Abstract
Understanding the pathophysiology of neurological symptoms observed after severe acute respiratory syndrome coronavirus 2 (SARS-CoV2) infection is essential to optimizing outcomes and therapeutics. To date, small sample sizes and narrow molecular profiling have limited the generalizability of findings. In this study, we profiled multiple cortical and subcortical regions in postmortem brains of patients with coronavirus disease 2019 (COVID-19) and controls with matched pulmonary pathology (total n = 42) using spatial transcriptomics, bulk gene expression and proteomics. We observed a multi-regional antiviral response without direct active SARS-CoV2 infection. We identified dysregulation of mitochondrial and synaptic pathways in deep-layer excitatory neurons and upregulation of neuroinflammation in glia, consistent across both mRNA and protein. Remarkably, these alterations overlapped substantially with changes in age-related neurodegenerative diseases, including Parkinson’s disease and Alzheimer’s disease. Our work, combining multiple experimental and analytical methods, demonstrates the brain-wide impact of severe acute/subacute COVID-19, involving both cortical and subcortical regions, shedding light on potential therapeutic targets within pathways typically associated with pathological aging and neurodegeneration.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
All processed data are available in the main text or the supplementary materials and source data. The proteomics data have been deposited to MassIVE with accession number MSV000094297 (ftp://massive.ucsd.edu/v07/MSV000094297/). Raw data of GeoMx spatial transcriptomics (.DCC, .pkc files and Q3 normalized counts) and nCounter direct gene expression assays (.RCC files) have been deposited in the National Center for Biotechnology Information’s Gene Expression Omnibus (GEO) and are accessible through GEO Series with accession number GSE274267.
Code availability
The code used in this study can be found at https://github.com/dhglab/Brain-wide-alterations-revealed-by-spatial-transcriptomics-and-proteomics-in-COVID-19-infection.
References
Zhu, N. et al. A novel coronavirus from patients with pneumonia in China, 2019. N. Engl. J. Med. 382, 727–733 (2020).
Xu, E., Xie, Y. & Al-Aly, Z. Long-term neurologic outcomes of COVID-19. Nat. Med. 28, 2406–2415 (2022).
Moriguchi, T. et al. A first case of meningitis/encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 94, 55–58 (2020).
Helms, J. et al. Neurologic features in severe SARS-CoV-2 infection. N. Engl. J. Med. 382, 2268–2270 (2020).
Spudich, S. & Nath, A. Nervous system consequences of COVID-19. Science 375, 267–269 (2022).
Mao, L. et al. Neurologic manifestations of hospitalized patients with coronavirus disease 2019 in Wuhan, China. JAMA Neurol. 77, 683–690 (2020).
Helbok, R. et al. Guillain–Barre syndrome in a patient with antibodies against SARS-COV-2. Eur. J. Neurol. 27, 1754–1756 (2020).
Badenoch, J. B. et al. Persistent neuropsychiatric symptoms after COVID-19: a systematic review and meta-analysis. Brain Commun. 4, fcab297 (2022).
Novak, P. et al. Multisystem involvement in post-acute sequelae of coronavirus disease 19. Ann. Neurol. 91, 367–379 (2022).
Gandal, M. J. et al. Shared molecular neuropathology across major psychiatric disorders parallels polygenic overlap. Science 359, 693–697 (2018).
Parikshak, N. N., Gandal, M. J. & Geschwind, D. H. Systems biology and gene networks in neurodevelopmental and neurodegenerative disorders. Nat. Rev. Genet. 16, 441–458 (2015).
Gandal, M. J. et al. Broad transcriptomic dysregulation occurs across the cerebral cortex in ASD. Nature 611, 532–539 (2022).
Ma, Y., Klein, H. U. & De Jager, P. L. Considerations for integrative multi-omic approaches to explore Alzheimer’s disease mechanisms. Brain Pathol. 30, 984–991 (2020).
Olah, M. et al. A transcriptomic atlas of aged human microglia. Nat. Commun. 9, 539 (2018).
Castanares-Zapatero, D. et al. Pathophysiology and mechanism of long COVID: a comprehensive review. Ann. Med. 54, 1473–1487 (2022).
Iadecola, C., Anrather, J. & Kamel, H. Effects of COVID-19 on the nervous system. Cell 183, 16–27 (2020).
Douaud, G. et al. SARS-CoV-2 is associated with changes in brain structure in UK Biobank. Nature 604, 697–707 (2022).
Thakur, K. T. et al. COVID-19 neuropathology at Columbia University Irving Medical Center/New York Presbyterian Hospital. Brain 144, 2696–2708 (2021).
Lou, J. J. et al. Neuropathology of COVID-19 (neuro-COVID): clinicopathological update. Free Neuropathol. https://doi.org/10.17879/freeneuropathology (2021).
Solomon, I. H. et al. Neuropathological features of Covid-19. N. Engl. J. Med. 383, 989–992 (2020).
Manca, R., De Marco, M., Ince, P. G. & Venneri, A. Heterogeneity in regional damage detected by neuroimaging and neuropathological studies in older adults with COVID-19: a cognitive-neuroscience systematic review to inform the long-term impact of the virus on neurocognitive trajectories. Front. Aging Neurosci. 13, 646908 (2021).
Coolen, T. et al. Early postmortem brain MRI findings in COVID-19 non-survivors. Neurology 95, e2016–e2027 (2020).
Serrano, G. E. et al. SARS-CoV-2 brain regional detection, histopathology, gene expression, and immunomodulatory changes in decedents with COVID-19. J. Neuropathol. Exp. Neurol. 81, 666–695 (2022).
Mavrikaki, M., Lee, J. D., Solomon, I. H. & Slack, F. J. Severe COVID-19 is associated with molecular signatures of aging in the human brain. Nat. Aging 2, 1130–1137 (2022).
Yang, A. C. et al. Dysregulation of brain and choroid plexus cell types in severe COVID-19. Nature 595, 565–571 (2021).
Fullard, J. F. et al. Single-nucleus transcriptome analysis of human brain immune response in patients with severe COVID-19. Genome Med. 13, 118 (2021).
Goytain, A. & Ng, T. NanoString nCounter technology: high-throughput RNA validation. Methods Mol. Biol. 2079, 125–139 (2020).
Merritt, C. R. et al. Multiplex digital spatial profiling of proteins and RNA in fixed tissue. Nat. Biotechnol. 38, 586–599 (2020).
Morabito, S. et al. Single-nucleus chromatin accessibility and transcriptomic characterization of Alzheimer’s disease. Nat. Genet. 53, 1143–1155 (2021).
Morabito, S., Reese, F., Rahimzadeh, N., Miyoshi, E. & Swarup, V. hdWGCNA identifies co-expression networks in high-dimensional transcriptomics data. Cell Rep. Methods 3, 100498 (2023).
Huang, T. et al. MSstatsTMT: statistical detection of differentially abundant proteins in experiments with isobaric labeling and multiple mixtures. Mol. Cell Proteom. 19, 1706–1723 (2020).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Bates, D., Mächler, M., Bolker, B. & Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. https://doi.org/10.18637/jss.v067.i01 (2015).
Shen, L. GeneOverlap: an R package to test and visualize gene overlaps. R package version 1.38.0 https://bioconductor.org/packages/release/bioc/html/GeneOverlap.html (2024).
Wang, K. et al. CD147-spike protein is a novel route for SARS-CoV-2 infection to host cells. Signal Transduct. Target. Ther. 5, 283 (2020).
Hoffmann, M. et al. SARS-CoV-2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell 181, 271–280(2020).
Shang, J. et al. Cell entry mechanisms of SARS-CoV-2. Proc. Natl Acad. Sci. USA 117, 11727–11734 (2020).
Skene, N. G. & Grant, S. G. Identification of vulnerable cell types in major brain disorders using single cell transcriptomes and expression weighted cell type enrichment. Front. Neurosci. 10, 16 (2016).
Wamsley, B. et al. Molecular cascades and cell type-specific signatures in ASD revealed by single-cell genomics. Science 384, eadh2602 (2024).
Bakken, T. E. et al. Comparative cellular analysis of motor cortex in human, marmoset and mouse. Nature 598, 111–119 (2021).
Velmeshev, D. et al. Single-cell genomics identifies cell type-specific molecular changes in autism. Science 364, 685–689 (2019).
Zhou, Y. et al. Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat. Commun. 10, 1523 (2019).
Szklarczyk, D. et al. The STRING database in 2021: customizable protein–protein networks, and functional characterization of user-uploaded gene/measurement sets. Nucleic Acids Res. 49, D605–D612 (2021).
Juarez-Rebollar, D., Rios, C., Nava-Ruiz, C. & Mendez-Armenta, M. Metallothionein in brain disorders. Oxid. Med. Cell. Longev. 2017, 5828056 (2017).
Ionescu-Tucker, A. & Cotman, C. W. Emerging roles of oxidative stress in brain aging and Alzheimer’s disease. Neurobiol. Aging 107, 86–95 (2021).
Lau, V., Ramer, L. & Tremblay, M. E. An aging, pathology burden, and glial senescence build-up hypothesis for late onset Alzheimer’s disease. Nat. Commun. 14, 1670 (2023).
Yu, N., Pasha, M. & Chua, J. J. E. Redox changes and cellular senescence in Alzheimer’s disease. Redox Biol. 70, 103048 (2024).
Johnson, E. C. B. et al. Large-scale deep multi-layer analysis of Alzheimer’s disease brain reveals strong proteomic disease-related changes not observed at the RNA level. Nat. Neurosci. 25, 213–225 (2022).
Teuscher, A. C. et al. Longevity interventions modulate mechanotransduction and extracellular matrix homeostasis in C. elegans. Nat. Commun. 15, 276 (2024).
Wang, K. et al. Epigenetic regulation of aging: implications for interventions of aging and diseases. Signal Transduct. Target. Ther. 7, 374 (2022).
Irier, H. A. & Jin, P. Dynamics of DNA methylation in aging and Alzheimer’s disease. DNA Cell Biol. 31, S42–S48 (2012).
Muzio, L., Viotti, A. & Martino, G. Microglia in neuroinflammation and neurodegeneration: from understanding to therapy. Front. Neurosci 15, 742065 (2021).
Litvinchuk, A. et al. Amelioration of Tau and ApoE4-linked glial lipid accumulation and neurodegeneration with an LXR agonist. Neuron 112, 384–403 (2024).
Adibhatla, R. M. & Hatcher, J. F. Role of lipids in brain injury and diseases. Future Lipidol. 2, 403–422 (2007).
Johnson, A. A. & Stolzing, A. The role of lipid metabolism in aging, lifespan regulation, and age-related disease. Aging Cell 18, e13048 (2019).
Kuleshov, M. V. et al. Enrichr: a comprehensive gene set enrichment analysis web server 2016 update. Nucleic Acids Res. 44, W90–W97 (2016).
Krzywinski, M. et al. Circos: an information aesthetic for comparative genomics. Genome Res. 19, 1639–1645 (2009).
Bader, G. D. & Hogue, C. W. An automated method for finding molecular complexes in large protein interaction networks. BMC Bioinformatics 4, 2 (2003).
Swarup, V. et al. Identification of conserved proteomic networks in neurodegenerative dementia. Cell Rep. 31, 107807 (2020).
Subramanian, A. et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc. Natl Acad. Sci. USA 102, 15545–15550 (2005).
Mootha, V. K. et al. PGC-1α-responsive genes involved in oxidative phosphorylation are coordinately downregulated in human diabetes. Nat. Genet. 34, 267–273 (2003).
Zhang, P. P. et al. COVID-19-associated monocytic encephalitis (CAME): histological and proteomic evidence from autopsy. Signal Transduct. Target. Ther. 8, 24 (2023).
Dickson, D. W. Parkinson’s disease and parkinsonism: neuropathology. Cold Spring Harb. Perspect. Med. 2, a009258 (2012).
Emmi, A. et al. Detection of SARS-CoV-2 viral proteins and genomic sequences in human brainstem nuclei. NPJ Parkinsons Dis. 9, 25 (2023).
Bulfamante, G. et al. Brainstem neuropathology in two cases of COVID-19: SARS-CoV-2 trafficking between brain and lung. J. Neurol. 268, 4486–4491 (2021).
Hoffman, G. E. & Roussos, P. Dream: powerful differential expression analysis for repeated measures designs. Bioinformatics 37, 192–201 (2021).
Freeze, W. M. et al. Blood–brain barrier leakage and microvascular lesions in cerebral amyloid angiopathy. Stroke 50, 328–335 (2019).
Zhang, W. et al. Integrated transcriptomic and metabolomic analysis of cortical neurons reveals dysregulated lipid metabolism, enhanced glycolysis and activated HIF-1 signaling pathways in acute hypoxia. Heliyon 9, e14949 (2023).
Heng, J. S. et al. Hypoxia tolerance in the Norrin-deficient retina and the chronically hypoxic brain studied at single-cell resolution. Proc. Natl Acad. Sci. USA 116, 9103–9114 (2019).
Montine, T. J. et al. National Institute on Aging–Alzheimer’s Association guidelines for the neuropathologic assessment of Alzheimer’s disease: a practical approach. Acta Neuropathol. 123, 1–11 (2012).
Stein, S. R. et al. SARS-CoV-2 infection and persistence in the human body and brain at autopsy. Nature 612, 758–763 (2022).
Alexandersen, S., Chamings, A. & Bhatta, T. R. SARS-CoV-2 genomic and subgenomic RNAs in diagnostic samples are not an indicator of active replication. Nat. Commun. 11, 6059 (2020).
Bocci, M. et al. Infection of brain pericytes underlying neuropathology of COVID-19 patients. Int. J. Mol. Sci. 22, 11622 (2021).
Boluda, S. et al. Golgi localization of SARS-CoV-2 spike protein and interaction with furin in cerebral COVID-19 microangiopathy: a clue to the central nervous system involvement? Free Neuropathol. https://doi.org/10.17879/freeneuropathology-2023-4584 (2023).
Lake, B. B. et al. A comparative strategy for single-nucleus and single-cell transcriptomes confirms accuracy in predicted cell-type expression from nuclear RNA. Sci. Rep. 7, 6031 (2017).
Hodge, R. D. et al. Conserved cell types with divergent features in human versus mouse cortex. Nature 573, 61–68 (2019).
Reveret, L. et al. Higher angiotensin-converting enzyme 2 (ACE2) levels in the brain of individuals with Alzheimer’s disease. Acta Neuropathol. Commun. 11, 159 (2023).
Lopera, F. et al. Resilience to autosomal dominant Alzheimer’s disease in a Reelin-COLBOS heterozygous man. Nat. Med. 29, 1243–1252 (2023).
Sleigh, J. N. et al. Neuropilin 1 sequestration by neuropathogenic mutant glycyl-tRNA synthetase is permissive to vascular homeostasis. Sci. Rep. 7, 9216 (2017).
Schwab, C., Hosokawa, M., Akiyama, H. & McGeer, P. L. Familial British dementia: colocalization of furin and ABri amyloid. Acta Neuropathol. 106, 278–284 (2003).
Hur, J. Y. et al. The innate immunity protein IFITM3 modulates γ-secretase in Alzheimer’s disease. Nature 586, 735–740 (2020).
She, X. et al. Immune surveillance of brain metastatic cancer cells is mediated by IFITM1. EMBO J. 42, e111112 (2023).
Song, H. et al. Detection of blood–brain barrier disruption in brains of patients with COVID-19, but no evidence of brain penetration by SARS-CoV-2. Acta Neuropathol. 146, 771–775 (2023).
Baker, A. et al. Specialized subpopulations of deep-layer pyramidal neurons in the neocortex: bridging cellular properties to functional consequences. J. Neurosci. 38, 5441–5455 (2018).
Schapira, A. H. Mitochondria in the aetiology and pathogenesis of Parkinson’s disease. Lancet Neurol 7, 97–109 (2008).
Malpartida, A. B., Williamson, M., Narendra, D. P., Wade-Martins, R. & Ryan, B. J. Mitochondrial dysfunction and mitophagy in parkinson’s disease: from mechanism to therapy. Trends Biochem. Sci 46, 329–343 (2021).
Toomey, C. E. et al. Mitochondrial dysfunction is a key pathological driver of early stage Parkinson’s. Acta Neuropathol. Commun. 10, 134 (2022).
Ashleigh, T., Swerdlow, R. H. & Beal, M. F. The role of mitochondrial dysfunction in Alzheimer's disease pathogenesis. Alzheimers Dement. 19, 333–342 (2023).
Chakravorty, A., Jetto, C. T. & Manjithaya, R. Dysfunctional mitochondria and mitophagy as drivers of Alzheimer’s disease pathogenesis. Front. Aging Neurosci. 11, 311 (2019).
Misrani, A., Tabassum, S. & Yang, L. Mitochondrial dysfunction and oxidative stress in Alzheimer’s disease. Front. Aging Neurosci. 13, 617588 (2021).
Carmo, C., Naia, L., Lopes, C. & Rego, A. C. Mitochondrial dysfunction in Huntington’s disease. Adv. Exp. Med. Biol. 1049, 59–83 (2018).
Damiano, M., Galvan, L., Deglon, N. & Brouillet, E. Mitochondria in Huntington’s disease. Biochim. Biophys. Acta 1802, 52–61 (2010).
Amorim, J. A. et al. Mitochondrial and metabolic dysfunction in ageing and age-related diseases. Nat. Rev. Endocrinol. 18, 243–258 (2022).
Campisi, J. et al. From discoveries in ageing research to therapeutics for healthy ageing. Nature 571, 183–192 (2019).
Baranova, A., Cao, H. & Zhang, F. Causal effect of COVID-19 on Alzheimer’s disease: a Mendelian randomization study. J. Med. Virol. 95, e28107 (2022).
Li, C., Liu, J., Lin, J. & Shang, H. COVID-19 and risk of neurodegenerative disorders: a Mendelian randomization study. Transl. Psychiatry 12, 283 (2022).
Shanley, J. E. et al. Longitudinal evaluation of neurologic-post acute sequelae SARS-CoV-2 infection symptoms. Ann. Clin. Transl. Neurol. 9, 995–1010 (2022).
Silva, J. et al. Neuropathological aspects of SARS-CoV-2 infection: significance for both Alzheimer’s and Parkinson’s disease. Front. Neurosci. 16, 867825 (2022).
Golzari-Sorkheh, M., Weaver, D. F. & Reed, M. A. COVID-19 as a risk factor for Alzheimer’s disease. J. Alzheimers Dis. 91, 1–23 (2023).
Green, R. et al. SARS-CoV-2 infection increases the gene expression profile for Alzheimer’s disease risk. Mol. Ther. Methods Clin. Dev. 27, 217–229 (2022).
Ryback, R. & Eirin, A. Mitochondria, a missing link in COVID-19 heart failure and arrest? Front. Cardiovasc. Med. 8, 830024 (2021).
De la Cruz-Enriquez, J., Rojas-Morales, E., Ruiz-Garcia, M. G., Tobon-Velasco, J. C. & Jimenez-Ortega, J. C. SARS-CoV-2 induces mitochondrial dysfunction and cell death by oxidative stress/inflammation in leukocytes of COVID-19 patients. Free Radic. Res. 55, 982–995 (2021).
Zhu, B. et al. Inhibition of the mitochondrial pyruvate carrier simultaneously mitigates hyperinflammation and hyperglycemia in COVID-19. Sci. Immunol. 8, eadf0348 (2023).
Nunn, A. V. W., Guy, G. W., Brysch, W. & Bell, J. D. Understanding long COVID; mitochondrial health and adaptation—old pathways, new problems. Biomedicines 10, 3113 (2022).
Gordon, D. E. et al. A SARS-CoV-2 protein interaction map reveals targets for drug repurposing. Nature 583, 459–468 (2020).
Priemer, D. S., Rhodes, C. H., Karlovich, E., Perl, D. P. & Goldman, J. E. Abeta deposits in the neocortex of adult and infant hypoxic brains, including in cases of COVID-19. J. Neuropathol. Exp. Neurol. 81, 988–995 (2022).
Tayeb-Fligelman, E. et al. Low complexity domains of the nucleocapsid protein of SARS-CoV-2 form amyloid fibrils. Nat. Commun. 14, 2379 (2023).
Nystrom, S. & Hammarstrom, P. Amyloidogenesis of SARS-CoV-2 spike protein. J. Am. Chem. Soc. 144, 8945–8950 (2022).
Charnley, M. et al. Neurotoxic amyloidogenic peptides in the proteome of SARS-COV2: potential implications for neurological symptoms in COVID-19. Nat. Commun. 13, 3387 (2022).
Johnson, V. E., Stewart, W. & Smith, D. H. Traumatic brain injury and amyloid-β pathology: a link to Alzheimer’s disease? Nat. Rev. Neurosci. 11, 361–370 (2010).
Eimer, W. A. et al. Alzheimer’s disease-associated β-amyloid is rapidly seeded by herpesviridae to protect against brain infection. Neuron 99, 56–63 (2018).
Vojtechova, I., Machacek, T., Kristofikova, Z., Stuchlik, A. & Petrasek, T. Infectious origin of Alzheimer’s disease: amyloid beta as a component of brain antimicrobial immunity. PLoS Pathog. 18, e1010929 (2022).
Kumar, D. K. et al. Amyloid-β peptide protects against microbial infection in mouse and worm models of Alzheimer’s disease. Sci. Transl. Med. 8, 340ra372 (2016).
Tian, W. et al. Immune suppression in the early stage of COVID-19 disease. Nat. Commun. 11, 5859 (2020).
Wauters, E. et al. Discriminating mild from critical COVID-19 by innate and adaptive immune single-cell profiling of bronchoalveolar lavages. Cell Res. 31, 272–290 (2021).
Leng, L. et al. Pathological features of COVID-19-associated lung injury: a preliminary proteomics report based on clinical samples. Signal Transduct. Target. Ther. 5, 240 (2020).
Sacco, K. et al. Immunopathological signatures in multisystem inflammatory syndrome in children and pediatric COVID-19. Nat. Med. 28, 1050–1062 (2022).
Cabral-Marques, O. et al. Autoantibodies targeting GPCRs and RAS-related molecules associate with COVID-19 severity. Nat. Commun. 13, 1220 (2022).
Wallukat, G. et al. Functional autoantibodies against G-protein coupled receptors in patients with persistent Long-COVID-19 symptoms. J. Transl. Autoimmun. 4, 100100 (2021).
Frere, J. J. et al. SARS-CoV-2 infection in hamsters and humans results in lasting and unique systemic perturbations after recovery. Sci. Transl. Med. 14, eabq3059 (2022).
Nie, X. et al. Multi-organ proteomic landscape of COVID-19 autopsies. Cell 184, 775–791 (2021).
Kramer, A., Green, J., Pollard, J. Jr & Tugendreich, S. Causal analysis approaches in Ingenuity Pathway Analysis. Bioinformatics 30, 523–530 (2014).
Schweizer, L. et al. Quantitative multiorgan proteomics of fatal COVID-19 uncovers tissue-specific effects beyond inflammation. EMBO Mol. Med. 15, e17459 (2023).
Vanderboom, P. M. et al. Proteomic signature of host response to SARS-CoV-2 infection in the nasopharynx. Mol. Cell. Proteom. 20, 100134 (2021).
Paranjpe, I. et al. Proteomic characterization of acute kidney injury in patients hospitalized with SARS-CoV2 infection. Commun. Med. (Lond.) 3, 81 (2023).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Coscia, F. et al. A streamlined mass spectrometry-based proteomics workflow for large-scale FFPE tissue analysis. J. Pathol. 251, 100–112 (2020).
Hughes, C. S. et al. Ultrasensitive proteome analysis using paramagnetic bead technology. Mol. Syst. Biol. 10, 757 (2014).
Deng, W., Sha, J., Plath, K. & Wohlschlegel, J. A. Carboxylate-modified magnetic bead (CMMB)-based isopropanol gradient peptide fractionation (CIF) enables rapid and robust off-line peptide mixture fractionation in bottom-up proteomics. Mol. Cell. Proteom. 20, 100039 (2021).
McAlister, G. C. et al. MultiNotch MS3 enables accurate, sensitive, and multiplexed detection of differential expression across cancer cell line proteomes. Anal. Chem. 86, 7150–7158 (2014).
Cox, J. & Mann, M. MaxQuant enables high peptide identification rates, individualized p.p.b.-range mass accuracies and proteome-wide protein quantification. Nat. Biotechnol. 26, 1367–1372 (2008).
Gregory, J. M. et al. Spatial transcriptomics identifies spatially dysregulated expression of GRM3 and USP47 in amyotrophic lateral sclerosis. Neuropathol. Appl. Neurobiol. 46, 441–457 (2020).
Sankowski, R. et al. Multiomic spatial landscape of innate immune cells at human central nervous system borders. Nat. Med. 30, 186–198 (2024).
Langfelder, P. & Horvath, S. WGCNA: an R package for weighted correlation network analysis. BMC Bioinformatics 9, 559 (2008).
Langfelder, P. & Horvath, S. Fast R functions for robust correlations and hierarchical clustering. J Stat. Softw. https://doi.org/10.18637/jss.v046.i11 (2012).
Risso, D., Ngai, J., Speed, T. P. & Dudoit, S. Normalization of RNA-seq data using factor analysis of control genes or samples. Nat. Biotechnol. 32, 896–902 (2014).
Knight, J. D. et al. A web-tool for visualizing quantitative protein–protein interaction data. Proteomics 15, 1432–1436 (2015).
Knight, J. D. R. et al. ProHits-viz: a suite of web tools for visualizing interaction proteomics data. Nat. Methods 14, 645–646 (2017).
Szklarczyk, D. et al. STRING v11: protein–protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 47, D607–D613 (2019).
Shannon, P. et al. Cytoscape: a software environment for integrated models of biomolecular interaction networks. Genome Res. 13, 2498–2504 (2003).
Goedhart, J. & Luijsterburg, M. S. VolcaNoseR is a web app for creating, exploring, labeling and sharing volcano plots. Sci. Rep. 10, 20560 (2020).
Kanehisa, M., Sato, Y., Kawashima, M., Furumichi, M. & Tanabe, M. KEGG as a reference resource for gene and protein annotation. Nucleic Acids Res. 44, D457–D462 (2016).
Ashburner, M. et al. Gene Ontology: tool for the unification of biology. Nat. Genet. 25, 25–29 (2000).
Gene Ontology Consortium et al. The Gene Ontology knowledgebase in 2023. Genetics 224, iyad031 (2023).
Gillespie, M. et al. The reactome pathway knowledgebase 2022. Nucleic Acids Res. 50, D687–D692 (2022).
Ruepp, A. et al. CORUM: the comprehensive resource of mammalian protein complexes—2009. Nucleic Acids Res. 38, D497–D501 (2010).
Martens, M. et al. WikiPathways: connecting communities. Nucleic Acids Res. 49, D613–D621 (2021).
Stark, C. et al. BioGRID: a general repository for interaction datasets. Nucleic Acids Res. 34, D535–D539 (2006).
Turei, D., Korcsmaros, T. & Saez-Rodriguez, J. OmniPath: guidelines and gateway for literature-curated signaling pathway resources. Nat. Methods 13, 966–967 (2016).
Li, T. et al. A scored human protein–protein interaction network to catalyze genomic interpretation. Nat. Methods 14, 61–64 (2017).
Acknowledgements
We thank the autopsy service chief and staff at the Department of Pathology and Laboratory Medicine, G. Fishbein, K. Ellis, C. E. Ramos and G. Ceballos for facilitating the tissue procurement. We thank C. K. Williams at the section of neuropathology for helpful discussion and advice on tissue processing. We thank members of the Geschwind laboratory (R. Kawaguchi, L. Bricks, Y. Tan, X. Huan and L. O. Chen) for helpful discussion and advice on data analyses. This work was funded by the Eli and Edythe Broad Center of Regenerative Medicine and Stem Cell Research (BSCRC); the UCLA COVID-19 Research Award (OCRC no. 20-70) to D.H.G., H.V.V. and T.Z.; the Dr. Miriam and Sheldon G. Adelson Medical Research Foundation (20231126) to D.H.G.; the Translational Research Fund of the UCLA Department of Pathology and Laboratory Medicine to T.Z and H.V.V.; and the UCLA Intercampus Medical Genetics Training Program (T32GM008243) to T.Z. The funders had no role in study design, data collection and analysis, decision to publish or preparation of the manuscript.
Author information
Authors and Affiliations
Contributions
Experiments were designed by D.H.G, H.V.V. and T.Z. Spatial transcriptomics data were generated by T.Z., Y. Li, L.P., C.N., Y. Lee, Y.-C.S. and X.L. Bulk RNA direct gene expression (nCounter) data were generated by T.Z., Y. Li, M.B. and E.F.-K. Proteomics data were generated by T.Z., Y. Li, J.S. and J.W. Data analysis was performed by T.Z., Y.-C.S., J.S. and D.H.G. The manuscript was written by T.Z., S.M., H.V.V. and D.H.G. The work was supervised by D.H.G. and H.V.V.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Aging thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Gene expression profile from the frontal grey matter correlates with nCounter bulk tissue direct gene expression.
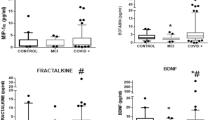
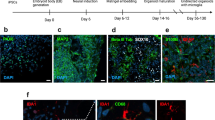
a) Bar plots showing the number of differentially expressed molecules in different assays (BH-adjusted p < 0.1, MSstatsTMT31 for proteomics; DESeq232 for nCounter direct gene expression; lme433 for spatial transcriptomics). b) Venn diagrams and Pearson’s correlation of overlapped differentially expressed genes from nCounter direct gene expression of the frontal lobe and spatial transcriptomics of frontal grey matter (p = 1.60E-11, GeneOverlap34, Pearson’s correlation, two-sided, r = 0.77, p = 1.62E-33) and frontal white matter (p = 0.60, GeneOverlap34, Pearson’s correlation, two-sided, r = -0.42, p = 0.72). c) 3 SARS-CoV2 transcripts in nCounter direct gene expression (basal ganglia) indicated there was no increased expression of viral mRNA (N = 6 for controls, N = 6 for COVID-19 patients, two-tailed unpaired t test). d) Bar plots showing the results of cell type enrichment analysis (EWCE38) of NRGN neuron from Yang, et al, 202125 and its celltype markers’ enrichment when comparing to the major cell types (level 1) and subclass cell types (level 2) in Wamsley, et al, 202439 and Bakken, et al, 202140. The red dashed line represents a threshold of 2.5 for the sd_from_mean. Cell types with a sd_from_mean value > 2.5 and a BH-adjusted p < 0.05 are considered statistically significant.
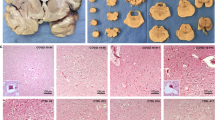
Extended Data Fig. 2 Increased ACE2 and FURIN protein expression in the frontal cortex of severe acute/subacute COVID-19 patients.
Anti-ACE2 (a) and Anti-FURIN (b) immunohistochemistry highlight the increased protein expression at the vascular/perivascular structures (magenta arrowhead) in the white matter of the COVID-19 patients (N = 9) comparing to the non-COVID controls (N = 8), one-tailed unpaired t-test of area percentage for positive immunoreactivity, p = 3.95E-2 (ACE2) and p = 3.00E-3 (FURIN).
Extended Data Fig. 3 Increased NRP1 protein expression in the frontal cortex of severe acute/subacute COVID-19 patients.
a) Anti-NRP1 immunohistochemistry in the frontal cortex of non-COVID controls (N = 8) and COVID-19 patients (N = 9) are displayed. Within the non-COVID controls, NRP1 immunoreactivity has a predominant astrocytic expression pattern (cyan arrowhead) in five out of eight cases, while a predominant neuronal expression pattern (purple arrowhead) is observed in the remaining three. In the COVID-19 patients, NRP1 immunoreactivity is seen more frequently in both neurons and astrocytes. Moreover, increased neuronal immunoreactivity is found in the nucleus of five out of nine patients, and in the cytosol of two out of nine patients. The overall immunopositive area percentage is increased in the COVID-19 patients (p = 3.20E-3, one-tailed unpaired t-test). b) Anti-Fibrin immunohistochemistry in the frontal white matter of non-COVID controls (N = 5) and COVID-19 patients (N = 4). Increased intravascular and perivascular fibrin deposition (yellow arrowhead), indicative of thrombi formation and blood brain barrier/neurovascular unit defect, is seen in both non-COVID controls and COVID-19 patients (one-tailed unpaired t-test of area percentage for positive immunoreactivity, p = 2.68E-1).
Extended Data Fig. 4 Network dendrogram, EWCE results of WGCNA and GO enrichment analysis of hdWGCNA.
a) Consensus WGCNA network dendrogram from co-expression topological overlap of genes in frontal grey matter and pontine nuclei spatial transcriptomics. b-d) Dot plots illustrating cell type enrichment analysis (EWCE38) results of consensus modules, using major cell types from Wamsley, et al, 202439(b), subclass cell types from Bakken, et al, 202140(c), and Yang, et al, 202125(d). The color intensity corresponds to the sd_from_mean of each cell type. The greyscale of the dot edge corresponds to the FDR-adjusted p value for each cell type. The dot size in each row represents the relative abundance across all the modules. e) Top GO enrichment terms of all the excitatory neuron modules (EnrichR56).
Extended Data Fig. 5 Additional GO enrichment, PPI and MCODE network analyses of differentially expressed proteins in eight regions.
a) The graph reflects the same networks depicted in Fig. 6b (downregulated GO cluster, left panel) and Fig. 6c (upregulated GO cluster, right panel), but with the regional information displayed as a pie chart for each gene. Each GO term is proportioned within the pie chart and color-coded based on the number of proteins under the term from the corresponding region (key below)42. Most of the terms displayed are shared by more than three regions. b) Merged protein-protein interaction and MCODE network analyses42 of differentially expressed proteins in eight regions. Top GO terms for each MCODE network are selected for labeling.
Extended Data Fig. 6 Brain-wide differential gene expression in severe acute/subacute COVID-19 patients shows minimal overlap with hypoxia-related changes.
a) Volcano plots depict the NES values from Gene Set Enrichment Analysis (GSEA60,61, GeneSet Hallmark) and FDR-q values across 10 areas of the frontal lobe, pons, and midbrain from spatial transcriptomics results. Notably, hypoxia-related genes are not significantly enriched in any of the profiled areas. b) Venn diagrams illustrate the results of comparison between the top 500 differentially up-and down-regulated genes observed in primary cortical neurons under hypoxia conditions68 and those identified in the grey matter of COVID-19 patients (BH-adjusted p < 0.05) in this study, no significant overlap are identified (for upregulated genes, the comparisons between Zhang, et al68 and frontal grey matter, pontine nuclei, and substantia nigra, p = 9.04E-1, 8.13E-1, and 1.78E-1, respectively; for downregulated genes, the corresponding p = 9.35E-1, 1.00, 3.71E-1, GeneOverlap34). We also compared the 500 upregulated genes of mouse brains under hypoxic conditions for 2 days (gene pattern 1) and 7 days (gene pattern 2)69 to the upregulated genes in frontal grey matter, finding no significant overlap (p = 4.70E-1 and 1.88E-1, GeneOverlap34).
Extended Data Fig. 7 Increased amyloid deposition including senile plaques and cerebral amyloid angiopathy, not Tau deposition, in severe acute/subacute COVID-19 patients.
a) Anti-Aβ42 immunohistochemistry highlights the amyloid deposition including senile plaques (orange arrowhead) and cerebral amyloid angiopathy (CAA, purple arrowhead) in the COVID-19 patients (N = 9) which are not seen in the non-COVID controls (N = 8) (one-tailed unpaired t-test of area percentage for positive immunoreactivity, p = 4.61E-2). b) Anti-phospho-Tau immunohistochemistry does not detect significant positive immunoreactivity and difference in positive immunoreactivity area between non-COVID control (N = 8) and COVID-19 patient (N = 9) (one-tailed unpaired t-test, p = 4.84E-1) groups. Rare neurons containing neurofibrillary tangles and neuropil threads (cyan arrowhead) are observed. As positive controls, Aβ42 immunopositivity highlights the characteristic senile plaques and cerebral amyloid angiopathy of advanced stage Alzheimer’s disease (Braak stage V-VI). Anti-phospho-Tau antibody highlights abundant neurofibrillary tangles and neuropil threads (cyan arrowhead), and neuritic plaques (yellow arrowhead) in AD patient. Anti-Aβ42 and phospho-Tau IHCs were repeated once.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–6 and legends and Supplementary Tables 1–4.
Supplementary Data 1 (download XLSX )
Differential_Expression_Spatial_Transcriptomics.
Supplementary Data 2 (download XLSX )
Differential_Expression_nCounter_bulk_direct_gene_expression.
Supplementary Data 3 (download XLSX )
Differential_Expression_Proteomics.
Supplementary Data 4 (download XLSX )
Differential_expression_of_controls_with_lung_pathology_and_with_no_significant_pathology.
Supplementary Data 5 (download XLSX )
Statistical source data.
Supplementary Data 6 (download CSV )
Statistical source data.
Supplementary Data 7 (download ZIP )
PPT file and images for Fig. 1.
Supplementary Data 8 (download ZIP )
PPT file and images for Fig. 2.
Supplementary Data 9 (download ZIP )
PPT file and images for Fig. 3.
Supplementary Data 10 (download ZIP )
PPT file and images for Fig. 4.
Supplementary Data 11 (download ZIP )
PPT file and images for Fig. 5.
Supplementary Data 12 (download ZIP )
PPT file and images for Fig. 6.
Supplementary Data 13 (download ZIP )
PPT file and images for Fig. 7.
Supplementary Data 14 (download ZIP )
PPT file and images for Extended Fig. 1.
Supplementary Data 15 (download ZIP )
PPT file and images for Extended Fig. 2.
Supplementary Data 16 (download ZIP )
PPT file and images for Extended Fig. 3.
Supplementary Data 17 (download ZIP )
PPT file and images for Extended Fig. 4.
Supplementary Data 18 (download ZIP )
PPT file and images for Extended Fig. 5.
Supplementary Data 19 (download ZIP )
PPT file and images for Extended Fig. 6.
Supplementary Data 20 (download ZIP )
PPT file and images for Extended Fig. 7.
Supplementary Data 21 (download ZIP )
PPT files for supplementary figures.
Source data
Source Data Fig. 1 (download XLSX )
Statistical Source data
Source Data Fig. 2 (download XLSX )
Statistical Source data
Source Data Fig. 3 (download XLSX )
Statistical Source data
Source Data Fig. 4 (download XLSX )
Statistical Source data
Source Data Fig. 5 (download XLSX )
Statistical Source data
Source Data Fig. 6 (download XLSX )
Statistical Source data
Source Data Fig. 7 (download XLSX )
Statistical Source data
Source Data Extended Data Fig. 1 (download XLSX )
Statistical Source data
Source Data Extended Data Fig. 2 (download XLSX )
Statistical Source data
Source Data Extended Data Fig. 3 (download XLSX )
Statistical Source data
Source Data Extended Data Fig. 4 (download XLSX )
Statistical Source data
Source Data Extended Data Fig. 5 (download XLSX )
Statistical Source data
Source Data Extended Data Fig. 6 (download XLSX )
Statistical Source data
Source Data Extended Data Fig. 7 (download XLSX )
Statistical Source data
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, T., Li, Y., Pan, L. et al. Brain-wide alterations revealed by spatial transcriptomics and proteomics in COVID-19 infection. Nat Aging 4, 1598–1618 (2024). https://doi.org/10.1038/s43587-024-00730-z
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s43587-024-00730-z
This article is cited by
-
Advances in the pathophysiological study of brain development: application of cerebral organoid combined with Spatial omics technology
Stem Cell Research & Therapy (2026)
-
Spatial architecture of development and disease
Nature Reviews Genetics (2026)
-
The synergistic role of viral infection and immune response in the pathogenesis of facial palsy
Journal of NeuroVirology (2025)