PE and related complications18. However, some studies have yielded inconsistent results. One prospective study found no significant difference in serum UA levels between those who did and did not develop PE during early and mid-pregnancy, although elevated serum UA levels in late pregnancy were associated with the onset and severity of PE19. Building upon previous research, our study categorized early pregnancy UA levels and used dose-response analysis to identify a nonlinear relationship between UA and PE, emphasizing the varying degrees of impact different UA levels have on PE. Notably, when early pregnancy UA levels are ≥ 360 μmol/L, the risk of PE significantly increases. This association is unaffected by factors such as age and parity.
Additionally, we found that among pregnant women with serum UA levels greater than 240 μmol/L before gestational age of 20 weeks, the incidence of PE reached 8.05%, which is 1.8 times higher than the incidence observed in those with serum UA levels less than 240 μmol/L. Among all cases of PE, 41.04% of the pregnant women had serum UA levels greater than 240 μmol/L before gestational age of 20 weeks. These cases can be defined as a UA-related PE, which has important implications for the classification and treatment of PE and may become a focus for future research.
Further subgroup analysis yielded interesting results: compared to pregnant women with a BMI ≥ 24 kg/m2, those with a BMI < 24 kg/m2 had a higher risk of PE at equivalent UA levels. This suggests that UA, as a serum biomarker predicting PE, may be even more relevant for underweight or normal-weight pregnant women. However, the underlying mechanisms by which UA influences the age-related aspects of PE remain unclear and require further exploration in future scientific research.
Moreover, we observed that elevated serum UA levels before gestational age of 20 weeks increased the risk of preterm birth, with the risk of preterm birth increasing as UA levels rose. Previous research supports our conclusions. Prior studies have found that high UA levels are associated with preterm birth20, and that elevated UA levels increase the risk of PE19,21. However, our study focuses on UA levels before gestational age of 20 weeks, whereas the timing of UA measurements in these previous studies was mainly during late pregnancy or after the diagnosis of PE.
In normal early pregnancy, due to increased blood volume, the concentration of serum UA tends to decrease relatively. In PE patients, impaired renal function leading to a decreased glomerular filtration rate is considered the cause of elevated UA levels22. Indeed, during the progression of PE, UA plays multiple pathological roles. Previous studies have shown that elevated UA can induce endothelial dysfunction, oxidative stress, and mitochondrial dysfunction23,24, which may represent a potential mechanism by which UA contributes to the development of PE. Additionally, UA reduces nitric oxide production in endothelial cells, leading to impaired trophoblast invasion and resulting in defective remodeling of spiral arteries25. UA can also induce trophoblast shedding, contributing to the occurrence of placental abnormalities26. Combining these mechanistic studies with our findings, elevated UA may be a critical factor in the early pathological processes of the placenta.
Previous studies have consistently demonstrated that both PE and fetal growth restriction (FGR) are major adverse pregnancy outcomes associated with underlying placental insufficiency, often arising from impaired trophoblast invasion and defective remodeling of the uterine spiral arteries during early placentation27,28,29. As such, PE and FGR are widely regarded as clinical manifestations of abnormal placentation and can serve as external indicators of placental dysfunction. However, due to the retrospective nature of this study and limitations in data availability, we were unable to systematically collect FGR diagnoses based on standardized criteria, such as customized fetal growth charts, serial ultrasound assessments of fetal biometry, Doppler velocimetry of the umbilical or middle cerebral arteries, or composite clinical algorithms. This represents a key limitation of our analysis. In the absence of rigorously defined FGR, we cannot disentangle whether the observed associations between maternal uric acid levels and adverse birth outcomes are primarily driven by preterm delivery, true fetal growth pathology, or a combination of both. The use of less specific outcomes such as low birth weight or gestational age at delivery may lead to outcome misclassification and reduce the precision of our estimates. This lack of granularity limits the interpretability of the relationship between early uric acid elevation and distinct placental phenotypes. Future prospective studies incorporating serial fetal growth monitoring and standardized FGR definitions are needed to clarify the specific role of maternal uric acid in different manifestations of placental insufficiency.
Our study has several advantages. Firstly, it included a large sample size of 44,609 singleton pregnant women from multiple centers, providing objective and robust results for accurately assessing the association between early pregnancy UA levels and PE. Secondly, the study employed various statistical methods, including GAM, smooth curve fitting, and threshold effect analysis, which confirmed the non-linear relationship between UA and PE and identified 240 μmol/L as the turning point in this association. Lastly, considering previous research and clinical experience, we accounted for various confounding covariates, thereby minimizing potential biases introduced by these factors. Our study has several limitations that should be considered. Firstly, due to its observational design, we cannot establish causality between UA levels and PE. The observed associations may be influenced by unmeasured confounding factors. Secondly, the study population was selected from retrospective datas, which may introduce selection bias and limit the generalizability of our findings. Moreover, the lack of multiple testing correction in the analyses of secondary outcomes may increase the possibility of type I error. But we believe that the exploratory nature of these analyses mitigates this concern to some extent. Thirdly, UA levels were measured once during early pregnancy, which may not reflect potential changes later in gestation, possibly affecting the accuracy of exposure classification. Lastly, while we found an association between UA and PE, the biological mechanisms underlying this relationship remain unclear, and the clinical utility of UA as a predictive marker for specific subtypes of PE requires further investigation. Lastly, the association between early-pregnancy uric acid levels and preterm birth may be confounded by preeclampsia-related clinical decisions, as many preterm deliveries in our cohort were iatrogenic. We cannot determine from this observational data whether uric acid has an independent effect on preterm birth or if the observed association is mediated through preeclampsia. Further prospective and mechanistic studies are needed to clarify this relationship.
Conclusion
In summary, our study demonstrates a significant positive correlation between elevated serum UA levels before gestational age of 20 weeks and PE, with this positive correlation presenting a non-linear relationship unaffected by confounding factors such as age and parity. This association is more pronounced in patients with a BMI < 24 kg/m2, indicating that elevated UA levels before gestational age of 20 weeks may lead to an increased risk of PE. These findings underscore the importance of monitoring early pregnancy UA levels for predicting disease progression in PE, which holds significant clinical implications for the prediction and treatment of PE.
Data availability
Data will be provided by the corresponding author upon reasonable request.
References
Li, G. et al. Hypoxic stress disrupts HGF/Met signaling in human trophoblasts: implications for the pathogenesis of preeclampsia. J. Biomed. Sci. 29(1), 8 (2022).
Yang, Y. et al. Preeclampsia prevalence, risk factors, and pregnancy outcomes in Sweden and China. JAMA Netw. Open 4(5), e218401 (2021).
Burton, G. J., Redman, C. W., Roberts, J. M. & Moffett, A. Pre-eclampsia: pathophysiology and clinical implications. BMJ (Clin. Res. ed.) 366, l2381 (2019).
Dalbeth, N.; Gosling, A. L.; Gaffo, A.; Abhishek, A., Gout. Lancet (London, England) 397 (10287), 1843-1855. (2021)
Yue, C., Ying, C. & Li, X. Association of first trimester serum uric acid with preeclampsia: an observational cohort study with propensity score matching. Hypertens. Res. : off. J. Jpn. Soc. Hypertens. 46(2), 377–385 (2023).
Khaliq, O. P., Konoshita, T., Moodley, J. & Naicker, T. The role of uric acid in preeclampsia: is uric acid a causative factor or a sign of preeclampsia?. Curr. Hypertens. Rep. 20(9), 80 (2018).
Chen, Y. et al. Increased uric acid, gamma-glutamyl transpeptidase and alkaline phosphatase in early-pregnancy associated with the development of gestational hypertension and preeclampsia. Front. Cardiovasc. Med. 8, 756140 (2021).
Wolak, T., Sergienko, R., Wiznitzer, A., Paran, E. & Sheiner, E. High uric acid level during the first 20 weeks of pregnancy is associated with higher risk for gestational diabetes mellitus and mild preeclampsia. Hypertens. Pregnancy 31(3), 307–15 (2012).
Chen, Q. et al. Serum uric acid may not be involved in the development of preeclampsia. J. Hum. Hypertens. 30(2), 136–40 (2016).
Sanchez-Lozada, L. G. et al. Uric acid and hypertension: an update with recommendations. Am. J. Hypertens. 33(7), 583–594 (2020).
Lam, C., Lim, K. H., Kang, D. H. & Karumanchi, S. A. Uric acid and preeclampsia. Semin. Nephrol. 25(1), 56–60 (2005).
Shakarami, A., Ghafarzadeh, M., Yari, F. & Fathi, L. Association between maternal serum uric acid and preeclampsia. Arch. Physiol. Biochem. 128(6), 1434–1437 (2022).
Li, Q. et al. Association between triglyceride-glucose index in early pregnancy and risk of preeclampsia: a multicenter retrospective cohort study. Lipids Health Dis. 24(1), 152 (2025).
Yue, C. Y., Gao, J. P., Zhang, C. Y. & Ying, C. M. Is serum vitamin D deficiency before gestational 20 weeks a risk factor for preeclampsia?. Clin. Nutr. (Edinburgh, Scotland) 40(6), 4430–4435 (2021).
Croke, L. Gestational hypertension and preeclampsia: a practice bulletin from ACOG. Am. Fam. Phys. 100(10), 649–650 (2019).
ACOG Practice Bulletin No. 202: gestational hypertension and preeclampsia. Obstet. Gynecol. 133(1), 1 (2019).
Nair, A. & Savitha, C. Estimation of serum uric acid as an indicator of severity of preeclampsia and perinatal outcome. J. Obstet. Gynaecol. India 67(2), 109–118 (2017).
Bellos, I., Pergialiotis, V., Loutradis, D. & Daskalakis, G. The prognostic role of serum uric acid levels in preeclampsia: a meta-analysis. J. Clin. Hypertens. (Greenwich, Conn.) 2(5), 826–834 (2020).
Zhao, X., Frempong, S. T. & Duan, T. Uric acid levels in gestational hypertensive women predict preeclampsia and outcome of small-for-gestational-age infants. J. Matern.-fetal Neonatal Med. : Off. J. Eur. Assoc. Perinat. Med., Fed. Asia Ocean. Perinat. Soc., Int. Soc. Perinat. Obstet. 34(17), 2825–2831 (2021).
Roberts, J. M. et al. Uric acid is as important as proteinuria in identifying fetal risk in women with gestational hypertension. Hypertension (Dallas, Tex. : 1979) 46(6), 1263–9 (2005).
Xiong, Y. et al. Association of maternal serum uric acid levels with LBW/SGA: a large retrospective cohort study. BMC Pregnancy Childbirth 24(1), 160 (2024).
Cheung, K. L. & Lafayette, R. A. Renal physiology of pregnancy. Adv. Chronic Kidney Dis. 20(3), 209–14 (2013).
Kang, D. H. et al. Uric acid, endothelial dysfunction and pre-eclampsia: searching for a pathogenetic link. J. Hypertens. 22(2), 229–35 (2004).
Sánchez-Lozada, L. G. et al. Uric acid-induced endothelial dysfunction is associated with mitochondrial alterations and decreased intracellular ATP concentrations Nephron. Exp. Nephrol. 121(3–4), e71-8 (2012).
Bainbridge, S. A. & Roberts, J. M. Uric acid as a pathogenic factor in preeclampsia. Placenta 29(Suppl A), S67-72 (2008).
Bainbridge, S. A. et al. Uric acid attenuates trophoblast invasion and integration into endothelial cell monolayers. Am. J. Physiol. Cell Physiol. 297(2), C440-50 (2009).
Burton, G. J. & Jauniaux, E. Pathophysiology of placental-derived fetal growth restriction. Am. J. Obstet. Gynecol. 218(2s), S745-s761 (2018).
Redman, C. W. & Sargent, I. L. Placental stress and pre-eclampsia: a revised view. Placenta 30(Suppl A), S38-42 (2009).
Farah, O., Nguyen, C., Tekkatte, C. & Parast, M. M. Trophoblast lineage-specific differentiation and associated alterations in preeclampsia and fetal growth restriction. Placenta 102, 4–9 (2020).
Acknowledgements
The authors acknowledge the physicians, nurses, and scientific staff of Obstetrics and Gynecology Hospital affiliated with Fudan University, the First People’s Hospital of Chenzhou, and the Wuxi Maternity and Child Health Care Hospital.
Funding
This study is supported by the Pregnancy Cohort of the scientific collaboration between the First People’s Hospital of Chenzhou and the Tenth People’s Hospital of Shanghai (2023-09) and Natural Science Foundation of Shanghai Basic Research Program (25ZR1402039).
Author information
Authors and Affiliations
Contributions
CYZ designed the study, collected and analyzed the data, interpreted the results, wrote and reviewed the manuscript. ML collected the data, reviewed the manuscript. QL designed the study, collected and analyzed the data, interpreted the results, wrote the manuscript.YJP and YL interpreted the results and reviewed the manuscript. CYY designed the study, collected and analyzed the data, interpreted the results, and revised the manuscript. YC collected the data, reviewed the manuscript. DD interpreted the results, revised the manuscript. All authors approved the final version of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethics
This study was approved by the ethics committees of Fudan University Obstetrics and Gynecology Hospital and Chenzhou First People’s Hospital and Wuxi Maternity and Child Health Care Hospital. All participants provided broad informed consent.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhao, C., Li, M., Li, Q. et al. Elevated serum uric acid before 20 weeks of gestation increases the risk of preeclampsia. Sci Rep 16, 5335 (2026). https://doi.org/10.1038/s41598-026-36218-6
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-026-36218-6