Abstract
Intracellular accumulation of α-synuclein (αSyn) aggregates is a hallmark of synucleinopathies, such as Parkinson’s disease (PD) and multiple system atrophy (MSA). In MSA, αSyn aggregates form glial cytoplasmic inclusions (GCIs) in oligodendrocytes, despite their low expression of αSyn. Here, we demonstrate that neuron-to-oligodendrocyte propagation of αSyn, via Toll-like receptor 2 (TLR2) contributes to GCI formation. Male transgenic mice expressing the A53T mutant human αSyn exclusively in neurons, and the preformed fibril injection model exhibited MSA-like pathology, including GCI formation, gliosis, and neuroinflammation in the white matter. Notably, administration of NM-101, an anti-TLR2 antibody, significantly alleviated these pathological features. Transcriptome analyses revealed demyelination-related features in MSA oligodendrocytes and experimental models. Elevated TLR2 expression in MSA oligodendrocytes inversely correlated with MBP expression, and the correlation was absent in PD. In the transgenic mouse model, NM-101 administration rescued the demyelination phenotype. These findings highlight anti-TLR2 immunotherapy as a potential disease-modifying approach for MSA.
Similar content being viewed by others
Introduction
Many neurodegenerative diseases, such as Parkinson’s disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA), are characterized by abnormal aggregation of α-synuclein (αSyn) and are thus classified as synucleinopathies1. In PD and DLB, αSyn accumulates within neurons as Lewy bodies (LBs) and Lewy neurites (LNs), whereas in MSA, αSyn predominantly accumulates in oligodendrocytes, forming glial cytoplasmic inclusions (GCIs)1. Clinically, these diseases exhibit overlapping symptoms, particularly Parkinsonian motor symptoms. Among these synucleinopathies, MSA is the most rapidly progressing disease, presenting with Parkinsonism, cerebellar ataxia, and autonomic syndrome2.
There is a strong correlation between αSyn aggregation and clinical symptoms3. For example, in PD, αSyn aggregates spread from distinct regions to more widespread brain areas as the disease progresses, with the complexity of clinical symptoms positively correlating with aggregate spreading4. Previous studies have demonstrated that αSyn spreading is mediated by cell-to-cell propagation via exo/endocytosis and tunneling nanotube5,6. Additionally, an increasing body of evidence suggests that this propagation is mediated by cell-surface receptors for αSyn7. Among these receptors, Toll-like receptor 2 (TLR2) is the only receptor that has been shown to mediate both neuron-to-neuron propagation of αSyn and αSyn-induced microglial activation8,9,10.
αSyn is highly expressed in neurons. Therefore, in PD and DLB, αSyn aggregates deposit in neurons in forms of LBs and LNs in a cell-autonomous fashion. In MSA, however, the origin of αSyn in GCIs has been an enigma. Both conventional RT-PCR and single-nucleus RNA sequencing have shown that αSyn expression is either undetectable or extremely low in oligodendrocytes11,12. In vitro and in vivo studies have demonstrated that the expression of αSyn was decreased during oligodendrocyte maturation13,14. Since neurons secrete αSyn aggregates, we hypothesized that neuron-originated αSyn can be transferred to oligodendrocytes and form GCIs. Here, we demonstrate in both in vitro and in vivo models that αSyn can be propagated from neurons to oligodendrocytes through TLR2, leading to demyelination in oligodendrocytes, and that anti-TLR2 treatment can inhibit the GCI formation and demyelination.
Results
αSyn can be propagated from neurons to oligodendrocytes
Previous studies have reported limited expression of SNCA in oligodendrocytes13,14. To further investigate this, we analyzed single-nuclei transcriptomes from healthy controls to assess SNCA expression levels in human neurons and oligodendrocytes from Adams et al.15. As shown in Fig. 1a, SNCA expression in oligodendrocytes was markedly lower than in neurons. To further evaluate the levels of expression of SNCA in oligodendrocytes, we analyzed its expression in oligodendrocytes and neurons isolated from post-mortem brain tissues. Consistent with the single-nuclei transcriptome data, qRT-PCR data demonstrated that SNCA expression in oligodendrocytes isolated from post-mortem brains of individuals with primary age-related tauopathy (PART) was also substantially lower than in neurons from the same brain (Fig. 1b).
a Comparison of the expression level of SNCA in neurons and oligodendrocytes obtained from young and aged healthy controls. The dataset was obtained from Adams et al.15. b Fold change of expression values of SNCA mRNA in neurons and oligodendrocytes isolated from the cerebellum from control (PART). c Experimental scheme. Oligodendrocytes were differentiated from hNSCs after treatment with either αSyn CM (αSCM) or control LacZ CM (β-galactosidase conditioned media; LZCM) and subsequently incubated for an additional 3 days. d Representative confocal images showing the formation of pS129-positive inclusion bodies in MBP-positive oligodendrocytes. e Quantification of αSyn puncta containing cell (%). Statistical significance was evaluated using two-sided unpaired t-tests with Welch’s correction. f Quantification of relative αSyn intensity in MBP-positive oligodendrocytes. Statistical significance was evaluated using two-sided unpaired t-tests with Welch’s correction. g Colocalization analysis between αSyn and pS129. M1: fraction of αSyn, and M2: fraction of pS129. h Characterization of αSyn-positive inclusion bodies in oligodendrocytes, revealing co-localization with TPPP/p25α and ubiquitin. i Quantification of TPPP/p25α–positive αSyn puncta. j Quantification of ubiquitin-positive αSyn puncta. k Experimental scheme. The deposition of pS129-positive GCIs was examined in the white matter of WT and hA53T Tg mice. l Relative expression of SNCA and oligodendrocyte markers in oligodendrocytes from WT and hA53T Tg mice. No leaky expression of SNCA was detected in oligodendrocytes from hA53T Tg mice, which express SNCA exclusively in neurons. m Representative confocal images showing accumulation of GCIs in hA53T Tg mice. n Quantification of the percentage of TPPP-positive oligodendrocytes containing pS129 in hA53T Tg mice. Statistical significance was evaluated using two-sided unpaired t-tests with Welch’s correction. Scale: 20 μm in (d, h), 10 μm in (m). All data are presented as the mean ± SEM. n = 3 biologically independent samples for each in (e, i, j, l). n = 6 biologically independent samples for each in (f). n = 4 biologically independent samples for each in (g). n = 7 biologically independent samples for WT, and n = 8 biologically independent samples for hA53T Tg mice in (n). c Created in BioRender. Bae, E. (2025) https://BioRender.com/v0eyjrp. k Created in BioRender. Bae, E. (2025) https://BioRender.com/hup3r8o.
To test the hypothesis that oligodendrocytes develop GCI-like inclusions through neuron-to-oligodendrocyte propagation of αSyn, we generated an oligodendrocyte-rich culture from human neural stem cells (hNSC) and treated these cells with conditioned medium from differentiated SH-SY5Y cells overexpressing αSyn (Fig. 1c). The conditioned medium contains secreted α-synuclein (αSyn) species, including both monomeric and oligomeric forms of αSyn (Supplementary Fig. 1a). To assess potential viral contamination in αSyn conditioned medium (αSCM), RT-PCR was performed targeting human SNCA using the undiluted conditioned medium as the template. No amplification of SNCA was observed in αSCM (Supplementary Fig. 1b). Oligodendrocytes exposed to αSyn-containing conditioned medium (αSCM) exhibited diverse patterns of intracellular αSyn accumulation (Fig. 1d), including dispersed small puncta, dense puncta, and thread-like inclusions, similar to those observed in the brains of MSA patients16 (Supplementary Fig. 2a, b). Treatment with αSCM led to an increase in αSyn signal intensity and a higher proportion of αSyn-positive cells (Fig. 1d–f). These αSyn-positive aggregates were partially immunoreactive for the Ser129-phosphorylated form of αSyn (pS129) (Fig. 1g and Supplementary Fig. 2a, b), as well as for TPPP/p25α and ubiquitin (Fig. 1h–j), markers characteristic of glial cytoplasmic inclusions (GCIs) in MSA. Z-stack imaging revealed that these aggregates were localized within intracellular regions, thereby excluding the possibility that the αSyn signals arose from nonspecific adhesion of sticky αSyn in αSCM to the cell surface. Instead, these findings suggest that internalized αSyn from αSCM can form intracellular aggregates in NSC-derived oligodendrocytes (Supplementary Fig. 2c and Supplementary Movie 1). To further validate these findings, we employed primary mouse oligodendrocyte cultures. In these cultures, αSCM treatment was sufficient to induce GCI-like inclusions in MBP-positive oligodendrocytes (Supplementary Fig. 2d and Supplementary Movie 2). To extend this observation in vivo and assess neuron-to-oligodendrocyte propagation of αSyn, we examined white matter oligodendrocytes from transgenic (Tg) mice overexpressing human A53T (hA53T) αSyn under the control of a neuron-specific promoter (Fig. 1k). To rule out the possibility of PrP promoter-driven expression of human αSyn in oligodendrocytes in this mouse, we evaluated the expression of human SNCA mRNA in oligodendrocytes isolated from Tg mice using laser capture microdissection. The SNCA expression levels in oligodendrocytes from Tg mice were comparable to those in wild-type (WT) mice, demonstrating that PrP promoter-driven expression of human αSyn is negligible in oligodendrocytes (Fig. 1l). Since these animals express human αSyn only in neurons (Fig. 1l), any human αSyn detected in oligodendrocytes would have originated from neurons. In these Tg animals, we found the accumulation of human phosphorylated αSyn (pS129) in TPPP/p25α-positive oligodendrocytes in the white matter regions, which is similar to those observed in the brains (Fig. 1m). Given that white matter primarily consists of myelinated neuronal axons, it is essential to differentiate glial inclusions from neuronal ones. To exclude the possibility that observed inclusions were located within neuronal processes, we analyzed pS129 signals within mature oligodendrocytes, identified by immunoreactivity to TPPP, a marker characterized by a distinct perinuclear staining pattern (Supplementary Fig. 3a, b and Supplementary Movie 3). Among the 1565 pS129-positive signals per mm2 detected in the white matter, 12.32% were localized within TPPP-positive oligodendrocytes (Supplementary Fig. 3c). Additionally, approximately 8.4% of TPPP-positive oligodendrocytes in the white matter displayed pS129-positive glial cytoplasmic inclusion (GCI)-like structures in Tg mice (Fig. 1n). These findings provide both in vitro and in vivo evidence supporting the hypothesis that neuron-secreted αSyn can propagate into oligodendrocytes and form GCI-like inclusions.
Blockade of TLR2 inhibits the formation of GCI-like inclusions in hNSC-derived OLs
We have previously shown in various studies that TLR2 is a receptor for αSyn and that blockade of TLR2 could reverse neuron-to-neuron propagation of αSyn and αSyn-induced microglia activation10,17. To assess the potential role of TLR2 in MSA, we investigated changes in TLR2 expression levels in oligodendrocytes from MSA patients. Transcriptomic analyses of oligodendrocytes from MSA patients18 showed a significant increase in TLR2 transcript levels, compared to controls (Fig. 2a). To further confirm this finding, we isolated oligodendrocytes from cerebellar tissues of postmortem brains from either primary age-related tauopathy (PART; control) or MSA patients (Fig. 2b). Consistent with the previous transcriptomic findings, conventional qRT-PCR of oligodendrocytes isolated from postmortem brains demonstrated that levels of expression of TLR2 in MSA oligodendrocytes were higher than those in PART oligodendrocytes (control) (Fig. 2c). These results prompted us to hypothesize that TLR2 mediates the neuron-to-oligodendrocyte propagation of αSyn.
a Comparison of TLR2 expression levels in oligodendrocytes derived from healthy controls and MSA patients. The dataset was obtained from Piras et al.18. b Experimental scheme. Oligodendrocytes were isolated from cerebellar tissues of postmortem brains from PART (control) or MSA patients using anti-O4 MicroBeads. The cells collected in the eluate fraction were subsequently lysed to extract RNA for further analysis. c Relative expression levels of TLR2 in oligodendrocytes isolated from the cerebellum of control (PART) and MSA patients. d Experimental scheme. Oligodendrocytes were differentiated from hNSCs and treated with NM-101, a TLR2 antagonizing antibody, for 30 min prior to treatment with either αSyn CM (αSCM) or LacZ CM (LZCM) (used as a control), followed by an additional 3-day incubation. e Representative confocal images depicting the formation of αSyn-positive inclusion bodies in hNSC-derived oligodendrocytes. Scale: 10 μm. f Quantification of relative αSyn intensity in hNSC-derived oligodendrocytes. g Representative confocal images depicting the accumulation of αSyn-positive inclusion bodies in primary mouse oligodendrocytes. Scale: 20 μm. h Quantification of relative αSyn intensity in primary mouse oligodendrocytes. Statistical significance was evaluated using two-sided unpaired t-tests with Welch’s correction (a, c) and one-way ANOVA followed by Tukey’s post hoc comparisons between groups (f, h). All data are presented as the mean ± SEM. n = 25 biologically independent samples for each in (a). n = 8 biologically independent samples for PART, and n = 7 biologically independent samples for MSA in (c). n = 6 biologically independent samples for LZCM, n = 8 biologically independent samples for αSCM + IgG, and n = 6 biologically independent samples for αSCM + NM-101 in (f). n = 3 biologically independent samples for each in (h). b Created in BioRender. Bae, E. (2025) https://BioRender.com/z13e601. d Created in BioRender. Bae, E. (2025) https://BioRender.com/v0eyjrp.
Using NM-101, an antibody that blocks the ectodomain of TLR2, we determined whether the TLR2 blockade inhibits the neuron-to-oligodendrocyte propagation of αSyn and GCI formation. We evaluated the functional blockade of TLR2 by NM-101 using a reporter cell line, THP1-Blue NF-кB cells, to measure TLR2 activity. As shown in Supplementary Fig. 4a, the IC50 value of NM-101 for neuron-released αSyn-mediated TLR2 activation was calculated to be 13.516 ng/ml, approximately 0.1 nM (Supplementary Fig. 4a and Supplementary Table 1). We next evaluated whether NM-101 could inhibit the deposition of neuron-released αSyn in hNSC-derived oligodendrocytes. To this end, hNSC-derived oligodendrocytes were pretreated with NM-101 for 30 min prior to αSyn exposure (Fig. 2d). NM-101 treatment significantly suppressed the formation of GCI-like inclusions (Fig. 2e, f). To further validate this finding, primary mouse oligodendrocytes were also pretreated with NM-101 30 min before αSyn exposure. Similar to the results observed in hNSC-derived cells, NM-101 treatment attenuated the accumulation of GCI-like inclusions in primary mouse oligodendrocytes (Fig. 2g, h).
Effects of NM-101 immunotherapy on white matter pathology in αSyn Tg mice
To assess the pharmacokinetic (PK) properties of NM-101 in the periphery and brain, we analyzed NM-101 levels following intravenous administration to mice. αSyn Tg mice received either a single (30 mg/kg) or repeated injection (10, 30, or 100 mg/kg) of NM-101, and plasma and brain samples were collected as described in Supplementary Fig. 4b. In the repeated injection experiment, it was observed that the mean concentration increased per dose (from 10 to 100 mg/kg) and injection number (up to 5 times) in a proportional manner, with the exception of stationary concentration within the brain with repeated injections with a dose of 100 mg/kg (Supplementary Fig. 4c, d).
To investigate the changes of drug concentration in the brain and plasma across the different timepoints after a single injection of NM-101, 8-month-old αSyn Tg mice were administered a single intravenous injection of NM-101 at a dose of 30 mg/kg (Supplementary Fig. 4e). The concentration versus time profiles of NM-101 in plasma and brain are shown in Supplementary Fig. 4f, g, and the analyzed PK parameters are listed in Table 1. In plasma, NM-101 exhibited a mean clearance (CL) of 8.64 ml/day/kg, and a mean terminal volume of distribution (Vd) of 60 ml/kg, resulting in a plasma half-life (t1/2) of 7.57 days, which falls within the recently reported t1/2 range of 3–10 days for a variety of different human antibodies in mice following intravenous administration. The mean maximum concentration (Cmax) and area under the concentration-time curve from time zero to the last measurable timepoint (AUClast) were 637 μg/ml and 3930 μg × day/ml, respectively. The brain PK parameters of NM-101 showed that the mean brain Cmax was 0.77 μg/g and AUClast was 5.89 μg × day/g, with a brain-to-plasma ratio of 0.15%, calculated by comparing the mean AUClast values for brain and plasma.
Subsequently, we evaluated the in vivo efficacy of NM-101 on white matter pathology. Six-month-old αSyn Tg mice received weekly intravenous administrations of NM-101 (3 or 10 mg/kg) or placebo for 13 weeks. Following treatment, the animals were subject to neuropathological, biochemical, and behavioral analyses (Fig. 3a). To assess TLR2 pathway activation, we measured levels of downstream effectors in whole brain lysates. αSyn Tg mice exhibited elevated levels of phosphorylated Erk, which was attenuated by NM-101 treatment (10 mg/kg) (Supplementary Fig. 4h, i). Myd88, another TLR2 downstream effector, was also increased in αSyn Tg mice and reduced with NM-101 treatment, though this reduction did not reach statistical significance (Supplementary Fig. 4h, j). As shown in Fig. 1m, αSyn Tg mice exhibited GCI-like inclusions that were positive for pS129 within TPPP-positive oligodendrocytes of the white matter regions (Fig. 3b–d). Treatment with NM-101 led to a dose-dependent reduction in pS129 levels in the white matter of αSyn Tg mice compared to placebo-treated αSyn Tg mice, with a 23.9% decrease observed at 3 mg/kg and a 39.4% decrease at 10 mg/kg (Fig. 3c). Similarly, the relative intensity of pS129-positive GCI-like inclusions within TPPP-positive oligodendrocytes was reduced following NM-101 treatment, with decreases of 28.3% at 3 mg/kg and 38.4% at 10 mg/kg compared to placebo (Fig. 3d). These results suggest that αSyn can translocate from neurons to oligodendrocytes to form GCIs, and that NM-101 treatment can inhibit GCI formation in oligodendrocytes. Correspondingly, immunoblotting experiments confirmed that NM-101 treatment significantly reduced the extent of insoluble αSyn in the white matter of αSyn Tg mice (Fig. 3e–g).
a Experimental scheme. Six-month old WT or hA53T Tg mice received weekly intravenous injections of either vehicle or NM-101 at 3 and 10 mg/kg for 4 months. This image was created in BioRender. Bae, E. (2025) https://BioRender.com/46xygey. b Representative confocal images depicting GCI formation in the cerebellum. c Quantification of relative pS129 intensity in the white matter. d Quantification of relative pS129 intensity in TPPP-positive oligodendrocytes. e Western blots showing the level of αSyn in cerebellar tissue lysate. Tx-soluble: Triton X-100 soluble fraction, Tx-insoluble: Triton X-100 insoluble fraction. f Quantification of the relative level of αSyn at approximately 15 kDa in the Triton X-100 insoluble fraction. g Quantification of the relative level of high molecular weight αSyn in the Triton X-100 insoluble fraction. h Representative confocal images showing Iba-1, a marker of microgliosis, and Cd68, a marker of activated microglia in the cerebellum. i Quantification of the relative Iba-1 intensity. j Quantification of the number of Iba-1-positive cells per mm2. k Quantification of the number of Cd68-positive cells per mm2. l Representative skeletonized images of microglia used for the Sholl analysis. m Quantification of the number of intersections in microglia. n Quantification of proportion of proximal intersections in microglia. o Representative confocal images showing GFAP, a marker of astrogliosis in the cerebellum. p Quantification of the relative GFAP intensity. q Quantification of the number of GFAP-positive cells per mm2. r Representative confocal images showing FLICA puncta in Olig2-positive cells within the cerebellum. s Quantification of the relative FLICA intensity. t Life span analysis. u Four-limb hanging test. Scale: 10 μm in (b), 20 μm in (h), 10 μm in (i), 50 μm in (o), 20 μm in (r). Statistical significance was evaluated using one-way ANOVA followed by Tukey’s post hoc test for comparisons (c, d, f, g, i, j, k, m, n, p, q, u) and Brown–Forsythe and Welch ANOVA with two-sided Dunnett’s T3 post hoc test (s). All data are presented as the mean ± SEM. n = 4 biologically independent samples for WT, and n = 5 biologically independent samples for hA53T Tg mice, 3 and 10 mg/kg in (c). n = 7 biologically independent samples for WT, and n = 8 biologically independent samples for hA53T Tg mice, 3 and 10 mg/kg in (d). n = 3 biologically independent samples for each in (f, g). n = 14 biologically independent samples for WT, n = 13 biologically independent samples for hA53T Tg mice, and n = 8 biologically independent samples for 3 and 10 mg/kg in (i). n = 8 biologically independent samples for each in (j). n = 7 biologically independent samples for WT, hA53T Tg mice, and 10 mg/kg and n = 6 biologically independent samples for 3 mg/kg in (k). n = 8 biologically independent samples for WT, n = 7 biologically independent samples for hA53T Tg mice, and 10 mg/kg, and n = 9 biologically independent samples for 3 mg/kg in (m, n). n = 8 biologically independent samples for each in (p, q, s). n = 20 biologically independent samples for WT, n = 16 biologically independent samples for hA53T Tg mice, 3 mg/kg and 10 mg/kg in (t). n = 22 biologically independent samples for WT, n = 8 biologically independent samples for hA53T Tg mice, and n = 16 biologically independent samples for 3 mg/kg and 10 mg/kg in (u).
To investigate the efficacy of NM-101 on glial pathology, we analyzed gliosis and inflammasome activation in αSyn Tg mice treated with either NM-101 or placebo. For gliosis analysis, the brain sections were immunostained with anti-Iba-1, anti-Cd68, or anti-GFAP antibodies. Compared to wild-type controls, αSyn Tg mice exhibited a 1.4-fold increase in Iba-1-positive signals and a 1.8-fold increase in Cd68 signals, both of which were attenuated following treatment with NM-101 at 10 mg/kg. A reduction in Iba-1 immunoreactivity was also observed in the 3 mg/kg NM-101 group; however, this difference did not reach statistical significance (Fig. 3h-j). Importantly, the number of Iba-1-positive microglia, which was increased 2.2-fold in αSyn Tg mice relative to controls, was significantly reduced by NM-101 treatment at 10 mg/kg (Fig. 3h, j). Furthermore, Cd68 signals, indicative of activated microglia with phagocytic activity, were fully normalized in both the 3 and 10 mg/kg NM-101 treatment groups (Fig. 3h, k). To quantify morphological changes in microglia, we performed Sholl analysis. αSyn Tg mice displayed a greater number of intersections, reflecting a shift toward a hypertrophic microglial phenotype with increased process ramification, consistent with observations in the PLP-αSyn Tg mouse model19 (Fig. 3l–n). This morphological change was attenuated in the 10 mg/kg NM-101 treatment group (Fig. 3l–n). Additionally, αSyn Tg mice exhibited significantly elevated GFAP intensity and the number of GFAP-positive cells in white matter regions (2.2-fold increase compared to wild-type), which was significantly reduced in the 10 mg/kg NM-101 treatment group (Fig. 3o–q).
To determine inflammasome activation in oligodendrocytes, we measured the levels of active caspase-1 using the fluorescent labeled inhibitor of caspases (FLICA) assay. αSyn Tg mice showed increased caspase-1 activity in Olig2-positive oligodendrocytes in the white matter regions. The increased caspase-1 activities were effectively dampened to basal levels by NM-101 administration in a dose-dependent manner (Fig. 3r, s).
Consistent with these pathological improvements, we confirmed increased survival rates with NM-101 administration in αSyn Tg mice. These mice typically begin to die at around 6 months of age, with a median survival time of 10 months. However, NM-101 treatment resulted in a remarkable 100% improvement in survival (Fig. 3t). We also determined the effects of NM-101 on motor deficits in αSyn Tg mice and found that the weakened limb muscle strength was ameliorated by NM-101 treatment (Fig. 3u).
To validate the therapeutic effects of NM-101 on αSyn-induced pathologies in oligodendrocytes within the white matter, we employed an intrastriatal injection model of αSyn preformed fibrils (PFFs). In line with Uemura et al.20, who showed that pSyn-positive glia emerged at 12 months post-injection (PI) and increased by 18 months PI as neuronal pathology declined, we assessed oligodendroglial pathology in the corpus callosum at 12 months PI. Two-month-old WT mice received intrastriatal injections of mouse αSyn PFFs, followed by weekly intravenous administration of NM-101 (3 or 10 mg/kg) or placebo over a 48-week period (Supplementary Fig. 5a). The injection of αSyn PFFs resulted in the deposition of pS129 in the TPPP-positive oligodendrocytes in the white matter. NM-101 treatment led to a dose-dependent reduction in pS129 in the white matter of αSyn PFFs-injected mice (Supplementary Fig. 5b, c). To specifically assess the effects of NM-101 on oligodendroglial αSyn deposition, we quantified pS129 signals within TPPP-positive cells on the ipsilateral side of the white matter. NM-101 treatment effectively abolished pS129-positive puncta in TPPP-positive cells at both 3 and 10 mg/kg doses (Supplementary Fig. 5b, d). Mice injected with αSyn PFFs exhibited an accumulation of high molecular weight of αSyn as well as pS129 in the insoluble fractions of cerebellar tissue lysates, which was significantly attenuated by NM-101 in a dose-dependent manner (Supplementary Fig. 5e–i). Consistent with results observed in αSyn Tg mice, PFFs-injected mice also showed increased gliosis. NM-101 treatment alleviated astrogliosis (Supplementary Fig. 5j, k) and showed a trend toward reduced microgliosis (Supplementary Fig. 5j, l) as well. Behavior assessments revealed that the NM-101 treatment improved αSyn-induced motor defects in PFFs-injected mice (Supplementary Fig. 5m–o).
Demyelination signatures in MSA patients and models
To examine the effects of αSyn aggregates on oligodendrocytes, hNSC-derived oligodendrocyte-enriched culture was exposed to either αSCM or control LZCM (β-galactosidase-containing conditioned media), and analyzed using single-cell RNA sequencing. A total of 12,547 cells were embedded using the UMAP algorithm. Based on the Panglao database21 and a two-tailed Fisher’s exact test, cells were annotated with astrocytes (AQP4, GFAP), neurons (NGRN), neural stem cells (STMN1, 2), oligodendrocytes (CNP, PLP1), and oligodendrocyte progenitor cells (OPC) (OLIG1) (Fig. 4a, b). To investigate transcriptomic differences between αSyn-treated oligodendrocyte lineage cells and controls, we isolated oligodendrocyte fractions and performed re-clustering (Fig. 4b, c). Based on established oligodendrocyte maturation markers, we identified and annotated four distinct oligodendroglial lineage cell types: mature oligodendrocytes (mature OLs), oligodendrocyte precursor cells (OPCs), proliferating OPCs, and immature oligodendrocytes (immature OLs) (Fig. 4c). Further analysis revealed that αSyn treatment led to a reduction in the proportion of mature oligodendrocytes (Fig. 4d). To characterize transcriptomic alterations specific to mature oligodendrocytes following αSyn treatment, we extracted and re-clustered the mature oligodendrocyte population using myelination-associated markers (Fig. 4e, f). We then examined the expression of genes involved in oligodendrocyte maturation and myelination. Myelinating oligodendrocytes exhibited more pronounced transcriptomic changes related to these processes compared to pre-myelinating oligodendrocytes upon αSyn exposure (Fig. 4g). Notably, a heatmap of log₂ fold changes in gene expression (relative to LZCM-treated controls) and violin plots showing gene expression levels in myelinating and pre-myelinating oligodendrocytes across treatment groups revealed marked downregulation of key myelination and maturation genes, including CNP, PLP1, GSTP1, and FOXO4, in αSyn-treated myelinating oligodendrocytes (Fig. 4g, h). In addition, αSyn treatment was associated with a modest upregulation of markers corresponding to OPCs, immature OLs, and neural stem cells (NSCs) (Fig. 4g). We also observed a significant reduction in the expression of NRG1, a key regulator of oligodendrocyte differentiation and myelination, specifically in αSyn-treated myelinating OLs (Fig. 4h).
a UMAP representation of cell subsets, with each dot corresponding to an individual cell. b Heatmap illustrating differences in clusters based on the most variable genes (FDR < 0.05). Cells were annotated according to feature genes. c UMAP representation depicting sub-clusters of oligodendrocyte lineage cells in the control group (LZCM) and the αSyn-treated group (αSCM), with each dot representing an individual cell. d Stacked bar plots displaying the percentages of sub-clusters of oligodendrocyte lineage cells in each group. e UMAP representation illustrating cell subsets within mature oligodendrocytes, with each dot representing an individual cell. f Dot plot displaying the percentage of expressed cells and the average expression levels of feature genes across each subpopulation. g Heatmap depicting the expression profiles of oligodendroglial marker genes. *p < 0.05, ****p < 0.00005. h Violin plots illustrating the differential expression of genes associated with oligodendrocyte maturation and myelination in the two subpopulations. i Bar plot indicating that the increased DEGs in myelinating oligodendrocytes are associated with the enrichment of oligodendrocyte progenitor cell markers. Statistical significance was evaluated using two-sided unpaired t-tests with Welch’s correction between groups (e). All data are presented as the mean ± SEM.
To further explore the effects of αSyn on myelinating OLs, we identified differentially expressed genes (DEGs) through iterative differential analysis in myelinating OLs using a pseudo-bulk approach. The upregulated DEGs showed significant enrichment in markers associated with oligodendrocyte progenitor cells (OPCs) (Fig. 4i), suggesting that the uptake and accumulation of αSyn aggregates disrupt both oligodendrocyte maturation and myelination.
To confirm the impact of αSyn accumulation in oligodendrocytes in vivo, we analyzed the transcriptome of GSTP1-positive oligodendrocytes within the white matter of αSyn Tg mice. Laser-captured oligodendrocytes from αSyn Tg mice displayed 296 DEGs compared to those from control mice. The most significant Gene Ontology (GO) term enriched in the downregulated DEGs was central nervous system myelination (Fig. 5a; Supplementary Tables 2; 3 and Supplementary Data 1), suggesting defects in myelination in oligodendrocytes in αSyn Tg mice. To assess gene expression changes in humans, we also analyzed published transcriptomic data from MSA patients and healthy controls18. This study compared gene expressions in laser-captured oligodendrocytes from patients and controls. Transcriptomic analysis of MSA patients revealed 747 DEGs compared to healthy controls. These DEGs showed a reduction in genes related to neuronal ensheathment (Fig. 5b; Supplementary Tables 4; 5, and Supplementary Data 2), findings that align with our transcriptomic data from the αSyn Tg mice. There were 195 common DEGs identified from these two analyses (αSyn Tg mouse vs. WT, and MSA patients vs. Controls) (Fig. 5c; Supplementary Data 1 and 2). GO analysis of these 195 common DEGs identified reduced central nervous system myelination as a prominent feature. Specifically, genes that are involved in myelination and oligodendrocyte differentiation, such as MBP, PLP1, and SOX10, were commonly downregulated in oligodendrocytes from both MSA patients and αSyn Tg mice (Fig. 5d; Supplementary Tables 6; 7; Supplementary Data 1 and 2). Consistent with the transcriptome analyses, immunofluorescence analysis of tissue sections showed reduced PLP expression in the white matter of MSA patients (Fig. 5e, f).
a Bar plot illustrating the significant GO enrichment terms in biological processes based on DEGs from oligodendrocytes in hA53T Tg mice. b Bar plot depicting the significant GO enrichment terms in biological processes derived from DEGs in oligodendrocytes obtained from MSA patients. c Venn diagram showing the number of DEGs in oligodendrocytes from both MSA patients and hA53T Tg mice. d Bar plot displaying the significant GO enrichment terms in biological processes using the common DEGs from (c). e Representative confocal images showing PLP expression in the cerebellum from PART controls and MSA patients. Scale: 20 μm. f quantification of PLP in the cerebellum from PART and MSA patients. Statistical significance was assessed using the two-tailed Student’s t tests with Tukey’s post hoc comparison. All data are presented as the mean ± SEM. n = 4 biologically independent samples for each in (f). g–j Dot plot representing the correlation between the expression of TLR2 and MBP. g MSA patients with high average oligodendrocyte TLR2 levels display decreased MBP expression (R2 = 0.601, p = 0.014). Analyses of three independent single-nucleus transcriptome datasets from Adams et al.15 (h), Martirosyan et al.22 (i), and Wang et al.23 (j) revealed no significant correlation between TLR2 and MBP expression in oligodendrocytes from the substantia nigra of PD patients (R² = 0.168, p = 0.058; R² = 0.061, p = 0.197; R² = 0.021, p = 0.634). Statistical significance in was evaluated using simple linear regression model (g–j). The p-values were calculated by two-sided t-tests (g–j).
Based on our previous findings demonstrating TLR2 engagement in αSyn-induced oligodendrogliopathy (Figs. 2 and 3), we investigated whether oligodendrocyte TLR2 expression is associated with the myelin defects in MSA patients. As shown in Fig. 5g, MSA patients with elevated TLR2 levels exhibited significant downregulation of MBP, indicating a negative correlation between TLR2 and MBP expression (R2 = 0.601, p = 0.014). However, previously reported single-nucleus analyses of PD patients15,22,23 revealed little correlation between TLR2 and MBP expression in oligodendrocytes isolated from the substantia nigra (R² = 0.168, p = 0.058 (h); R² = 0.061, p = 0.197 (i); R² = 0.021, p = 0.634 (j)) (Fig. 5h–j). These findings suggest that the induction of TLR2 expression in oligodendrocytes may contribute to myelination defects in MSA.
Recovery of myelination by NM-101 in αSyn Tg mice
To evaluate the impact of NM-101 on transcriptomic alterations in oligodendrocytes, we conducted a transcriptomic analysis of oligodendrocytes isolated from αSyn Tg mice treated with either NM-101 or a placebo. In oligodendrocytes from NM-101-treated αSyn Tg mice, 162 DEGs were identified (Supplementary Data 3). NM-101 treatment increased the expression of genes associated with oligodendrocyte differentiation and glial cell differentiation (Fig. 6a; Supplementary Tables 8 and 9). Among the DEGs upregulated or downregulated in αSyn Tg mice, 11 genes were reversed following NM-101 administration. These reversed genes were enriched in processes related to central nervous system myelination (Fig. 6b, c and Supplementary Data 3). These results suggest that NM-101 administration reverses defects in myelination and oligodendrocyte differentiation in αSyn Tg mice.
a Bar plot illustrating the significant GO enrichment terms in biological processes based on DEGs from oligodendrocytes in NM-101-treated hA53T Tg mice. b Scatter plot displaying the overlap of DEGs between Tg mice and WT mice, as well as between NM-101-treated Tg mice and untreated Tg mice. c Enriched GO clusters based on DEGs that were reversed by NM-101 treatment. The ClueGO app was used to find enriched GO clusters, and a network of connected GO terms was created. d Representative confocal microscopy images depicting the expression of MBP and PLP in the NSC-derived oligodendrocytes. Scale: 20 μm. e Quantification of the relative MBP in the NSC-derived oligodendrocytes. f Quantification of the relative PLP in the NSC-derived oligodendrocytes. g Representative transmission electron microscopy images depicting myelinated axons in the cerebellum. Scale: 2 μm. h Quantification of the relative G-ratio. i Quantification of the percentage of myelination. j Scatter plot illustrating the correlation between G-ratio and axonal diameter. Representative confocal images of PLP (k) and CNPase (m) in the white matter. Scale: 20 μm. Quantification of the relative PLP (l) and CNPase (n) intensity in the white matter. Statistical significance was evaluated using one-way ANOVA with two-sided Tukey’s post hoc comparison for (e, f, h, i, l) and Brown–Forsythe and Welch ANOVA with two-sided Dunnett’s T3 post hoc test for (n) between groups. All data are presented as the mean ± SEM. n = 3 biologically independent samples for each in (e, f). n = 7 biologically independent samples for each in (h, i, j). n = 8 biologically independent samples for each in (l). n = 9 biologically independent samples for WT, n = 8 biologically independent samples for hA53T Tg mice, n = 7 biologically independent samples for 3 mg/kg in (n).
To experimentally validate the effects of NM-101 on myelination in oligodendrocytes, we assessed the expression levels of myelin-associated proteins in hNSC-derived oligodendrocytes following αSyn treatment. In line with transcriptomic findings, levels of MBP and PLP were reduced upon αSyn exposure. Notably, NM-101 treatment attenuated this reduction, partially restoring the expression of both proteins (Fig. 6d–f). To further validate the impact of NM-101 on myelin integrity, we examined the ultrastructure of myelin in the white matter of αSyn Tg mice treated with NM-101 or placebo using transmission electron microscopy (Fig. 6g). The g-ratio, defined as the ratio of the axonal diameter to the total outer diameter of myelinated fibers, serves as a reliable structural and functional index for evaluating axonal myelination. Higher g-ratio, which reflects proportionally thinner myelin sheaths, is indicative of reduced myelination or demyelination24. In our study, the g-ratio, representing the thickness of the myelin sheath relative to the inner diameter of myelinated fibers, was significantly higher in αSyn Tg mice (0.64) compared to WT mice (0.48), indicating disorganized myelin. Administration of NM-101 reduced g-ratios to 0.54 at 3 mg/kg and 0.52 at 10 mg/kg, suggesting recovery from the demyelination phenotype (Fig. 6h). Scatter plots correlating axon diameter with the g-ratio revealed a significant correlation (Fig. 6j). Additionally, we evaluated the extent of myelination in the same Tg model. αSyn Tg mice exhibited a lower percentage of myelination (74.8%) compared to WT mice (91.3%). NM-101 administration led to the recovery of this phenotype, with myelination percentages reaching 87.1% and 91.2% at 3 and 10 mg/kg, respectively (Fig. 6i). These results demonstrate that NM-101 effectively reversed demyelination, a key pathological feature of MSA, in the white matter regions of αSyn Tg mice (Fig. 6g–j). Furthermore, immunostaining of brain white matter regions revealed that PLP, a marker of myelin sheaths, was reduced in αSyn Tg mice, whereas it was restored to normal levels following NM-101 treatment (Fig. 6k, l). Likewise, CNPase, which was also decreased in αSyn Tg mice, showed a statistically significant recovery by NM-101 administration (Fig. 6m, n).
Discussion
In this study, we propose a potential mechanism underlying GCI formation in MSA, suggesting that neuron-derived αSyn serves as a major source of αSyn aggregates that contribute to oligodendroglial inclusions through neuron-to-oligodendrocyte propagation. Exposure to neuron-derived αSyn induced the formation of GCI-like inclusions in hNSC-derived oligodendrocytes. Consistently, αSyn transgenic mice expressing human αSyn exclusively in neurons exhibited αSyn-positive inclusions within oligodendrocytes, accompanied by shortened lifespan and motor impairments. This neuron-to-oligodendrocyte αSyn transfer was associated with transcriptional dysregulation linked to impaired myelination and oligodendrocyte maturation across hNSC-derived oligodendrocytes, transgenic mice, and MSA patient samples. Notably, treatment with a TLR2-blocking antibody ameliorated both pathological and behavioral abnormalities—including GCI formation, gliosis, neuroinflammation, demyelination, and motor deficits—in transgenic mice. Collectively, these findings support a model in which TLR2-dependent neuron-to-oligodendrocyte αSyn propagation drives GCI formation and highlight TLR2 inhibition as a promising therapeutic strategy for MSA.
The origin of αSyn proteins in GCIs has been debated, since αSyn expression in oligodendrocytes is significantly lower than in neurons. Our RNA sequencing analysis confirmed that αSyn expression in oligodendrocytes is only a fraction of that in neurons in humans (Fig. 1a). αSyn can be secreted from neurons and transferred to other neurons and astrocytes25,26. This led to the hypothesis that αSyn propagates from neurons to oligodendrocytes, forming GCIs in the latter cells. Previous studies have shown the uptake of recombinant αSyn monomers, oligomers, and fibrils by oligodendrocytes27. In this study, we demonstrated the neuron-to-oligodendrocyte transfer and GCI formation in two models. First, human oligodendrocytes derived from neural stem cells formed GCI-like inclusions after exposure to conditioned media from αSyn-expressing neuroblastoma cells. Second, Tg mice that express human αSyn exclusively in neurons (no human αSyn expression in oligodendrocytes) developed human αSyn-positive GCIs in oligodendrocytes. These results unequivocally demonstrate GCI formation via neuron-to-oligodendrocyte transfer. The reverse (oligodendrocyte-to-neuron propagation) may also occur when αSyn was overexpressed in oligodendrocytes in a mouse model28. Although neuron-derived αSyn accumulates in oligodendrocytes in our models, we do not intend to exclude the role of the endogenous oligodendroglial αSyn in GCI formation. It has been shown that the treatment of exogenous αSyn preformed fibrils to the oligodendrocyte precursor cells increases αSyn protein levels through inhibiting autophagic degradation without increasing the gene expression29.
In the study by Uemura et al.20, abundant pSyn-positive neurons were detected in the ipsilateral piriform cortex at 1 and 3 months post-injection (PI) of mouse α-Syn PFFs, but their numbers gradually declined by 7 months PI. Notably, no pSyn-positive glial cells were observed at 1 or 3 months PI, and only limited glial pathology was detected at 7 months PI. Robust pSyn-positive glial pathology emerged at 12 months PI and became more prominent by 18 months PI. Similarly, Masuda-Suzukake et al.30 demonstrated that in mice injected with human α-Syn PFFs into the striatum, pSyn pathology appeared in the corpus callosum at 15 months PI but was absent at 9 months PI. Interestingly, pSyn pathology in the ipsilateral piriform cortex had largely resolved by 15 months PI, suggesting that white matter pathology may arise following the attenuation of grey matter pathology. Collectively, these studies indicate that oligodendroglial pathology in the white matter tends to emerge after neuronal pathology has subsided in α-Syn PFF-injected mice. This temporal pattern aligns with our findings showing oligodendroglial synucleinopathies at 12 months PI. The significance of this delayed onset of oligodendroglial pathology in the context of MSA pathogenesis remains to be elucidated.
There have been multiple Tg mouse models that were designed to generate GCIs through the overexpression of αSyn in oligodendrocytes. In these mice, αSyn expression was driven by oligodendrocyte-specific promoters, such as MBP, CNPase, and PLP31,32,33. Although these mice develop pathological features akin to MSA pathology, including GCI-like inclusions, demyelination, and neurodegeneration, the pathophysiological relevance of oligodendroglial αSyn overexpression has been in question. In this study, we used a Tg model that expresses human A53T αSyn. Although the A53T mutation is not found in MSA patients, we selected this mouse model to achieve exclusive expression of human αSyn in neurons and to take advantage of its well-characterized neuronal synucleinopathy lesions. Notably, several studies have reported that PD patients with mutations at codons 53 and 51 of the SNCA gene (A53T, A53E, or A51D) exhibit MSA-like clinical and pathological features, including the accumulation of GCIs34,35,36. Although the role of these mutations in GCI formation remains unclear, they are not required for GCI pathology, as injection of PFFs of wild-type human αSyn also induces GCI formation in oligodendrocytes. Nonetheless, the A53T Tg mouse model serves as a valuable tool for investigating neuron-to-oligodendrocyte propagation of αSyn. Our present study demonstrates GCI formation in oligodendrocytes without overexpressing αSyn in these cells, akin to the situation in MSA. Analysis of the white matter in neuronal αSyn Tg mice may provide a more relevant approach to study mechanisms of GCI formation and the downstream effects in myelination defects.
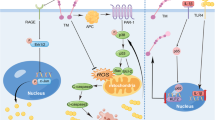
The pathogenic actions of αSyn are not limited to the intraneuronal space, but also extend to the extracellular space, affecting surrounding cells5. The latter is mediated by cell surface receptors for αSyn. Several αSyn receptors have been reported, including TLR28, LAG337, PrPc38, Na-K ATPase39, and FcγR40. Among these, TLR2 is upregulated in PD and DLB patients8,9, and its role in synucleinopathies has been independently validated by several researchers9,41,42 TLR2 is involved in both neuron-to-neuron αSyn propagation and αSyn oligomer-induced microglial activation. In this study, we showed that TLR2 plays a pivotal role in neuron-to-oligodendrocyte propagation of αSyn and pathological changes in oligodendrocyte phenotypes, such as transcriptional alterations and myelin defects. Administration of TLR2-blocking antibody effectively alleviated MSA-related pathologies, such as GCI formation, oligodendroglial gene expression changes, white matter inflammation, demyelination, and behavioral deficits. Therefore, antagonizing TLR2 itself or its downstream signaling may represent a promising approach for MSA therapy.
The mechanism by which TLR2 regulates synucleinopathy is an important topic. We previously demonstrated that TLR2 directly mediates neuron-to-neuron propagation of αSyn10 and promotes synucleinopathy by inhibiting neuronal autophagy17. With respect to neuronal synucleinopathy, we propose that TLR2 contributes to disease progression through both autophagy inhibition and facilitation of cell-to-cell transmission of αSyn. The role of TLR2 in autophagy regulation appears to be both cell type- and stimulus-dependent. In microglia, for example, TLR2 deficiency has been associated with reduced autophagic activity, enhanced M2 polarization, and attenuated neuroinflammation43. Conversely, TLR2 activation has been shown to induce microglial cell death via stimulation of autophagic pathways44. In addition, pathogenic α-synuclein aggregates can impair autophagy via TLR2 activation, leading to protein accumulation in neurons and astrocytes17,45. However, the role of TLR2 in autophagy regulation in oligodendrocytes remains unclear and warrants further investigation. Considering previous studies showing that inhibition of TLR2 can also reduce neuronal inclusions17, it remains uncertain whether the improvements in behavioral outcomes and survival observed upon TLR2 blockade arise predominantly from effects on oligodendrocytes, neurons, or both. Future studies in mice with neuron- or oligodendrocyte-specific deletions of TLR2 might be useful in resolving this question.
Lewy body diseases and MSA share considerable clinical and pathological overlap46,47. Likewise, synuclein pathologies in these diseases are not exclusive. In PD and DLB, although synuclein pathology is dominant in neurons, glial synuclein deposition has also been observed48. In MSA, synuclein deposition is dominant in oligodendrocytes in the form of GCIs, but αSyn pathology is also present in neurons in the form of neuronal cytoplasmic inclusions49. How these related synucleinopathies are differentiated remains mysterious. One interesting hypothesis employs the aggregate “strain” idea to explain this enigma. This hypothesis explains that strains with different structures, thus with different pathogenic properties, determine the disease phenotype. Evidence has been accumulating to support the presence of disease-specific αSyn aggregate strains50,51,52. Although the origins of these strains remain unknown, intracellular milieu, which is specific to cell types, might determine distinct strains of αSyn aggregates53, and ultimately the type of disease. Alternatively, cell type-specific responses to αSyn might differentiate synucleinopathies. For example, in Lewy body diseases, neurons are more vulnerable to αSyn, while in MSA, oligodendrocytes are more vulnerable to αSyn. Our study showed that TLR2 levels are higher in oligodendrocytes of MSA patients than in those of controls and that a negative correlation between TLR2 and MBP expression is observed in oligodendrocytes exclusively in MSA, not in PD. This suggests that oligodendrocytes in MSA are modified to become responsive to αSyn, whereas oligodendrocytes in PD are relatively spared.
In conclusion, GCI formation and the associated MSA pathology can be induced by neuron-to-oligodendrocyte propagation of αSyn. This propagation is mediated by TLR2, whose expression is increased in oligodendrocytes in MSA patients. Anti-TLR2 antibody can block neuron-to-oligodendrocyte propagation of αSyn and alleviate MSA-like pathologies, providing promising therapeutic approaches for MSA.
Methods
Compliance with ethical regulations
Mouse maintenance and experiments were conducted by CHAON Ltd. in compliance with the NIH guideline for the Institutional Animal Care and Use Committee (IACUC) (CE20026). Primary culture were prepared in accordance with the regulations of the Seoul National University Institutional Animal Care and Use Committee (SNU-230511-2). Human postmortem brain tissue was obtained from Seoul National University Hospital, Seoul, Republic of Korea (SNUH Brain Bank) after approval of the Institutional Review Board of SNUH (E-2312-016-1488). All participants provided written informed consent.
Differentiation of hNSCs into oligodendrocytes
The detailed method for hNSC differentiation into oligodendrocytes has been described in Yoon et al.54. Briefly, Stempro hNSCs (Thermo Fisher Scientific, Waltham, MA, USA) were cultured in the OPC medium (CTS KnockOut DMEM/F12 with B-27™ supplement minus vit. A (12587-010, 1:50; Thermo Fisher Scientific), N2 supplement (17502-048, 1:100; Thermo Fisher Scientific), human FGFβ (100–188, final 20 ng/ml; PeproTech, Rocky Hill, NJ, USA), PDGF-AA (CYT-341, 20 ng/mL, ProSpec, Rehovet, Israel), GlutaMAX Supplement (1:100), and penicillin-streptomycin (1:100)) in the Nunclon Sphera dishes to form oligospheres. After 7 days, OPC-containing oligospheres were dissociated with a warm Accutase solution (Thermo Fisher Scientific). Cells were transferred to poly-L-ornithine/laminin-coated dishes. After culturing for 5 days, cells were detached with Accutase again and placed in a regular tissue culture dish for 30 min at 37 °C to allow the removal of sticky non-OPCs. The remaining suspension cells were placed in poly-ornithine/laminin-coated cells with OPC medium. The medium was switched to the oligodendrocyte differentiation (OD) medium and cultured for 14 days. After cells were differentiated for 11 days, 10 mg/ml of NM-101 or hIgG (HG4K, Sino Biological, Beijing, China) was pre-treated for 30 min. Cells were then treated with the conditioned medium containing α-synuclein (αSCM) or β-galactosidase (LZCM) (500 ng/ml) for 72 h54.
Culture of primary mouse oligodendrocytes
The detailed protocol for the isolation and maturation of primary oligodendrocytes was previously described by Kim et al.55 were isolated from the cerebral cortices of C57BL/6 mice using a two-step differential centrifugation method, as previously reported55. The isolated OPCs were seeded into poly-D-Lysine-coated T75 flasks (P7280, Sigma-Aldrich, St. Louis, MO, USA) and expanded for 5 days in OPC proliferation medium. This medium consisted of Dulbecco’s Modified Eagle Medium/Ham’s F12 (#11320033, Thermo Fisher Scientific) supplemented with 1% GlutaMAX (#35050-061, Thermo Fisher Scientific), 1% penicillin/streptomycin, 2% B-27 supplement (12587-010, Thermo Fisher Scientific), 30 ng/mL platelet-derived growth factor-AA (CYT-341, ProSpec), 10 ng/mL fibroblast growth factor (100–188, PeproTech), and 10 ng/mL epidermal growth factor (AF-100-15, PeproTech). After 5 days, confluent OPC colonies were observed. Cells were detached using 5 mL/flask of Accutase, followed by centrifugation at 200 × g. The resulting cell pellet was resuspended in 1 mL of OPC stabilization medium (identical to OPC proliferation medium but without EGF) and seeded onto poly-D-Lysine-coated coverslips at a density of 1 × 104 live cells/cm2. Cells were maintained in stabilization medium for 2 days prior to differentiation. Differentiation was initiated by replacing the stabilization medium with oligodendrocyte (OL) differentiation medium, composed of Neurobasal medium (#21103049, Thermo Fisher Scientific), supplemented with 1% GlutaMAX, 1% penicillin/streptomycin, 2% B-27, 1% N2 supplement, and 40 ng/mL triiodothyronine. Cells were differentiated for 4 days under these conditions. After differentiation to mature oligodendrocytes, 10 mg/ml of NM-101 or hIgG (HG4K, Sino Biological) was pre-treated for 30 min. Cells were then treated with the conditioned medium containing α-synuclein (αSCM) or β-galactosidase (LZCM) (500 ng/ml) for 72 h.
Immunofluorescence cell staining
Cells were then fixed and stained as described in previously54. Briefly, the cells were fixed in 4% paraformaldehyde in PBS and then permeabilized with 0.1% Triton X-100. After washing with PBS, coverslips were placed in the blocking solution (5% bovine serum albumin and 3% goat serum in PBS). The primary antibodies were diluted in a blocking solution and added to the coverslips. The following primary antibodies were used: anti-α-synuclein (#610787, 1:100; BD Biosciences, San Diego, CA, USA), anti-phospho-α-synuclein (ab59264, 1:1,000; Abcam, Waltham, MA, USA), anti-MBP (ab7349, 1:500; Abcam), anti-TPPP (EPR3316, ab92305, 1:200; Abcam), and anti-ubiquitin (ab7780, 1:50; Abcam). After washing in PBS, the coverslips were covered with the fluorescent dye-conjugated secondary antibodies (Jackson Immunoresearch Laboratories, PA, USA) for 30 min and then washed again in PBS. The nuclei were stained with TOPRO-3 dye (Thermo Fisher) or Hoechst 33342 (Sigma-Aldrich). Coverslips were mounted on slides using the Antifade reagent (Thermo Fisher).
Animals and NM-101 administration
αSyn A53T Tg mice (line G2-3), which express human αSyn A53T under the PrP promoter were purchased from Jackson Laboratory (stock #006823; Bar Harbor, ME, USA). Male mice (6-month-old) were administered with NM-101 or placebo for 13 weeks (weekly, intravenous injection). The placebo is the buffer solution that has been used in clinical trials with the composition of 25 mmol/l sodium citrate at pH 5.5, 125 mmol/l sodium chloride, and 6 mmol/l citric acid in 0.01% wt/vol polysorbate 20. Behavioral tests were performed to assess motor functions.
Brain tissue preparation for immunoblot analysis
After anesthesia using 2% isoflurane in oxygen (700 ml/min), blood was collected through the abdominal vein. After blood collection, mice were perfused with 1X PBS for 10 min (flow rate: 5 ml/min). After excision, the brains were dissected to sub-region and stored in a deep freezer until use. To separate the Triton X-100 soluble and insoluble fractions, white matter (anterior commissure and corpus callosum) region samples were collected and homogenized with 1X PBS containing 1% Triton X-100, protease inhibitors cocktail, and phosphatase inhibitor using a dounce homogenizer (#357538; DWK Life Sciences, Mainz, Germany). The homogenates were incubated for 20 min in ice and then centrifuged at 15,000 × g for 20 min at 4 °C. The resulting supernatants (soluble fractions) were collected. The pellet was solubilized with 1X RIPA buffer containing 1% protease inhibitor cocktail, and phosphatase inhibitor for 20 min in ice and further centrifuged at 15,000 × g for 20 min at 4 °C. The resulting supernatants (Triton X-100 insoluble fractions) were collected and stored in a deep freezer until use. Protein concentrations were determined using the BCA assay according to the manufacturer’s instruction (#23225; Thermo Fisher Scientific).
Brain tissue preparation for immunofluorescence
At the conclusion of the study, a behavioral test was conducted, followed by a necropsy. Following blood collection, PBS perfusion was performed using a peristaltic pump. The right hemisphere was then stored in 4% PFA, while the left hemisphere was stored in an ultra-low temperature freezer (−80 °C). To prepare the tissue for further analysis, the 4% PFA solution was replaced with 30% sucrose and the samples were incubated at 4 °C overnight or until they had sunk. A brain mold was created using OCT compound, and the mold was stored at −80 °C. The brain was subsequently coronally sectioned (30 μm-thick) using a Leica cryostat, and the sections were stored in a stock solution (composed of 30% glycerol, 30% ethylene glycol, 10% 0.2 M phosphate buffer, and 30% distilled water (v/v)) at 4 °C.
Immunoblot analysis
Brain homogenates were boiled with Laemmli sample buffer (#161-0747; Bio-Rad, Hercules, CA, USA) at 95 °C for 5 min. Ten micrograms of protein samples were loaded in 10% SDS-PAGE gel, and electrophoresis was conducted at 100 volts for 90 min. Proteins were transferred from SDS-PAGE gel to nitrocellulose membrane at 300 milliamperes for 90 min. The membrane was incubated with blocking buffer (5% skim milk in TBS containing 0.05% Tween 20) at room temperature for 30 min and then with primary antibody diluted with buffer (5% BSA in TBS containing 0.05% Tween 20) at 4 °C for 15 h. After washing three times with TBS containing 0.05% Tween 20, the membrane was incubated with the secondary antibody at room temperature for 1 h. For data analysis, western blot intensities of the proteins were quantified by ImageJ software and then normalized with those of β-actin. The following primary antibodies were used: anti-α-synuclein (#610787, 1:1,500; BD Biosciences), anti-phospho-α-synuclein (ab51253, 1:1,000; Abcam), and anti-β-actin (AC-15, 1:10,000; Sigma-Aldrich).
Fluorescence imaging and quantification
Fixed mouse brain sections were blocked in a solution containing 5% normal goat serum (Jackson ImmunoResearch) and 0.1% Triton X-100 (Sigma-Aldrich) for 1 h at room temperature. The samples were then incubated with primary antibodies against TPPP, pS129-αSyn, Olig2, GFAP or Iba-1 at 4 °C overnight. Briefly, floating brain sections were washed with PBS containing 0.1% Triton X-100 and incubated with fluorescence-conjugated secondary antibodies (Invitrogen, Waltham, MA, USA) at room temperature for 1 h. The sections were mounted with or without DAPI. For staining of GSTP1, CNPase, PLP, and MBP, sections were treated with an antigen-retrieval solution (10 mM sodium citrate in 0.05% Tween 20, pH 6.0) at 98 °C for 5 min prior to blocking in PBS containing 0.1% Triton X-100, 5% BSA, and 5% normal donkey serum (Jackson ImmunoResearch). The samples were then incubated with primary antibodies against GSTP1, CNPase, PLP, or MBP at 4 °C overnight. Briefly, floating brain sections were subsequently washed with PBS containing 0.1% Triton X-100 and incubated with fluorescence-conjugated secondary antibodies at room temperature for 90 min. The sections were mounted following DAPI (D1306, 1:1,000; Invitrogen) staining. For FLICA staining, freshly frozen sections of mouse brain were examined to determine the level of Inflammasome (caspase-1 activity). This was done by employing the FAM-FLICA Caspase-1 (YVAD) Assay Kit (SKU-98; ImmunoChemistry Technologies, Bloomington, MN, USA). The FLICA reagent is a dye that can penetrate cells and attach to activated Caspase-1. Once FLICA staining was completed, we performed immunofluorescence staining in additional imaging channels, as outlined above. The following primary antibodies were used: anti-TPPP (ab92305, 1:250; Abcam), anti-phospho-α-synuclein (#825702, 1:1,000; Biolegend, San Diego, CA, USA), anti-olig2 (AB9610, 1:500; EMD Millipore, Billerica, MA, USA), anti-GFAP (ab7260, 1:500; Abcam), anti-Iba-1 (ab5076, 1:500; Abcam), anti-Cd68 (MCA1957, 1:100; Bio-Rad), anti-GSTP1 (LS-B2376-50, 1:500; LifeSpan Biosciences, Seattle, WA), anti-CNPase (PA1-4712, 1:500; Thermo Fisher Scientific), anti-PLP (ab254363, 1:1000; Abcam), anti-MBP (ab209328, 1:250; Abcam), and anti-IBA1 (019-19741, 1:500; FUJIFILM Wako, Osaka, Japan). Fluorescent images were obtained using a confocal microscope (Zeiss LSM 980) or Microscope Slide Scanner (Axio Scan Z1). Fluorescence intensity was analyzed with Fiji software (ImageJ, NIH, version 1.54.p) unless otherwise specified.
Sholl analysis of microglial morphology
Sholl analysis was performed to quantify microglial branching complexity. Only well-isolated microglia without overlapping processes from neighboring cells were selected for analysis to avoid confounding branch intersections. Image processing and analysis were performed using Fiji software. The Iba1 channel was separated from other channels, and background fluorescence was subtracted. For each selected cell, the center of the soma was manually defined as the active region of interest (ROI). Sholl analysis was conducted using the simple neurite tracer (SNT) plugin. Concentric radii were drawn starting at 6 µm from the soma, with a step size of 3 µm, and ending at 50 µm. The number of intersections at each radius was calculated, and primary branches were inferred from the starting radius. For curve fitting, the Sholl profile was approximated using the best-fitting polynomial degree determined by the plugin. Sholl decay was automatically chosen by the software between semi-logarithmic and log-log transformations using the default normalization settings. From the resulting Sholl profiles, parameters including total intersections and maximum intersections were extracted for quantitative comparison between experimental conditions. Statistical analysis was performed using one-way ANOVA followed by Tukey’s multiple comparison test, with p < 0.05 considered statistically significant.
scRNA-seq data analysis
R version 4.2.2 was used for statistical analysis and plotting. The filtered gene-cell barcode matrices, which were generated with CellRanger 3.0.2 (10x Genomics) based on the human GRCh38 reference genome were used for further analysis with the R package Seurat v4.3.0. For quality control, nuclei with less than 200 genes, more than 5% mitochondrial content or an extremely high number of detected genes or UMIs were filtered out. After the quality control process, we obtained 12,547 single cell expression profiles. The Function SelectIntegrationFeatures (nfeatures = 2000) was used to identify variable genes. Clusters were identified with the functions FindNeighbors (dims = 1:10) and FindClusters (resolution = 0.15). To identify marker genes, the function FindAllMarkers (min.pct = 0.25, log2fc.threshold = 0.3) was used. P < 0.05 was used as a cutoff to determine the marker genes. Cell types were annotated based on the Panglao database. After data integration and the unsupervised clustering process, marker genes were identified using the function FindAllMarkers (min.pct = 0.25, log2fc.threshold = 0.3). To determine differentially expressed genes (DEGs), a gene with a Benjamini–Hochberg (FDR) adjusted P < 0.05 and log2 of fold change > 0.3 or < −0.3 was determined to be statistically significant.
Tissue preparation for laser capture microdissection
Blocks of tissue containing the corpus callosum (CC) were frozen in OCT medium on dry ice. Tissue blocks were sectioned (5 μm) with a cryostat microtome, mounted on membrane slide PET 1.0, and stored at −80 °C. Staining procedures were conducted at 4 °C. Frozen tissue sections were fixed with methanol/acetone solution (1:1) for 10 min. After blocking with 10% normal horse serum in TBS (0.05 M Tris, 0.25 M NaCl, 0.25% Triton X-100, pH 7.6), tissues were stained with primary antibodies against phospho-α-synuclein (pS129) (ab51253, 1:500; Abcam) and GSTP1 (LS-B2376-50, 1:100; LifeSpan Biosciences). Sections were incubated with fluorescence dye-conjugated secondary antibodies, followed by dehydration using an ethanol series (70%, 95%, 100%). After dipping in xylene for 1 min, sections were dried and incubated at −80 °C.
Laser capture microdissection (LCM)
Cells in the CC were microdissected by “cut” and “laser pressure catapulting (LPC)” using a PALM Zeiss UV laser capture microdissection system. Approximately 300 selected cells were ejected against gravity into the cap of the adhesiveCap opaque tube, and dissolved with 20 µL RNA extraction lysis buffer. Libraries were prepared using a QuantSeq 3′ mRNA-Seq Library Prep Kit (Lexogen, Inc., Greenland, NH, USA). cDNA was sequenced with 75-bp single-end reads on a NextSeq 500 sequencing system (Illumina, Inc., San Diego, CA, USA).
Differentially expressed gene analysis
Reads were aligned using the STAR software v2.7.10b. For the LCM data set, reads were aligned against the mouse reference genome (GRCm39). The datasets of MSA patients and healthy controls were obtained from Piras et al.18. The reads were aligned to the human reference genome (GRCh38). Data were summarized as gene counts using featureCounts v1.16.1. Gene expression differential analyses were conducted using the R package DESeq2 v3.18. Enrichment analyses of Gene Ontology terms were analyzed using the Cytoscape plug-in ClueGO and EnrichR based on related terms and statistical significance56,57. For the correlation analysis between TLR2 and MBP, we excluded three outliers using principal component analysis after processing the data with the R package DESeq2 v3.18. For the remaining nine samples (4 MSA patients and 5 unaffected individuals), the average expression levels of TLR2 and MBP across all oligodendrocytes from each individual were normalized to the healthy control average. A linear regression model was then fitted to the data for all individuals.
snRNA-seq data analysis for SNCA levels in normal neurons and oligodendrocytes
R version 4.3.3 was used for statistical analysis and plotting. The filtered gene-nuclei barcode matrices of the healthy control human postmortem midbrain dataset (young control, aged control) were obtained from Adams et al.15. After excluding two samples with poor RNA read quality as indicated by Adams et al.15 the remaining 17 samples were used for further analysis with the R package Seurat v5.0.1. For quality control, nuclei with less than 100 or more than 20,000 detected genes, less than 500 or more than 100,000 reads, and more than 5% mitochondrial content were filtered out to yield 43,757 single nuclei expression profiles in total. Function FindVariableFeatures (nfeatures = 2000) was used to identify variable genes, which were used as anchors in integrating the 17 samples with 20 dimensions of canonical correlation analysis (CCA), as per procedures reported by Adams et al.15. Neurons and oligodendrocytes were respectively classified as STMN2+ and MBP+MOG+ cells (expression > 0.6). Expression of SNCA as well as neuron and oligodendrocyte markers were analyzed.
Pseudobulk analyses of snRNA-seq cohort data for oligodendrocyte TLR2 and MBP expression in PD patients
R version 4.3.3 was used for statistical analysis and plotting. The filtered gene-nuclei barcode matrices of postmortem substantia nigra from PD patients and healthy controls were obtained from Adams et al.15, Martirosyan et al.22, and Wang et al.23. After excluding samples with poor RNA read quality, the remaining 22, 29, and 13 samples (14 PD patients, 8 unaffected individuals; 15 PD patients, 14 unaffected individuals; 9 PD patients, 4 unaffected individuals) were used for further analysis with the R package Seurat v5.0.1.
For quality control of data from Adams et al.15, nuclei with less than 100 or more than 20,000 detected genes, less than 500 or more than 100,000 reads, and more than 5% mitochondrial content were filtered out. For quality control of data from Martirosyan et al.22 nuclei with less than 100 or more than 7000 detected genes, less than 500 or more than 6000 reads, and more than 5% mitochondrial content were filtered out. For quality control of data from Wang et al.23, nuclei with less than 200 or more than 2500 detected genes, less than 500 or more than 6000 reads, and more than 5% mitochondrial content were filtered out.
In each dataset, oligodendrocytes were classified as MBP+MOG+ cells (expression > 0.6). This selection yielded a total of 12,065, 21,284, and 20,786 oligodendrocyte nuclei, respectively. Average expression levels of TLR2 and MBP across all oligodendrocyte nuclei from each individual were normalized with the healthy control average in their cohort. Linear regression models were fitted on all individuals in each cohort.
Isolation of neurons and oligodendrocytes from MSA patients and controls
Frozen brain autopsy tissues of patients diagnosed with multiple system atrophy and primary age-related tauopathy (PART; control) were obtained from the SNUH Brain Bank (Seoul National University Hospital, Seoul, Republic of Korea). The available demographic. and clinical information of PART and MSA patients is described in Supplementary Table 10. Brain tissues were dissociated using the Adult Brain Dissociation Kit (#130-107-677; Miltenyi Biotec, Waltham, MA, USA) following the manufacturer’s protocol with some modification. Briefly, brain tissues were chopped in provided enzyme P before incubation with provided enzyme A at 37 °C for 20 min with occasional tapping and trituration. DPBS containing 0.5% fatty acid-free BSA and 5 mM EDTA was added to stop the reaction. The samples were filtered through a Falcon® 70 μm Cell Strainer (Corning, #352350), and the flow-through was centrifuged at 300 × g for 10 min at 4 °C. The pellets were resuspended in 60 µl of MACS buffer (PBS supplemented with 0.5% BSA and 2 mM EDTA) per 106 cells.
To label oligodendrocytes with O4+, cells were incubated with FcR Blocking Reagent (#130-059-901, 20 μL/107 cells; Miltenyi Biotec) on ice for 10 min, followed by incubation with anti-O4 MicroBeads (#130-096-670, 10 μL/107 cells; Miltenyi Biotec) on ice for 15 min. Cells were washed by adding DPBS containing 0.5% fatty acid-free BSA and 5 mM EDTA and centrifuging at 300 × g for 10 min at 4 °C. After resuspension in DPBS containing 0.5% fatty acid-free BSA and 5 mM EDTA, cells were applied through a MACS LS column (#130-042-401; Miltenyi Biotec). The resulting flow-through (O4− cells) was collected. The column was washed 3 times before flushing out the final eluate (O4+ cells). To enrich neurons, astrocytes and microglia were depleted from O4− cells using ACSA-1 MicroBeads (#130-095-826; Miltenyi Biotec) and Cd11b MicroBeads (#130-093-634; Miltenyi Biotec), respectively. The resulting oligodendrocytes and neurons were centrifuged at 300 × g for 10 min at 4 °C.
Expression analysis for SNCA levels in neurons and oligodendrocytes
Total RNA was isolated from neurons and oligodendrocytes using the RNeasy Mini Kit (#74104; QIAGEN, Hilden, Germany), and cDNA libraries were prepared using the iScriptTM cDNA Synthesis Kit (#1708890; Bio-Rad). Real-time PCR (qPCR) was performed with iTaqTM Universal SYBR Green Supermix (#1725121; Bio-Rad) for SNCA and GAPDH. Primers were designed with Primer bank and the NCBI Primer Blast software. The following primers were used: SNCA: 5′-ACCAAACAGGGTGTGGCAGAAG-3′ (forward), 5′-CTTGCTCTTTGGTCTTCTCAGCC-3′ (reverse); GAPDH: 5’-GTCTCCTCTGACTTCAACAGCG-3’ (forward), 5′-ACCACCCTGTTGCTGTAGCCAA-3′ (reverse).
Expression analysis for TLR2 levels in oligodendrocytes from MSA patients and controls
Total RNA was isolated from O4+ cells using the RNeasy Mini Kit (#74104; QIAGEN, Hilden, Germany), and cDNA libraries were prepared using the iScriptTM cDNA Synthesis Kit (#1708890; Bio-Rad). Real-time PCR (qPCR) was performed with iTaqTM Universal SYBR Green Supermix (#1725121; Bio-Rad) for TLR2, GAPDH, and OMG. Primers were designed with Primer Bank and the NCBI Primer Blast software. The following primers were used: TLR2: 5′-CTTCACTCAGGAGCAGCAAGCA-3′ (forward), 5′-ACACCAGTGCTGTCCTGTGACA-3′ (reverse); GAPDH: 5′-GTCTCCTCTGACTTCAACAGCG-3′ (forward), 5′-ACCACCCTGTTGCTGTAGCCAA-3′ (reverse); OMG: 5′- GGATGCAGACAGTGGACACCAT-3′ (forward), 5′-GGTGGTGTCTTTGCTTAGAGTGG-3′ (reverse).
Transmission electron microscope imaging and analysis
The brain tissue sections were washed with PBS and treated with a 1% solution of osmium (OsO4) for fixation. Following that, the tissues underwent dehydration using a series of ethanol and 100% propylene oxide and were then embedded in Epon plastic resin. Cross sections of the embedded samples were obtained at a thickness of 0.5 μm. To create ultrathin sections measuring 70–90 nm, the samples were placed on copper grids coated with formvar and subsequently stained with uranyl acetate and lead citrate. The samples were then examined using a FEI (Tecnai G2) transmission electron microscope (TEM). For analysis, EM images were composed of multiple pictures per group, and the MyelTracer program was used to analyze ten to twelve myelin structures per picture. Parameters such as axon size, internal and external myelin sizes, and the presence of unmyelinated or myelinated exons were manually selected. The program then automatically calculated values, such as the g-ratio, axonal diameter, and myelination percentage. The resulting values were exported to an Excel file and plotted on graphs.
Statistical analysis
Graphs were generated using the GraphPad Prism 9 software (GraphPad Software Inc., La Jolla, CA, USA), and statistical analysis was performed by one-way ANOVA with the Tukey’s post-hoc test unless otherwise described. Differences were considered significant at p < 0.05.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data generated or analyzed during this study are included in this published article, the Supplementary information, and Source data file. The gene expression data generated in this manuscript have been uploaded to the NCBI Sequence Read Archive (SRA) under BioProject ID PRJNA1180266 - SRA - NCBI, accession number GSE312641 GEO Accession viewer, GSE312642 GEO Accession viewer). The metadata for Adams et al. (accession number GSE193688 GEO Accession viewer) and Piras et al. (accession number GSE199724 GEO Accession viewer, GSE199715 GEO Accession viewer, and GSE199258 GEO Accession viewer), Martirosyan et al. (accession number GSE243639 GEO Accession viewer), and Wang et al. (accession number GSE184950 GEO Accession viewer) were downloaded from Gene Expression Omnibus (GEO). All materials are available from the corresponding author upon request. Source data are provided with this paper.
References
McCann, H., Stevens, C. H., Cartwright, H. & Halliday, G. M. Alpha-synucleinopathy phenotypes. Park. Relat. Disord. 20, S62–S67 (2014).
Krismer, F. & Wenning, G. K. Multiple system atrophy: insights into a rare and debilitating movement disorder. Nat. Rev. Neurol. 13, 232–243 (2017).
Calabresi, P. et al. Alpha-synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction. Cell Death Dis. 14, 176 (2023).
Muller, C. M. et al. Staging of sporadic Parkinson disease-related alpha-synuclein pathology: inter- and intra-rater reliability. J. Neuropathol. Exp. Neurol. 64, 623–628 (2005).
Lee, H. J., Bae, E. J. & Lee, S. J. Extracellular alpha-synuclein—a novel and crucial factor in Lewy body diseases. Nat. Rev. Neurol. 10, 92–98 (2014).
Abounit, S. et al. Tunneling nanotubes spread fibrillar alpha-synuclein by intercellular trafficking of lysosomes. EMBO J. 35, 2120–2138 (2016).
Ruf, W. P., Meirelles, J. L. & Danzer, K. M. Spreading of alpha-synuclein between different cell types. Behav. Brain Res. 436, 114059 (2023).
Kim, C. et al. Neuron-released oligomeric alpha-synuclein is an endogenous agonist of TLR2 for paracrine activation of microglia. Nat. Commun. 4, 1562 (2013).
Dzamko, N. et al. Toll-like receptor 2 is increased in neurons in Parkinson’s disease brain and may contribute to alpha-synuclein pathology. Acta Neuropathol. 133, 303–319 (2017).
Kim, C. et al. Immunotherapy targeting toll-like receptor 2 alleviates neurodegeneration in models of synucleinopathy by modulating alpha-synuclein transmission and neuroinflammation. Mol. Neurodegener. 13, 43 (2018).
Kon, T. et al. Neuronal SNCA transcription during Lewy body formation. Acta Neuropathol. Commun. 11, 185 (2023).
Han, X. et al. Construction of a human cell landscape at single-cell level. Nature 581, 303–309 (2020).
Djelloul, M. et al. Alpha-synuclein expression in the oligodendrocyte lineage: an in vitro and in vivo study using rodent and human models. Stem Cell Rep. 5, 174–184 (2015).
Kon, T. et al. SNCA and TPPP transcripts increase in oligodendroglial cytoplasmic inclusions in multiple system atrophy. Neurobiol. Dis. 198, 106551 (2024).
Adams, L., Song, M. K., Yuen, S., Tanaka, Y. & Kim, Y. S. A single-nuclei paired multiomic analysis of the human midbrain reveals age- and Parkinson’s disease-associated glial changes. Nat. Aging 4, 364–378 (2024).
Ishimoto, T. et al. A novel mouse model for investigating alpha-synuclein aggregates in oligodendrocytes: implications for the glial cytoplasmic inclusions in multiple system atrophy. Mol. Brain 17, 28 (2024).
Kim, C. et al. Antagonizing neuronal toll-like receptor 2 prevents synucleinopathy by activating autophagy. Cell Rep. 13, 771–782 (2015).
Piras, I. S. et al. Transcriptional profiling of multiple system atrophy cerebellar tissue highlights differences between the Parkinsonian and cerebellar sub-types of the disease. Acta Neuropathol. Commun. 8, 76 (2020).
Refolo, V. et al. Progressive striatonigral degeneration in a transgenic mouse model of multiple system atrophy: translational implications for interventional therapies. Acta Neuropathol. Commun. 6, 2 (2018).
Uemura, N. et al. Inoculation of alpha-synuclein preformed fibrils into the mouse gastrointestinal tract induces Lewy body-like aggregates in the brainstem via the vagus nerve. Mol. Neurodegener. 13, 21 (2018).
Franzen, O., Gan, L. M. & Bjorkegren, J. L. M. PanglaoDB: a web server for exploration of mouse and human single-cell RNA sequencing data. Database 2019, baz046 (2019).
Martirosyan, A. et al. Unravelling cell type-specific responses to Parkinson’s disease at single cell resolution. Mol. Neurodegener. 19, 7 (2024).
Wang, Q. et al. Molecular profiling of human substantia nigra identifies diverse neuron types associated with vulnerability in Parkinson’s disease. Sci. Adv. 10, eadi8287 (2024).
Fields, R. D. & Dutta, D. J. G-ratio commentary-why you’ve been doing it wrong. ASN Neuro 17, 2486962 (2025).
Desplats, P. et al. Inclusion formation and neuronal cell death through neuron-to-neuron transmission of alpha-synuclein. Proc. Natl. Acad. Sci. USA 106, 13010–13015 (2009).
Lee, H. J. et al. Direct transfer of alpha-synuclein from neuron to astroglia causes inflammatory responses in synucleinopathies. J. Biol. Chem. 285, 9262–9272 (2010).
Reyes, J. F. et al. Alpha-synuclein transfers from neurons to oligodendrocytes. Glia 62, 387–398 (2014).
Yamaguchi, H. et al. A rapidly progressive multiple system atrophy-cerebellar variant model presenting marked glial reactions with inflammation and spreading of alpha-synuclein oligomers and phosphorylated alpha-synuclein aggregates. Brain Behav. Immun. 121, 122–141 (2024).
Kaji, S. et al. Pathological endogenous alpha-synuclein accumulation in oligodendrocyte precursor cells potentially induces inclusions in multiple system atrophy. Stem Cell Rep. 10, 356–365 (2018).
Masuda-Suzukake, M. et al. Prion-like spreading of pathological alpha-synuclein in brain. Brain 136, 1128–1138 (2013).
Yazawa, I. et al. Mouse model of multiple system atrophy alpha-synuclein expression in oligodendrocytes causes glial and neuronal degeneration. Neuron 45, 847–859 (2005).
Stefanova, N. A mouse model of multiple system atrophy: bench to bedside. Neurotherapeutics 20, 117–126 (2023).
Shults, C. W. et al. Neurological and neurodegenerative alterations in a transgenic mouse model expressing human alpha-synuclein under oligodendrocyte promoter: implications for multiple system atrophy. J. Neurosci. 25, 10689–10699 (2005).
Markopoulou, K. et al. Clinical, neuropathological and genotypic variability in SNCA A53T familial Parkinson’s disease. Variability in familial Parkinson’s disease. Acta Neuropathol. 116, 25–35 (2008).
Pasanen, P. et al. Novel alpha-synuclein mutation A53E associated with atypical multiple system atrophy and Parkinson’s disease-type pathology. Neurobiol. Aging 35, 2180 e2181–2180 (2014).
Whittaker, H. T., Qui, Y., Bettencourt, C. & Houlden, H. Multiple system atrophy: genetic risks and alpha-synuclein mutations. F1000Research 6, 2072 (2017).
Mao, X. et al. Pathological alpha-synuclein transmission initiated by binding lymphocyte-activation gene 3. Science 353, aah3374 (2016).
Urrea, L., Ferrer, I., Gavin, R. & Del Rio, J. A. The cellular prion protein (PrPC) as neuronal receptor for alpha-synuclein. Prion 11, 226–233 (2017).
Shrivastava, A. N. et al. Alpha-synuclein assemblies sequester neuronal alpha3-Na+/K+-ATPase and impair Na+ gradient. EMBO J. 34, 2408–2423 (2015).
Choi, Y. R. et al. FcgammaRIIB mediates the inhibitory effect of aggregated alpha-synuclein on microglial phagocytosis. Neurobiol. Dis. 83, 90–99 (2015).
Scheiblich, H. et al. Microglial NLRP3 inflammasome activation upon TLR2 and TLR5 ligation by distinct alpha-synuclein assemblies. J. Immunol. 207, 2143–2154 (2021).
Dutta, D. et al. Selective targeting of the TLR2/MyD88/NF-kappaB pathway reduces alpha-synuclein spreading in vitro and in vivo. Nat. Commun. 12, 5382 (2021).
Ma, K., Guo, J., Wang, G., Ni, Q. & Liu, X. Toll-like receptor 2-mediated autophagy promotes microglial cell death by modulating the microglial M1/M2 phenotype. Inflammation 43, 701–711 (2020).
Arroyo, D. S. et al. Toll-like receptor 2 ligands promote microglial cell death by inducing autophagy. FASEB J. 27, 299–312 (2013).
Weiss, F. et al. Astrocytes contribute to toll-like receptor 2-mediated neurodegeneration and alpha-synuclein pathology in a human midbrain Parkinson’s model. Transl. Neurodegener. 13, 62 (2024).
Miki, Y. et al. Improving diagnostic accuracy of multiple system atrophy: a clinicopathological study. Brain 142, 2813–2827 (2019).
Miki, Y. et al. Identification of multiple system atrophy mimicking Parkinson’s disease or progressive supranuclear palsy. Brain 144, 1138–1151 (2021).
Wakabayashi, K., Hayashi, S., Yoshimoto, M., Kudo, H. & Takahashi, H. NACP/alpha-synuclein-positive filamentous inclusions in astrocytes and oligodendrocytes of Parkinson’s disease brains. Acta Neuropathol. 99, 14–20 (2000).
Yokoyama, T., Kusunoki, J. I., Hasegawa, K., Sakai, H. & Yagishita, S. Distribution and dynamic process of neuronal cytoplasmic inclusion (NCI) in MSA: correlation of the density of NCI and the degree of involvement of the pontine nuclei. Neuropathology 21, 145–154 (2001).
Ayers, J. I. et al. Different alpha-synuclein prion strains cause dementia with Lewy bodies and multiple system atrophy. Proc. Natl. Acad. Sci. USA 119, e2113489119 (2022).
Shahnawaz, M. et al. Discriminating alpha-synuclein strains in Parkinson’s disease and multiple system atrophy. Nature 578, 273–277 (2020).
Strohaker, T. et al. Structural heterogeneity of alpha-synuclein fibrils amplified from patient brain extracts. Nat. Commun. 10, 5535 (2019).
Peng, C. et al. Cellular milieu imparts distinct pathological alpha-synuclein strains in alpha-synucleinopathies. Nature 557, 558–563 (2018).
Yoon, Y. S. et al. Alpha-synuclein inclusion formation in human oligodendrocytes. Biomol. Ther. 29, 83–89 (2021).
Kim, H. et al. Protocol for the acquisition and maturation of oligodendrocytes from neonatal rodent brains. STAR Protoc. 5, 103327 (2024).
Bindea, G. et al. ClueGO: a cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics 25, 1091–1093 (2009).
Chen, E. Y. et al. Enrichr: interactive and collaborative HTML5 gene list enrichment analysis tool. BMC Bioinform. 14, 128 (2013).
Acknowledgements
We acknowledge the SNUH Brain Bank (Seoul National University Hospital, Seoul, Republic of Korea) and the SNUH Institution of Corpse-derived substance supply for assistance in providing post-mortem tissue samples and clinical data. We thank Seong-Ik Kim, Yu-Mi Shim, and Yiwook Choi in the SNUH Brain Bank for post-mortem brain sample preparation and data collection. We thank Jun Young Choi for his comments on the establishment of primary mouse oligodendrocyte culture. We also thank Hongsun Park for handling the documentation necessary for obtaining approval from the Institutional Review Board at SNUH. This research was supported by the National Research Foundation of Korea (RS-2024-00336045 to H-J.L., and RS-2023-00243474, RS-2025-16072952 to E-J.B.), the Korea Dementia Research Project through the Korea Dementia Research Center (KDRC), funded by the Ministry of Health and Welfare and Ministry of Science and ICT, Republic of Korea (RS-2024-00445582 to S-J.L.), the Korea Health Industry Development Institute (RS-2023-00266536 to S-J.L.), the BK21 FOUR funded by the Ministry of Education (MOE) and the National Research Foundation of Korea (to E-J.B.), and the 2024 Health Fellowship Foundation (to Y.W.J.).
Author information
Authors and Affiliations
Contributions
S-J.L., S.H., and E-J.B. conceptualized the study. E-J.B. and Y.W.J. performed transcriptomic analysis. W.J.A., Y-S.Y., and H-J.L. performed hNSC-derived oligodendrocyte cell experiments. E-J.B. performed primary mouse oligodendrocyte cell experiments. S.H., W.S.Y., Y.W.J., E-J.B., and Y.W.J. performed animal experiments and analyses. J.S. and W-J.L. performed in vitro experiments and analyses. S-H.P. provided the postmortem brain samples, clinical data, and technical support in human sample experiments. E-J.B., S.H., Y.W.J., W.S.Y., J.S., W-J.L., W.J.A., Y-S.Y., S.H.L., and H-J.L. performed quantification and statistical analyses. E-J.B., S.H., Y.W.J., S.H.L., H-J.L., and S-J.L. wrote the manuscript with input from all authors. Funding for this study was acquired by S-J.L., H-J.L., E-J.B., and Y.W.J. All the authors read and approved the manuscript.
Corresponding author
Ethics declarations
Competing interests
S.J.L. is a co-founder and CEO of Neuramedy Co., Ltd. The other authors declare that they have no competing interests.
Peer review
Peer review information
Nature Communications thanks Ryosuke Takahashi and the other anonymous reviewers for their contribution to the peer review of this work. A peer review file is available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Source data
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Bae, EJ., Ham, S., Jeong, Y.W. et al. Anti-TLR2 immunotherapy modulates neuron-to-oligodendrocyte propagation of α-synuclein in mouse and human models. Nat Commun 17, 2175 (2026). https://doi.org/10.1038/s41467-026-68870-x
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41467-026-68870-x