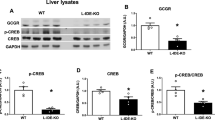
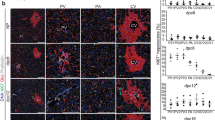
Abstract
Hepatocytes are organized along a spatial axis between the portal triad and the central vein to form functionally repetitive units known as lobules. The hepatocytes perform distinct metabolic functions depending on their location within the lobule. Single-cell analysis of hepatocytes across the liver lobule demonstrates that gluconeogenic gene expression is relatively low in the fed state and gradually increases in the periportal hepatocytes during the initial fasting period. As fasting progresses, pericentral hepatocyte gluconeogenic gene expression and gluconeogenic activity also increase and, following entry into a starvation state, the pericentral hepatocytes show similar gluconeogenic gene expression and activity to the periportal hepatocytes. In parallel, starvation suppresses canonical β-catenin signalling and modulates the expression of pericentral and periportal glutamine synthetase and glutaminase, respectively, resulting in enhanced incorporation of glutamine into glucose. Thus, hepatocyte gluconeogenic gene expression and glucose production are spatially and temporally plastic across the liver lobule, underscoring the complexity of defining hepatic insulin resistance and glucose production on a whole-organ level, as well as for a particular fasted or fed condition.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
Raw FASTQ files were deposited to the National Center for Biotechnology Information Gene Expression Omnibus database and are available under accession numbers GSE263415, GSE263418 and GSE263419. Proteomics raw files are available in ProteomeXchange (PRIDE) under accession number PXD051593. For KEGG reference pathways, mmu00010, mmu00061 and mmu00062 were used. Free access and query to the complete single-cell dataset is available through the Shiny interactive web application (https://pessinlab.shinyapps.io/liver). Source data are provided with this paper.
Code availability
This study was conducted using only publicly available software; no custom code was used.
References
Gebhardt, R. Metabolic zonation of the liver: regulation and implications for liver function. Pharmacol. Ther. 53, 275–354 (1992).
Martini, T., Naef, F. & Tchorz, J. S. Spatiotemporal metabolic liver zonation and consequences on pathophysiology. Annu. Rev. Pathol. 18, 439–466 (2023).
Trefts, E., Gannon, M. & Wasserman, D. H. The liver. Curr. Biol. 27, R1147–R1151 (2017).
Santoleri, D. & Titchenell, P. M. Resolving the paradox of hepatic insulin resistance. Cell Mol. Gastroenterol. Hepatol. 7, 447–456 (2019).
Ben-Moshe, S. & Itzkovitz, S. Spatial heterogeneity in the mammalian liver. Nat. Rev. Gastroenterol. Hepatol. 16, 395–410 (2019).
Braeuning, A. et al. Differential gene expression in periportal and perivenous mouse hepatocytes. FEBS J. 273, 5051–5061 (2006).
Jungermann, K. & Katz, N. Functional hepatocellular heterogeneity. Hepatology 2, 385–395 (1982).
Chen, K. S. & Katz, J. Zonation of glycogen and glucose syntheses, but not glycolysis, in rat liver. Biochem. J. 255, 99–104 (1988).
Tosh, D., Alberti, G. M. & Agius, L. Glucagon regulation of gluconeogenesis and ketogenesis in periportal and perivenous rat hepatocytes. Heterogeneity of hormone action and of the mitochondrial redox state. Biochem. J. 256, 197–204 (1988).
Evans, J. L., Quistorff, B. & Witters, L. A. Zonation of hepatic lipogenic enzymes identified by dual-digitonin-pulse perfusion. Biochem. J. 259, 821–829 (1989).
Guzman, M. & Castro, J. Zonation of fatty acid metabolism in rat liver. Biochem. J. 264, 107–113 (1989).
Katz, N. R., Fischer, W. & Giffhorn, S. Distribution of enzymes of fatty acid and ketone body metabolism in periportal and perivenous rat-liver tissue. Eur. J. Biochem. 135, 103–107 (1983).
Lin, P. et al. Single-cell and spatially resolved transcriptomics for liver biology. Hepatology 80, 698–720 (2024).
Droin, C. et al. Space–time logic of liver gene expression at sub-lobular scale. Nat. Metab. 3, 43–58 (2021).
Bahar Halpern, K. et al. Single-cell spatial reconstruction reveals global division of labour in the mammalian liver. Nature 542, 352–356 (2017).
Sun, T. et al. ZNRF3 and RNF43 cooperate to safeguard metabolic liver zonation and hepatocyte proliferation. Cell Stem Cell 28, 1822–1837.e10 (2021).
Wei, Y. et al. Liver homeostasis is maintained by midlobular zone 2 hepatocytes. Science 371, eabb1625 (2021).
He, L. et al. Proliferation tracing reveals regional hepatocyte generation in liver homeostasis and repair. Science 371, eabc4346 (2021).
Youn, D. Y. et al. The Mediator complex kinase module is necessary for fructose regulation of liver glycogen levels through induction of glucose-6-phosphatase catalytic subunit (G6pc). Mol. Metab. 48, 101227 (2021).
Acosta-Rodriguez, V. et al. Circadian alignment of early onset caloric restriction promotes longevity in male C57BL/6J mice. Science 376, 1192–1202 (2022).
Francois, M., Canal Delgado, I., Shargorodsky, N., Leu, C. S. & Zeltser, L. Assessing the effects of stress on feeding behaviors in laboratory mice. Elife 11, e70271 (2022).
Storch, K. F. et al. Extensive and divergent circadian gene expression in liver and heart. Nature 417, 78–83 (2002).
Geisler, C. E., Hepler, C., Higgins, M. R. & Renquist, B. J. Hepatic adaptations to maintain metabolic homeostasis in response to fasting and refeeding in mice. Nutr. Metab. (Lond.) 13, 62 (2016).
Carper, D., Coue, M., Laurens, C., Langin, D. & Moro, C. Reappraisal of the optimal fasting time for insulin tolerance tests in mice. Mol. Metab. 42, 101058 (2020).
Van der Mierden, S. et al. Measuring endogenous corticosterone in laboratory mice—a mapping review, meta-analysis, and open source database. ALTEX 38, 111–122 (2021).
Kuo, T., McQueen, A., Chen, T. C. & Wang, J. C. Regulation of glucose homeostasis by glucocorticoids. Adv. Exp. Med. Biol. 872, 99–126 (2015).
Stenvers, D. J., Scheer, F., Schrauwen, P., la Fleur, S. E. & Kalsbeek, A. Circadian clocks and insulin resistance. Nat. Rev. Endocrinol. 15, 75–89 (2019).
Wang, W. et al. Rotating day and night disturb growth hormone secretion profiles, body energy metabolism, and insulin levels in mice. Neuroendocrinology 112, 481–492 (2022).
Ben-Moshe, S. et al. Spatial sorting enables comprehensive characterization of liver zonation. Nat. Metab. 1, 899–911 (2019).
Kanehisa, M., Sato, Y., Kawashima, M., Furumichi, M. & Tanabe, M. KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res. 44, D457–D462 (2016).
Wagner, A. et al. Metabolic modeling of single Th17 cells reveals regulators of autoimmunity. Cell 184, 4168–4185.e21 (2021).
Thiele, I. et al. A community-driven global reconstruction of human metabolism. Nat. Biotechnol. 31, 419–425 (2013).
Chi, Y. et al. Regulation of gene expression during the fasting–feeding cycle of the liver displays mouse strain specificity. J. Biol. Chem. 295, 4809–4821 (2020).
Chen, G., Ning, B. & Shi, T. Single-cell RNA-seq technologies and related computational data analysis. Front. Genet. 10, 317 (2019).
Olsen, J. V. et al. Global, in vivo, and site-specific phosphorylation dynamics in signaling networks. Cell 127, 635–648 (2006).
Imbert, A. et al. FISH-quant v2: a scalable and modular tool for smFISH image analysis. RNA 28, 786–795 (2022).
Tunnacliffe, E. & Chubb, J. R. What is a transcriptional burst? Trends Genet. 36, 288–297 (2020).
Bahar Halpern, K. et al. Bursty gene expression in the intact mammalian liver. Mol. Cell 58, 147–156 (2015).
Quistorff, B. Gluconeogenesis in periportal and perivenous hepatocytes of rat liver, isolated by a new high-yield digitonin/collagenase perfusion technique. Biochem. J. 229, 221–226 (1985).
Lindros, K. O. & Penttila, K. E. Digitonin–collagenase perfusion for efficient separation of periportal or perivenous hepatocytes. Biochem. J. 228, 757–760 (1985).
Zhou, Y. et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 10, 1523 (2019).
Kurland, I. J., Alcivar, A., Bassilian, S. & Lee, W. N. Loss of [13C]glycerol carbon via the pentose cycle. Implications for gluconeogenesis measurement by mass isotoper distribution analysis. J. Biol. Chem. 275, 36787–36793 (2000).
Williams, H. C. et al. APOE alters glucose flux through central carbon pathways in astrocytes. Neurobiol. Dis. 136, 104742 (2020).
Kramer, A., Green, J., Pollard, J. Jr. & Tugendreich, S. Causal analysis approaches in Ingenuity Pathway Analysis. Bioinformatics 30, 523–530 (2014).
Rim, E. Y., Clevers, H. & Nusse, R. The Wnt pathway: from signaling mechanisms to synthetic modulators. Annu. Rev. Biochem. 91, 571–598 (2022).
Benhamouche, S. et al. Apc tumor suppressor gene is the ‘zonation-keeper’ of mouse liver. Dev. Cell 10, 759–770 (2006).
Hu, S. et al. Single-cell spatial transcriptomics reveals a dynamic control of metabolic zonation and liver regeneration by endothelial cell Wnt2 and Wnt9b. Cell Rep. Med. 3, 100754 (2022).
Fitamant, J. et al. YAP inhibition restores hepatocyte differentiation in advanced HCC, leading to tumor regression. Cell Rep. 10, 1692–1707 (2015).
Matz-Soja, M., Hovhannisyan, A. & Gebhardt, R. Hedgehog signalling pathway in adult liver: A major new player in hepatocyte metabolism and zonation? Med. Hypotheses 80, 589–594 (2013).
Burke, Z. D. et al. Liver zonation occurs through a β-catenin-dependent, c-Myc-independent mechanism. Gastroenterology 136, 2316–2324.e3 (2009).
Planas-Paz, L. et al. The RSPO-LGR4/5-ZNRF3/RNF43 module controls liver zonation and size. Nat. Cell Biol. 18, 467–479 (2016).
Yamaguchi, H. & Taouk, G. M. A potential role of YAP/TAZ in the interplay between metastasis and metabolic alterations. Front. Oncol. 10, 928 (2020).
Zanconato, F. et al. Genome-wide association between YAP/TAZ/TEAD and AP-1 at enhancers drives oncogenic growth. Nat. Cell Biol. 17, 1218–1227 (2015).
Kapoor, A. et al. Yap1 activation enables bypass of oncogenic Kras addiction in pancreatic cancer. Cell 179, 1239 (2019).
Skoda, A. M. et al. The role of the Hedgehog signaling pathway in cancer: a comprehensive review. Bosn. J. Basic Med. Sci. 18, 8–20 (2018).
Carballo, G. B., Honorato, J. R., de Lopes, G. P. F. & Spohr, T. A highlight on Sonic Hedgehog pathway. Cell Commun. Signal. 16, 11 (2018).
Katoh, Y. & Katoh, M. Hedgehog target genes: mechanisms of carcinogenesis induced by aberrant hedgehog signaling activation. Curr. Mol. Med. 9, 873–886 (2009).
Lee, E. Y. et al. Hedgehog pathway-regulated gene networks in cerebellum development and tumorigenesis. Proc. Natl Acad. Sci. USA 107, 9736–9741 (2010).
Cheng, X. et al. Glucagon contributes to liver zonation. Proc. Natl Acad. Sci. USA 115, E4111–E4119 (2018).
Cadoret, A. et al. New targets of beta-catenin signaling in the liver are involved in the glutamine metabolism. Oncogene 21, 8293–8301 (2002).
Hale, G. et al. Correlation of exon 3 beta-catenin mutations with glutamine synthetase staining patterns in hepatocellular adenoma and hepatocellular carcinoma. Mod. Pathol. 29, 1370–1380 (2016).
Curthoys, N. P. & Watford, M. Regulation of glutaminase activity and glutamine metabolism. Annu. Rev. Nutr. 15, 133–159 (1995).
Bauer, M. et al. Starvation response in mouse liver shows strong correlation with life-span-prolonging processes. Physiol. Genomics 17, 230–244 (2004).
Yelamanchi, S. D. et al. A pathway map of glutamate metabolism. J. Cell Commun. Signal. 10, 69–75 (2016).
Miller, R. A. et al. Targeting hepatic glutaminase activity to ameliorate hyperglycemia. Nat. Med. 24, 518–524 (2018).
Kosti, I., Jain, N., Aran, D., Butte, A. J. & Sirota, M. Cross-tissue analysis of gene and protein expression in normal and cancer tissues. Sci. Rep. 6, 24799 (2016).
Rolfs, Z. et al. An atlas of protein turnover rates in mouse tissues. Nat. Commun. 12, 6778 (2021).
Han, H. S., Kang, G., Kim, J. S., Choi, B. H. & Koo, S. H. Regulation of glucose metabolism from a liver-centric perspective. Exp. Mol. Med. 48, e218 (2016).
Stark, R. & Kibbey, R. G. The mitochondrial isoform of phosphoenolpyruvate carboxykinase (PEPCK-M) and glucose homeostasis: Has it been overlooked? Biochim. Biophys. Acta 1840, 1313–1330 (2014).
Sarkar, A. et al. Intermittent fasting induces rapid hepatocyte proliferation to restore the hepatostat in the mouse liver. Elife 12, e82311 (2023).
Russell, J. O. & Monga, S. P. Wnt/beta-catenin signaling in liver development, homeostasis, and pathobiology. Annu. Rev. Pathol. 13, 351–378 (2018).
Ip, W. et al. Liver-specific expression of dominant-negative transcription factor 7-like 2 causes progressive impairment in glucose homeostasis. Diabetes 64, 1923–1932 (2015).
Liu, H. et al. Wnt signaling regulates hepatic metabolism. Sci. Signal. 4, ra6 (2011).
Yamaguchi, K. et al. Bidirectional reporter assay using HAL promoter and TOPFLASH improves specificity in high-throughput screening of Wnt inhibitors. Biotechnol. Bioeng. 114, 2868–2882 (2017).
Stumvoll, M., Perriello, G., Meyer, C. & Gerich, J. Role of glutamine in human carbohydrate metabolism in kidney and other tissues. Kidney Int. 55, 778–792 (1999).
Sileo, P., Simonin, C., Melnyk, P., Chartier-Harlin, M. C. & Cotelle, P. Crosstalk between the Hippo pathway and the Wnt pathway in Huntington’s disease and other neurodegenerative disorders. Cells 11, 3631 (2022).
Cox, A. G. et al. Yap reprograms glutamine metabolism to increase nucleotide biosynthesis and enable liver growth. Nat. Cell Biol. 18, 886–896 (2016).
Verboven, E. et al. Regeneration defects in Yap and Taz mutant mouse livers are caused by bile duct disruption and cholestasis. Gastroenterology 160, 847–862 (2021).
Planas-Paz, L. et al. YAP, but not RSPO-LGR4/5, signaling in biliary epithelial cells promotes a ductular reaction in response to liver injury. Cell Stem Cell 25, 39–53.e10 (2019).
Daniels, L. J., Kay, D., Marjot, T., Hodson, L. & Ray, D. W. Circadian regulation of liver metabolism: experimental approaches in human, rodent, and cellular models. Am. J. Physiol. Cell Physiol. 325, C1158–C1177 (2023).
Stokkan, K. A., Yamazaki, S., Tei, H., Sakaki, Y. & Menaker, M. Entrainment of the circadian clock in the liver by feeding. Science 291, 490–493 (2001).
Damiola, F. et al. Restricted feeding uncouples circadian oscillators in peripheral tissues from the central pacemaker in the suprachiasmatic nucleus. Genes Dev. 14, 2950–2961 (2000).
Vollmers, C. et al. Time of feeding and the intrinsic circadian clock drive rhythms in hepatic gene expression. Proc. Natl Acad. Sci. USA 106, 21453–21458 (2009).
Ouyang, J. F., Kamaraj, U. S., Cao, E. Y. & Rackham, O. J. L. ShinyCell: simple and sharable visualization of single-cell gene expression data. Bioinformatics 37, 3374–3376 (2021).
Dong, S. et al. Monitoring spatiotemporal changes in chaperone-mediated autophagy in vivo. Nat. Commun. 11, 645 (2020).
Fernandez, C. A., Des Rosiers, C., Previs, S. F., David, F. & Brunengraber, H. Correction of 13C mass isotopomer distributions for natural stable isotope abundance. J. Mass Spectrom. 31, 255–262 (1996).
Bankhead, P. et al. QuPath: open source software for digital pathology image analysis. Sci. Rep. 7, 16878 (2017).
Yheskel, M., Sidoli, S. & Secombe, J. Proximity labeling reveals a new in vivo network of interactors for the histone demethylase KDM5. Epigenetics Chromatin 16, 8 (2023).
Aguilan, J. T., Kulej, K. & Sidoli, S. Guide for protein fold change and p-value calculation for non-experts in proteomics. Mol. Omics 16, 573–582 (2020).
Acknowledgements
This study was supported by grants from the National Institutes of Health (DK110063 (to F.Y., C.E., K.S. and J.E.P.), DK020541 (to V.L.S., I.J.K. and J.E.P.), DK110426 (to K.S.), T32GM007491 (to M.H.), S10OD030286 (to S.S.), P30CA013330 (to D. Reynolds, Genomics Core) and U2CDK135074 (National Mouse Metabolic Phenotyping Center at the University of California Davis). We thank S. Okada for data analysis, the National Mouse Metabolic Phenotyping Center at the University of California Davis, L. Leung for immunofluorescence assistance and D. Reynolds (Genomics Core) for the scRNA-seq processing.
Author information
Authors and Affiliations
Contributions
J.O. performed scRNA-seq, targeted scRNA-seq, smFISH and gluconeogenesis assays as well as drafting and editing the manuscript. A.L. performed periportal and pericentral hepatocyte isolation, bulk RNA-seq, proteomics sample processing, data analyses and manuscript editing. A.M.X. performed AAV-sgRNA preparation, generated the hepatocyte-specific Gls2-KO mice and edited the manuscript. L.L. performed data management and bioinformatic analyses, built the Shiny web application and edited the manuscript. M.H. performed proteomic analyses, bioinformatic data analyses and manuscript editing. V.L.S., F.Y., S.S. and J.E.P. provided experimental advice, data interpretation and manuscript editing. Y.Q. performed the mass spectrometry analyses for gluconeogenesis, experimental advice, data interpretation and manuscript editing. I.J.K. supervised the mass spectrometry, design for stable isotope usage, experimental advice, data interpretation and manuscript editing. C.E. supervised smFISH and provided experimental advice, data interpretation and manuscript editing. K.S. supervised scRNA-seq and provided experimental advice, data interpretation and manuscript editing.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Dale S. Edgerton, Jan Tchorz and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Revati Dewal, in collaboration with the Nature Metabolism team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Day-time and night-time feeding does not significantly affect Pck1/Fasn gene expression or metabolic factors in either male or female mice.
(a, b) Relative mRNA levels of Pck1 (A) and Fasn (B) from total hepatocytes in the feeding time course. N = 3 mice per time point/condition. Ordinary one-way ANOVA followed by Dunnett’s multiple comparisons test was performed to compare -4h with different time points. p values of *p < 0.05, **p < 0.01, and ***p < 0.001 was considered statistically significant when comparing two groups. (N.S.=not statistically significant). (c, d) Relative mRNA levels of lipogenic (Fasn) (C) and gluconeogenic (Pck1) (D) genes from total hepatocytes in the fed (0 h), fasted (8 h) and starvation (24 h) state. N = 3 mice per time point/condition. Ordinary one-way ANOVA followed by Dunnett’s multiple comparisons test was performed to compare 0 h with different time points. p values of *p < 0.05, **p < 0.01, and ***p < 0.001 was considered statistically significant when comparing two groups. (N.S.=not statistically significant). (e-m) Glucagon/insulin ratio, Relative mRNA levels, body weight, blood glucose, and glycogen measurements from male/female mice (day-time fed vs. night-time fed trained) in the fasting time course; fed4h (0 h), fast8h (8 h), fast16h (16 h), fast20h (20 h), and fast24h (24 h). N = 3-4 mice per time point/condition. (E) Glucagon/insulin ratio in plasma from day-time fed vs. night-time fed trained male mice. (f, g) Relative mRNA levels of lipogenic (Pck1) (F) and gluconeogenic (Fasn) (G) genes from day-time fed vs. night-time fed trained male mice. (h) Body weight (g) from day-time fed vs. night-time fed trained male mice. (i) Blood glucose (mg/dl) from day-time fed vs. night-time fed trained male mice. (j, k) Relative mRNA levels of lipogenic (Pck1) (J) and gluconeogenic (Fasn) (K) genes from day-time fed vs. night-time fed trained female mice. (l) Body weight (g) from day-time fed vs. night-time fed trained female mice. (m) Blood glucose (mg/dl) from day-time fed vs. night-time fed trained female mice. (n-q) Glycogen content (mg/g) from total hepatocytes in the fasting time course; day-time fed vs. night-time fed trained male mice (N), day-time fed vs. night-time fed trained female mice (O), day-time fed vs. night-time fed untrained male mice (P), and day-time fed vs. night-time fed untrained female mice (Q). N = 3-4 mice per time point/condition. (r-u) Body weight and blood glucose measurements from male/female mice (day-time fed vs. night-time fed untrained) in the fasting time course; fed4h (0 h), fast8h (8 h), fast16h (16 h), fast20h (20 h), and fast24h (24 h). N = 3-4 mice per time point/condition. (R) Body weight (g) from day-time fed vs. night-time fed untrained male mice. (S) Blood glucose (mg/dl) from day-time fed vs. night-time fed untrained male mice. (T) Body weight (g) from day-time fed vs. night-time fed untrained female mice. (U) Blood glucose (mg/dl) from day-time fed vs. night-time fed untrained female mice.
Extended Data Fig. 2 Food entrainment effects on the liver local clock and plasma circadian hormone levels.
(A-H) Relative mRNA levels of Bmal1 and Clock from male/female mice in the fasting time course; fed4h (0 h), fast8h (8 h), fast16h (16 h), fast20h (20 h), and fast24h (24 h). N = 3-4 mice per time point/condition. Results aligned to zeitgeber time (ZT); ZT0-12 had the light on, whereas ZT12-0 had the light off. (a, b) Relative mRNA levels of Bmal1 from day-time fed vs. night-time fed trained male (A) and female (B) mice. (c, d) Relative mRNA levels of Clock from day-time fed vs. night-time fed trained male (C) and female (D) mice. (e, f) Relative mRNA levels of Bmal1 from day-time fed vs. night-time fed untrained male (E) and female (F) mice. (g, h) Relative mRNA levels of Clock from day-time fed vs. night-time fed untrained male (G) and female (H) mice. (i-l) Corticosterone and Growth Hormone levels in plasma from male mice in the fasting time course; fed4h (0 h), fast8h (8 h), fast16h (16 h), fast20h (20 h), and fast24h (24 h). N = 3-4 mice per time point/condition. Results aligned to zeitgeber time (ZT); ZT0-12 had the light on, whereas ZT12-0 had the light off. (I, J) Corticosterone and growth hormone levels in plasma from day-time fed vs. night-time fed trained (I) and untrained (J) male mice. (K, L) Growth hormone levels in plasma from day-time fed vs. night-time fed trained (K) and untrained (L) male mice.
Extended Data Fig. 3 Compass analyses of several gene pathways that are not altered in the fed, fasted or starvation state.
(a, b) Compass-score differential activity analysis of the Vitamin A pathway between 0 h and 8 h pericentral/periportal hepatocytes (A) and between 0 h and 24 h pericentral/periportal hepatocytes (B) based on Cohen’s D analysis. BCDO (BCMO1) is highlighted. (c, d) Compass-score differential activity analysis of the Vitamin B6 pathway between 0 h and 8 h pericentral/periportal hepatocytes (C) and between 0 h and 24 h pericentral/periportal hepatocytes (D) based on Cohen’s D analysis. PNPO is highlighted. (e, f) Compass-score differential activity analysis of the Starch and sucrose (glycogen) pathway between 0 h and 8 h pericentral/periportal hepatocytes (E) and between 0 h and 24 h pericentral/periportal hepatocytes (F) based on Cohen’s D analysis. GLPASE (PYGL) is highlighted. (g) Representative immunoblotting of BCMO1, PNPO, PYGL, CYP2E1, E-Cadherin, and Vinculin from pericentral and periportal isolated hepatocytes.
Extended Data Fig. 4 Quantitated target scRNA-seq analyses for zonated gene expression.
(a) Sequencing saturation plot of each library. X-axis represents mean reads per cell; Y-axis represents percentage of transcripts sequenced; dotted line is the approximate saturation point. (b) Violin plots of Log2 expression Pck1 and Fasn comparing standard and targeted scRNA-seq in 0 h and 16 h. (c) Ridge plot analysis of gluconeogenic (Pck1, G6pc) and lipogenic (Acly, Fasn) genes from targeted scRNA-seq. X-axis represents log2 expression; Y-axis represents each time point/condition, red dotted line shows the threshold for cells positive of each gene. (d) MFuzz analysis of the targeted scRNA-seq to cluster similar expression profiles. Once percentage of positive cells were defined based on ridge plot analysis (Fig. S4C), each gene expressed in pericentral and periportal hepatocytes were assigned to 6 main clusters; early fasting induced, late fasting induced, cycling, early fasting suppressed, late fasting suppressed, constitutively expressed. X-axis represents time points; Y-axis represents percentage of positive cells.
Extended Data Fig. 5 Quantification of Pck1 and Fasn transcription sites by spatial smFISH analyses.
(a, b) Representative smFISH images of pericentral (PC) and periportal (PP) areas selected for quantification of TS (Pck1 (A), and Fasn (B)) from 0 h, 4 h, and 24 h in male mice. TS are highlighted with arrows. (c, d) Histogram based on the intensity of each TS (Pck1 (C), and Fasn (D)). X-axis represents the intensity of TS (A.U.); Y-axis represents the number of TS within each intensity. Dotted line shows the mean value of the intensity of TS with SD.
Extended Data Fig. 6 smFISH spatial analyses of Pck1 and Fasn expression in female mice livers.
(a, b) Representative smFISH images of pericentral (PC) and periportal (PP) areas from 0 h, 4 h, and 24 h in female mice. TS are highlighted with arrows. (c-h) Relative mRNA levels of zonation markers (Cyp2e1 pericentral (C), Cyp2f2 periportal (D)), gluconeogenic genes (Pck1 (E), G6pc (F)), and lipogenic genes (Fasn (G), Acly (H)) from pericentral and periportal isolated hepatocytes in female mice. N = 3 mice for 8hPC/PP and 24hPP, N = 4 mice for 0hPC/PP and 24hPC. Data are presented as mean values +/− SD. Student’s t-test with unpaired two-tailed p values of *p < 0.05, **p < 0.01, and ***p < 0.001 was considered statistically significant when comparing two groups. (N.S.=not statistically significant).
Extended Data Fig. 7 Identification of nutrition-independent zonation mRNA markers that are unchanged in the fed, fasted and starvation states.
(a) Principal Component Analysis (PCA) of RNA-seq from pericentral and periportal isolated hepatocytes. Each dot represents each time point/condition based on coloring and shape of dot (0 h=red, 8 h=yellow, 24 h=blue; circle=pericentral, triangle=periportal). Green arrow shows how fasting shift the pericentral hepatocytes. (b) The number of differentially expressed genes in pericentral and periportal hepatocytes throughout the fed (0 h), fasted (8 h) and starvation (24 h) state in the RNA-seq from pericentral and periportal isolated hepatocytes. Differentially expressed genes were defined based on log2Fold Change <-1 for pericentral, >1 for periportal, both with a cutoff of p-adj<0.05. (c, e) Venn diagram of differentially expressed genes in pericentral (C) and periportal (E) hepatocytes throughout the fed (0 h), fasted (8 h) and starvation (24 h) state in the RNA-seq from pericentral and periportal isolated hepatocytes. (d, f) Metascape analysis on nutrition-independent upregulated pericentral genes (D) and periportal genes (F). Top 5 pathways in each zone shown (-log10(P)).
Extended Data Fig. 8 The entire mass isotopologue distribution (MID) of glucose from [U-13C] pyruvate or glutamine.
(a) Steady state gluconeogenic time course experiment from [U-13C]-pyruvate using total hepatocytes isolated from 24 h fasted mice. N = 3 mice. Glucose isotopologues with one 13C to six 13C carbons (M1 thru M6 isotopologues) are shown. Data are presented as mean values + SD. (b) Fed, fasting and starvation response of glucose isotopologues with one 13C to six 13C carbons (M1 thru M6 isotopologues) derived from [U-13C]-pyruvate using pericentral and periportal isolated hepatocytes. N = 4 for all conditions except 0hPC, 8hPP, 16hPP, and 24hPC (N = 3). Data are presented as mean values + SD. (c) Fasted 24 hr response of glucose isotopologues with one to three 13C carbons (% enrichment) derived from [U-13C]-glutamine comparing pericentral vs. periportal isolated hepatocytes using Control vs GLS2KO mice 24 h mice. N = 3 mice per condition. Data are presented as mean values + SD.
Extended Data Fig. 9 Canonical WNT target gene expression decreases in pericentral hepatocytes in the starvations state.
(a, b) RNA-seq data (counts) of pericentral zonation markers (Gulo and Cyp2e1) and periportal zonation markers (Hsd17b13 and Cyp2f2) (A), gluconeogenic genes (Pck1, G6pc) and lipogenic genes (Acly, Fasn) (B) from pericentral and periportal isolated hepatocytes. N = 3 mice per time point/condition. Data are presented as mean values +/− SD. Student’s t-test with unpaired two-tailed p values of *p < 0.05, **p < 0.01, and ***p < 0.001 was considered statistically significant when comparing two groups. (N.S.=not statistically significant). (c) Heatmap of WNT target genes in RNA-seq from pericentral and periportal isolated hepatocytes. Average expression of N = 3 mice from each time point/condition is shown. Coloring based on z-score. 4 main clusters show PC upregulated/fast reduced, fast induced, PP upregulated fast reduced, fast reduced (D) RNA-seq data (counts) of WNT signaling related genes from pericentral and periportal isolated hepatocytes. N = 3 mice per time point/condition. Data are presented as mean values +/− SD. Student’s t-test with unpaired two-tailed p values of *p < 0.05, **p < 0.01, and ***p < 0.001 was considered statistically significant when comparing two groups. (N.S.=not statistically significant).
Extended Data Fig. 10 HIPPO/YAP and sonic hedgehog zonation gene pathway changes in the fed, fasted and starvation states.
(a) Heatmap of Yap target genes in RNA-seq from pericentral and periportal isolated hepatocytes. Average expression of N = 3 mice from each time point/condition is shown. Coloring based on z-score. (b) RNA-seq data (counts) of Yap related genes from pericentral and periportal isolated hepatocytes. N = 3 mice per time point/condition. Data are presented as mean values +/− SD. Student’s t-test with unpaired two-tailed p values of *p < 0.05, **p < 0.01, and ***p < 0.001 was considered statistically significant when comparing two groups. (N.S.=not statistically significant). (c) Heatmap of Shh target genes in RNA-seq from pericentral and periportal isolated hepatocytes. Average expression of N = 3 mice from each time point/condition is shown. Coloring based on z-score. (d) RNA-seq data (counts) of Shh related genes from pericentral and periportal isolated hepatocytes. N = 3 mice per time point/condition. Data are presented as mean values +/− SD. Student’s t-test with unpaired two-tailed p values of *p < 0.05, **p < 0.01, and ***p < 0.001 was considered statistically significant when comparing two groups. (N.S.=not statistically significant).
Supplementary information
Supplementary Information (download PDF )
Supplementary Tables 1–5.
Source data
Source Data Fig. 4 (download XLSX )
smFISH TS.
Source Data Fig. 5 (download XLSX )
RT–qPCR, western blot quantification.
Source Data Fig. 5 (download PDF )
Unprocessed western blots.
Source Data Fig. 6 (download XLSX )
Stable isotope.
Source Data Fig. 7 (download XLSX )
RNA-seq, IF, proteomics, RT–qPCR, stable isotope.
Source Data Fig. 7 (download PDF )
Unprocessed western blots.
Source Data Extended Data Fig./Table 1 (download XLSX )
RT–qPCR, Insulin/glucagon, BW, BG, glycogen.
Source Data Extended Data Fig./Table 2 (download XLSX )
RT–qPCR, hormones.
Source Data Extended Data Fig./Table 3 (download PDF )
Unprocessed western blots.
Source Data Extended Data Fig./Table 4 (download XLSX )
MFuzz analysis.
Source Data Extended Data Fig./Table 5 (download XLSX )
smFISH intensities.
Source Data Extended Data Fig./Table 8 (download XLSX )
Isotopologue distribution.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Okada, J., Landgraf, A., Xiaoli, A.M. et al. Spatial hepatocyte plasticity of gluconeogenesis during the metabolic transitions between fed, fasted and starvation states. Nat Metab 7, 1073–1091 (2025). https://doi.org/10.1038/s42255-025-01269-y
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s42255-025-01269-y
This article is cited by
-
Spatial protein gradients pattern liver metabolism
Nature Metabolism (2026)
-
Amygdala–liver signalling orchestrates glycaemic responses to stress
Nature (2025)
-
Starvation induces metabolic hepatocyte reprogramming
Nature Metabolism (2025)