Abstract
Maternal exposure to a Western-type diet (WD) increases the susceptibility of adult offspring to atherosclerosis, partly because fetal endothelial cells (ECs) become dysfunctional and inflamed due to risk factors transmitted via maternal–fetal blood exchange. However, the underlying mechanisms remain unclear. Here we show that maternal WD accelerates atherogenesis in adult offspring mice by regulating chromatin dynamics through activator protein-1 (AP-1) in aortic ECs, inducing inflammatory memory at the chromatin level. We found that 27-hydroxycholesterol is involved in memory establishment and also acts as a secondary stimulator, amplifying the expression of inflammatory factors and enhancing the enrichment of AP-1/p300 and H3K27ac in ECs. Inhibiting AP-1 binding to chromatin reduced the inflammatory response in human umbilical vein ECs from mothers with hypercholesterolemia and decreased atherogenesis in offspring mice exposed to maternal WD. Our findings demonstrate that maternal WD exacerbates EC dysfunction and atherosclerosis in adult offspring by inducing AP-1-associated epigenetic memory, which increases chromatin accessibility to inflammatory genes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Code availability
Analyses were performed using standard R packages. No custom analysis software was created to perform these analyses. Analyses were performed using published R packages that are cited within the text and Methods sections.
References
Fuster, V. Top 10 cardiovascular therapies and interventions for the next decade. Nat. Rev. Cardiol. 11, 671–683 (2014).
Soehnlein, O. et al. Targeting inflammation in atherosclerosis—from experimental insights to the clinic. Nat. Rev. Drug Discov. 20, 589–610 (2021).
Cooper, C. David Barker (1938–2013). Nature 502, 304 (2013).
Reynolds, R. M. et al. Maternal obesity during pregnancy and premature mortality from cardiovascular event in adult offspring: follow-up of 1 323 275 person years. BMJ 347, f4539 (2013).
Cao, N. et al. Prenatal lipopolysaccharides exposure induces transgenerational inheritance of hypertension. Circulation 146, 1082–1095 (2022).
Napoli, C. et al. Maternal hypercholesterolemia during pregnancy promotes early atherogenesis in LDL receptor-deficient mice and alters aortic gene expression determined by microarray. Circulation 105, 1360–1367 (2002).
Wakana, N. et al. Maternal high-fat diet exaggerates atherosclerosis in adult offspring by augmenting periaortic adipose tissue-specific proinflammatory response. Arterioscler. Thromb. Vasc. Biol. 35, 558–569 (2015).
de Nigris, F. et al. Epigenetic hallmarks of fetal early atherosclerotic lesions in humans. JAMA Cardiol. 3, 1184–1191 (2018).
Napoli, C. et al. Primary prevention of atherosclerosis: a clinical challenge for the reversal of epigenetic mechanisms? Circulation 125, 2363–2373 (2012).
Ordovas, J. M. et al. Epigenetics and cardiovascular disease. Nat. Rev. Cardiol. 7, 510–519 (2010).
Klemm, S. L. et al. Chromatin accessibility and the regulatory epigenome. Nat. Rev. Genet. 20, 207–220 (2019).
Chen, S. Y. et al. Maternal hypercholesterolemia exacerbates atherosclerosis lesions in female offspring through potentiating macrophage polarization toward an inflammatory M1 phenotype. J. Nutr. Biochem. 90, 108575 (2021).
Christ, A. et al. Western diet triggers NLRP3-dependent innate immune reprogramming. Cell 172, 162–175 (2018).
Drummer, C. T. et al. Trained immunity and reactivity of macrophages and endothelial cells. Arterioscler. Thromb. Vasc. Biol. 41, 1032–1046 (2021).
Levenson, J. M. et al. Epigenetic mechanisms in memory formation. Nat. Rev. Neurosci. 6, 108–118 (2005).
Gao, P. et al. Salt-induced hepatic inflammatory memory contributes to cardiovascular damage through epigenetic modulation of SIRT3. Circulation 145, 375–391 (2022).
Larsen, S. B. et al. Establishment, maintenance, and recall of inflammatory memory. Cell Stem Cell 28, 1758–1774 (2021).
Umetani, M. et al. The cholesterol metabolite 27-hydroxycholesterol promotes atherosclerosis via proinflammatory processes mediated by estrogen receptor alpha. Cell Metab. 20, 172–182 (2014).
Yu, L. et al. Macrophage-to-endothelial cell crosstalk by the cholesterol metabolite 27HC promotes atherosclerosis in male mice. Nat. Commun. 14, 4101 (2023).
Aikawa, Y. et al. Treatment of arthritis with a selective inhibitor of c-Fos/activator protein-1. Nat. Biotechnol. 26, 817–823 (2008).
Samir, P. et al. Food for training—Western diet and inflammatory memory. Cell Metab. 27, 481–482 (2018).
Kalna, V. et al. The transcription factor ERG regulates super-enhancers associated with an endothelial-specific gene expression program. Circ. Res. 124, 1337–1349 (2019).
Kim, B. K. et al. p300 cooperates with c-Jun and PARP-1 at the p300 binding site to activate RhoB transcription in NSC126188-mediated apoptosis. Biochim. Biophys. Acta 1839, 364–373 (2014).
Ma, Y. et al. Platelet-mimicking therapeutic system for noninvasive mitigation of the progression of atherosclerotic plaques. Adv. Sci. (Weinh.) 8, 2004128 (2021).
Barrett, T. J. et al. Chronic stress primes innate immune responses in mice and humans. Cell Rep. 36, 109595 (2021).
Beaulieu, A. M. Transcriptional and epigenetic regulation of memory nk cell responses. Immunol. Rev. 300, 125–133 (2021).
Sohrabi, Y. et al. OxLDL-mediated immunologic memory in endothelial cells. J. Mol. Cell. Cardiol. 146, 121–132 (2020).
Stark, R. J. et al. Endothelial cell tolerance to lipopolysaccharide challenge is induced by monophosphoryl lipid A. Clin. Sci. (Lond.) 130, 451–461 (2016).
Madrigal, P. et al. Ap-1 takes centre stage in enhancer chromatin dynamics. Trends Cell Biol. 28, 509–511 (2018).
Beisaw, A. et al. Ap-1 contributes to chromatin accessibility to promote sarcomere disassembly and cardiomyocyte protrusion during zebrafish heart regeneration. Circ. Res. 126, 1760–1778 (2020).
Luquain-Costaz, C. et al. Oxysterols in vascular cells and role in atherosclerosis. Adv. Exp. Med. Biol. 1440, 213–229 (2024).
Canfran-Duque, A. et al. Macrophage-derived 25-hydroxycholesterol promotes vascular inflammation, atherogenesis, and lesion remodeling. Circulation 147, 388–408 (2023).
Umetani, M. et al. 27-hydroxycholesterol is an endogenous SERM that inhibits the cardiovascular effects of estrogen. Nat. Med. 13, 1185–1192 (2007).
Burke, K. T. et al. Transport of maternal cholesterol to the fetus is affected by maternal plasma cholesterol concentrations in the golden Syrian hamster. J. Lipid Res. 50, 1146–1155 (2009).
Palinski, W. Maternal–fetal cholesterol transport in the placenta: good, bad, and target for modulation. Circ. Res. 104, 569–571 (2009).
Stefulj, J. et al. Human endothelial cells of the placental barrier efficiently deliver cholesterol to the fetal circulation via ABCA1 and ABCG1. Circ. Res. 104, 600–608 (2009).
Vurusaner, B. et al. The role of autophagy in survival response induced by 27-hydroxycholesterol in human promonocytic cells. Redox Biol. 17, 400–410 (2018).
Russell, D. W. The enzymes, regulation, and genetics of bile acid synthesis. Annu. Rev. Biochem. 72, 137–174 (2003).
Saeed, S. et al. Epigenetic programming of monocyte-to-macrophage differentiation and trained innate immunity. Science 345, 1251086 (2014).
Jin, Q. et al. Distinct roles of GCN5/PCAF-mediated H3K9ac and CBP/p300-mediated H3K18/27ac in nuclear receptor transactivation. EMBO J. 30, 249–262 (2011).
Hata, M. et al. Past history of obesity triggers persistent epigenetic changes in innate immunity and exacerbates neuroinflammation. Science 379, 45–62 (2023).
Smedts, H. P. et al. A derangement of the maternal lipid profile is associated with an elevated risk of congenital heart disease in the offspring. Nutr. Metab. Cardiovasc. Dis. 22, 477–485 (2012).
Versmissen, J. et al. Maternal inheritance of familial hypercholesterolemia caused by the V408M low-density lipoprotein receptor mutation increases mortality. Atherosclerosis 219, 690–693 (2011).
Napoli, C. et al. Maternal hypercholesterolemia enhances atherogenesis in normocholesterolemic rabbits, which is inhibited by antioxidant or lipid-lowering intervention during pregnancy: an experimental model of atherogenic mechanisms in human fetuses. Circ. Res. 87, 946–952 (2000).
Palinski, W. et al. The fetal origins of atherosclerosis: maternal hypercholesterolemia, and cholesterol-lowering or antioxidant treatment during pregnancy influence in utero programming and postnatal susceptibility to atherogenesis. FASEB J. 16, 1348–1360 (2002).
Nelson, E. R. et al. 27-hydroxycholesterol links hypercholesterolemia and breast cancer pathophysiology. Science 342, 1094–1098 (2013).
Nam, D. et al. Partial carotid ligation is a model of acutely induced disturbed flow, leading to rapid endothelial dysfunction and atherosclerosis. Am. J. Physiol. Heart Circ. Physiol. 297, H1535–H1543 (2009).
Chen, J. et al. Isolation and culture of murine aortic cells and RNA isolation of aortic intima and media: rapid and optimized approaches for atherosclerosis research. Atherosclerosis 347, 39–46 (2022).
Cao, X. et al. Chromatin accessibility dynamics dictate renal tubular epithelial cell response to injury. Nat. Commun. 13, 7322 (2022).
Acknowledgements
This work was supported by grants from the National Natural Science Foundation of China (81925003 and 82130014 to D.A.) and a grant from the National Key R&D Program of China (2019YFA0802502 to D.A.). Illustration items in the figures were created with BioRender.
Author information
Authors and Affiliations
Contributions
K.L., W.Q., L.S. and D.A. designed the experiments. K.L., W.Q., F.Z., W.Z., H.L., M.Q., X.C., R.L. and W.S. performed research. K.L., F.Z., W.Q. and H.J. analyzed data. K.L., Z.X., S.B. and C.H. collected the samples. M.Z., J.Z., J.D., H.J. and Y.C. oversaw the project and provided resources. K.L., W.Q., F.Z. and D.A. wrote the paper. All authors read and approved the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cardiovascular Research thanks Philip Shaul and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
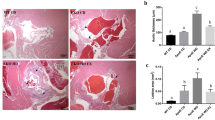
Extended Data Fig. 1 Maternal WD exacerbates atherosclerosis in offspring Ldlr-/- mice.
a, Representative ultrasound images of LCA from mice were obtained on day 0 and 14 after surgery. They revealed that partial ligation induced flow reversal in LCA during diastole. b, c, IPGTT of female (b; n = 9) and male (c; n = 8) mice in each group at 8 weeks of age. d, e, Plasma levels of TG, CHO, LDL-c, and HDL-c in female (d) and male (e) mice at 8-week-old. n = 6. f, g, Plasma levels of female (f) and male (g) mice at 11-week-old. n = 6. h, IPGTT of female mice in each group at 16 weeks of age. n = 9. i, Plasma levels of female mice at 16-week-old. n = 9. j, IPGTT of male mice in each group at 16 weeks of age. n = 8. k, Plasma levels of male mice at 16-week-old. n = 9 vs. 8. Data are presented as the mean ± SEM. An unpaired two-tailed Student’s t-test was used to determine statistical significance.
Extended Data Fig. 2 Distinct transcriptional modules are linked to WD stimulation in MAECs.
a, Diagram of MAECs sorting and transcriptomic analysis (SMART-seq). b, Principal component analysis (PCA) of replicates from both CDM-CDO, WDM-CDO, CDM-WDO and WDM-WDO groups in the dataset. c, Volcano plot of differentially expressed genes (WDM-CDO vs. CDM-CDO). |log2FC|≥ 1, p.adj < 0.05, n = 3. d, Heatmap showing mRNA levels for genes within the Up (above) and Down (below) groups. e, Scaled expression of Up and Down genes in each of the four groups. f, Bubble plot showing the GO BP and KEGG pathways by analyzing the Up and Down genes in MAECs. Gene expression is depicted in violin plots, displaying maximal, minimal, median values, and the 75th and 25th percentiles(e). Ordinary one-way ANOVA with Tukey’s multiple comparison test was used to determine statistical significance. p.adj: adjusted p-value.
Extended Data Fig. 3 Maternal WD endows MAECs inflammation memory in offspring at the chromatin level.
a, Schematic figure showing the process of chromatin preparation (ATAC-seq) in MAECs. b, Genomic distribution of differential accessibility regions in MAECs from four groups. c, Multivariate correlation matrix displayed graphically. d, Heatmaps (left) showing ATAC signals for each specified peak cluster in the indicated four groups. The signal strengths are denoted by color intensities. Line graphs (right) indicating differences in chromatin accessibility between WDM-CDO vs. CDM-CDO and WDM-WDO vs. CDM-WDO groups. e, Bubble plot showing important GO BP for each module. Significance calculated by hypergeometric test for genes in different modules and adjusted by Benjamini-Hochberg method.
Extended Data Fig. 4 AP-1 TF, accompanied by H3K27ac modification, drives memory recall in MAECs.
a, Motif enrichment analysis of MAECs genomic regions in module IV/V/VI/VII. b, Relative mRNA levels of Jun in CDM and WDM mice. n = 6. c, Relative mRNA levels of Jun in CDM-CDO, WDM-CDO, CDM-WDO and WDM-WDO mice. n = 3. Data are presented as the mean ± SEM. An unpaired two-tailed Student’s t-test (b) and ordinary two-way ANOVA with Sidak’s multiple comparison test (c) were used to determine statistical significance.
Extended Data Fig. 5 27-HC plays a critical role in epigenetic memory establishment in vivo.
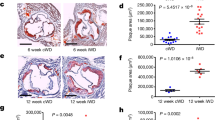
a, Plasma level of CHO in maternal mice just after the end of lactation. b, Plasma level of CHO in freshly weaned offspring mice. c, Relative mRNA levels of Tnf, Nlrp3 and Ccl3 in CDM and WDM mice. n = 6. d, C57BL/6 mice were subcutaneously treated with 27-OH (20 mg/kg/day) for 1 week, and CT (130 mg/kg) was subcutaneously injected daily on days 6 and 7 after 27-HC injection. Relative plasma levels of 27-HC in male C57BL/6 mice. n = 5. e, Relative plasma levels of 27-HC in maternal mice after cessation of lactation. n = 8. f, Relative plasma levels of 27-HC in female (left) and male (right) offspring mice at 3-week-old for four groups. n = 6. g, Representative images of male offspring mice arterial tissues isolated to examine atherosclerotic lesions after 3 weeks post-ligation. Scale bar, 2 mm. h, i, Representative HE staining images (h) and quantification of lesion areas (i) in ligated LCA sections. Dashed orange boxes represent LCA. n = 6. Scale bar, 100 μm. j, k, Relative plasma TNF (j) and CCL3 (k) level in CDM-WDO and WDM-WDO Ldlr-/- mice administered subcutaneously with Vec or CT. n = 6. l, Plasma levels of TG, CHO, LDL-c, and HDL-c in male mice at 11-week-old for four groups. n = 6. Data are presented as the mean ± SEM. An unpaired two-tailed Student’s t-test (a-c), ordinary one-way ANOVA with Sidak’s multiple comparison test (d), and ordinary two-way ANOVA with Sidak’s multiple comparison test (e, f, and i-l) were used to determine statistical significance.
Extended Data Fig. 6 27-HC plays a critical role in epigenetic memory establishment in vitro.
a, Relative ERα (a, left), NLRP3 (a, middle) and CCL3 (a, right) mRNA levels in HUVECs treated with siNC or siERα with or without 27-HC treatment. n = 6. b, c, CUT&RUN-qPCR analysis showing the enrichment of JUN (b) and H3K27ac (c) at NLRP3 (left) and CCL3 (right) in HUVECs treated with siNC or siERα with or without 27-HC treatment. n = 5. d, Relative NLRP3 (left) and CCL3 (right) mRNA levels in HUVECs treated with DMSO or T-5224 (20 μM) with or without 27-HC treatment. n = 6. e, f, CUT&RUN-qPCR analysis showing the enrichment of JUN (e) and H3K27ac (f) at NLRP3 (left) and CCL3 (right) in HUVECs treated with DMSO or T-5224 with or without 27-HC treatment. n = 5. Data are presented as the mean ± SEM. Ordinary two-way ANOVA with Sidak’s multiple comparison test were used to determine statistical significance.
Extended Data Fig. 7 T-5224 blocks 27-HC-stimulated inflammatory memory in MH HUVECs.
a, Relative mRNA levels of TNF, NLRP3, and CCL3 in P1 and P6 HUVECs from control pregnant patients (Ctrl; pink) and those with MH (MH; blue). n = 6. b, CUT&RUN-qPCR analyses showing the interaction of JUN with the gene bodies of 3 genes in Ctrl and MH HUVECs. n = 6. c, d, In the presence of 27-HC, CUT&RUN-qPCR analysis showing the enrichment of JUN (c) and H3K27ac (d) at TNF, NLRP3, and CCL3 in Ctrl and MH HUVECs treated with siNC or siERα. n = 5. e, Relative TNF (left) and CCL3 (right) levels by ELISA assay in Ctrl and MH HUVECs treated with DMSO or T-5224 with or without 27-HC treatment. n = 6. f, Representative western blot (above) and quantification of NLRP3 protein levels (below) in Ctrl and MH HUVECs treated with DMSO or T-5224 with or without 27-HC treatment, normalized to GAPDH. n = 6. g, Relative TNF, NLRP3, and CCL3 mRNA levels in Ctrl and MH HUVECs treated with DMSO or T-5224 with or without ox-LDL treatment. n = 6. h, CUT&RUN-qPCR analysis showing the enrichment of JUN at TNF, NLRP3, and CCL3 in Ctrl and MH HUVECs treated with DMSO or T-5224 with or without ox-LDL treatment. n = 6. i, Ctrl and MH HUVECs were subjected to 27-HC treatment for 24 hours. The lysate was immunoprecipitated with p300, followed by western blots with the indicated antibodies. n = 3. Data are presented as the mean ± SEM. Ordinary one-way ANOVA with Sidak’s multiple comparison test (a-d) and ordinary two-way ANOVA with Sidak’s multiple comparison test (e-h) were used to determine statistical significance.
Extended Data Fig. 8 T-5224 impairs epigenetic memory and alleviates atherosclerosis.
a, b, Relative plasma Tnf (a) and Ccl3 (b) level in CDM-CDO and WDM-CDO Ldlr-/- mice administered intragastrically with CMC-Na or T-5224. n = 6. c, Plasma levels of TG, CHO, LDL-c, and HDL-c in female mice at 11-week-old for four groups. n = 7. Data are presented as the mean ± SEM. Ordinary two-way ANOVA with Sidak’s multiple comparison test were used to determine statistical significance.
Extended Data Fig. 9 Mechanistic diagram of the effect of maternal high-fat diet on atherosclerosis in offspring.
a, The figure illustrates that maternal WD aggravating EC dysfunction and atherosclerosis in adult offspring acquires AP-1-associated epigenetic memory to increase chromatin accessibility of inflammatory genes, which is triggered by an elevation of 27-HC content.
Supplementary information
Supplementary Tables 1–4, (download PDF )
Supplementary Data 1 and Supplementary Fig. 1.
Supplementary Table 1 (download XLSX )
Patients without or with hyperlipemia. Supplementary Table 2 Primer sequences used in qPCR. Supplementary Table 3 JUN, H3K27ac and p300 CUT&RUN–qPCR. Supplementary Table 4 Target sequences for knockdown.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig./Table 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig./Table 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig./Table 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig./Table 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig./Table 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig./Table 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig./Table 7 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, K., Qian, W., Zhang, F. et al. Maternal high-fat diet exacerbates atherosclerosis development in offspring through epigenetic memory. Nat Cardiovasc Res 4, 362–379 (2025). https://doi.org/10.1038/s44161-025-00622-4
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44161-025-00622-4
This article is cited by
-
27-hydroxycholesterol and endothelial cell immune memory link maternal hypercholesterolemia with atherosclerosis in the offspring
Nature Cardiovascular Research (2025)