Abstract
When mammalian cells are exposed to stress, they co-ordinate the condensation of stress granules (SGs) through the action of proteins G3BP1 and G3BP2 (G3BPs) and, simultaneously, undergo a massive reduction in translation. Although SGs and G3BPs have been linked to this translation response, their overall impact has been unclear. Here we investigate the question of how, and indeed whether, G3BPs and SGs shape the stress translation response. We find that SGs are enriched for mRNAs that are resistant to the stress-induced translation shutdown. Although the accurate recruitment of these stress-resistant mRNAs does require the context of stress, a combination of optogenetic tools and spike-normalized ribosome profiling demonstrates that G3BPs and SGs are necessary and sufficient to both help prioritize the translation of their enriched mRNAs and help suppress cytosolic translation. Together, these results support a model in which G3BPs and SGs reinforce the stress translation programme by prioritizing the translation of their resident mRNAs.
Similar content being viewed by others
Main
When exposed to stresses, such as oxidative stress, high temperature and harmful chemicals, cells initiate the integrated stress response (ISR)1,2,3,4. The ISR integrates signals from different stress sensors to converge on the phosphorylation of translation initiation factor eif2α1,2,3,4,5, which causes reduced global translation while allowing selective translation of stress-responsive mRNAs that help cope with the stress2,4,6,7,8,9. During this dramatic alteration of the translation state, cytoplasmic puncta known as stress granules (SGs) form through the process of liquid–liquid phase separation1,6,10,11,12,13,14,15. These granules are non-membrane-bound collections of RNA-binding proteins, mRNAs and translation machinery1,11,16,17,18,19. Although SGs contain a diverse proteome, their formation requires the RNA-binding proteins G3BP1 and G3BP2 (G3BPs)12,16. Both SGs and G3BPs have been hypothesized to play roles in translation16,20,21,22,23, but their precise impacts are not well understood.
As they are highly enriched for translation machinery and form concurrently with translation shutdown, SGs were originally hypothesized to be required for the dramatic translation shutdown observed during the ISR20,24,25. However, G3BP-knockout (KO) cell lines, which lack microscopically visible SGs, still undergo the eif2α-dependent translation reduction associated with the ISR16,26.
As the formation of SGs is enhanced by polysome collapse, and SGs are enriched in small ribosomal subunits but not large ribosomal subunits, SGs are also proposed to simply be sites devoid of translation1,2,10,20,27,28. However, single-molecule analyses show that translating mRNAs are not forbidden inside of SGs29, and some report that translation inside of SGs might not be a rare event compared with translation in the cytosol30.
Despite their many connections to translation and the stress response, the overall impact that G3BPs and SGs have on translation during stress, or indeed whether they have any impact, is not fully understood10,31,32,33. Previous attempts to determine the effects of G3BPs or SGs on translation have relied either on bulk measurements of translation with limited sensitivity and specificity, such as 35S-labelling of nascent peptides, or have been restricted to observations of single mRNAs and reporters16,21,30. Here, we use spike-normalized ribosome profiling and optogenetic methods to measure the impact of G3BPs and SGs on translation with or without stress. These results support a model in which G3BPs and SGs reinforce the ISR translation programme by prioritizing the translation of their most enriched transcripts.
Results
Ribo-spike enables observation of absolute translation changes during the ISR
Global changes to translation, such as those observed during the ISR, have been challenging to observe by ribosome profiling, which typically lacks information on the absolute differences between samples34. RNA spike ins typically used to quantify absolute differences in other high-throughput approaches are not appropriate for ribosome profiling, as the RNAse treatment used to create ribosome-protected fragments (RPFs) would destroy the unprotected spike molecules. Normalizing to endogenous mitochondrial RPFs can bypass this problem34,35. However, this approach assumes that mitochondrial translation is unaffected by the experimental condition—an assumption that does not always hold, especially if cells are subjected to stress. To use ribosome profiling to measure absolute changes to translation efficiency (TE) occurring as a consequence of stress, we developed a ribosome profiling spike in (ribo-spike) consisting of a defined amount of rabbit reticulocyte lysate translating orthogonal firefly luciferase mRNA, which could then be spiked into each sample (Fig. 1a and Extended Data Fig. 1a).
a, A schematic representation of the ribo-spike. The ribo-spike consists of polysomes formed on an orthogonal mRNA sequence, in this case, firefly luciferase mRNA (fLuc) translated in a rabbit reticulocyte lysate. A defined amount of the ribo-spike sample is added to each experimental sample, ultimately generating orthogonal RPFs that enable normalization between samples. b, The ISR translation programme induced upon arsenite stress. Plotted for each mRNA is the log2 fold change (FC) in TE observed in HCT116 cells responding to 500 µM sodium arsenite (for 1 h) as a function of the mRNA expression. The dashed red line indicates unchanged TE, as determined by the ribo-spike. Points for stress-enhanced, stress-resistant and stress-hypersensitive mRNAs are indicated by colours, with exemplar mRNAs listed for each category. Stress response categories were determined by DESeq2 (log2 FC >1, adjusted P value <0.05). Mitochondrially translated mRNAs are indicated in orange. n indicates the number of unique mRNAs. c, The ISR translation programme induced upon heat shock (45 °C for 25 min), otherwise as in a.
Using our ribo-spike, we then compared translation between unstressed cells and cells treated with 500 µM sodium arsenite for 1 h. This dose of sodium arsenite activates the ISR, leading to eif2α phosphorylation and a substantial reduction in global translation16,20. By normalizing to the spike-in TE across samples, we were able to measure absolute changes in TE for mRNAs of >10,000 individual genes. Aggregating these measurements revealed a drastic (20 fold) reduction in global TE, consistent with previous bulk measurements16,36,37 (Fig. 1b and Extended Data Fig. 1b,c). Interestingly, TEs of mitochondrial mRNAs increased in the presence of sodium arsenite (Fig. 1b), validating our concern that they might not remain constant under stress.
Although translation was globally reduced during stress, some mRNAs retained or even enhanced their translation in response to stress. Among them were mRNAs of several stress-responsive genes previously reported to maintain their translation during the ISR, including ATF4, ATF5, CEBPα and DDIT3/CHOP (Fig. 1b). Many of these mRNAs contain upstream open reading frames (uORFs), which can help regulate their translation during stress38. Indeed, regulation by uORFs is proposed to help shape the general translation response to stress2,4,7,8,39,40,41. Supporting this proposal, mRNAs reported to contain one or more functional uORFs were significantly better translated during the stress response42 (Extended Data Fig. 1d). Moreover, while our manuscript was in review, another study published the development of a similar ribo-spike and also applied it to show that uORF-containing mRNAs are better translated during stress41.
In yeast, newly transcribed mRNAs tend to be preferentially translated during stress43,44. To assess this possibility in mammalian cells, we compared the translation change in response to stress to mRNA half-lives, reasoning that mRNAs with short half-lives would be predominantly composed of newly synthesized transcripts. Consistent with the observations made in yeast, we observed a moderate negative correlation between the translation change of an mRNA upon stress and its estimated half-life45 (Spearman R (RS) of −0.22; Extended Data Fig. 1e).
At the other extreme, some mRNAs were especially sensitive to translation shutdown, including CSDE1 and PCBP2, as reported previously40. Interestingly, other hypersensitive mRNAs encoded proteins involved in energetically expensive processes, such as vesicle trafficking (COPS3, COPS6, CLTC and SCFD1), which are strongly downregulated during stress46 (Fig. 1b).
To measure absolute TE changes in another stress context, we performed analogous experiments under 45 °C heat shock (Fig. 1c and Extended Data Fig. 1b,c). The results resembled those observed for arsenite stress (Fig. 1c and Extended Data Fig. 1f,g). However, the translation changes were not identical (RS of 0.63) and many mRNAs were called as differentially regulated in one stress but not the other (Extended Data Fig. 1h–j), consistent with previous reports that the type and dosage of stress shape the ISR3.
We consider the combination of widespread translation shutdown, the retained or increased translation of select transcripts and the hypersensitive downregulated translation of others to be the ISR translation programme. These results illustrate how using ribo-spike enables accurate and precise monitoring of absolute translation changes between samples and emphasize the utility of a truly orthogonal spike-in for studies monitoring global changes to translation.
G3BPs reinforce the ISR translation programme
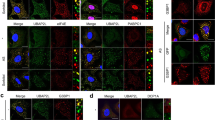
Having developed a method to globally monitor absolute TE changes, we wanted to use it to determine the effects of G3BPs and SGs on the ISR. To do so, we needed to be able deplete cells of G3BPs and SGs. Therefore, we engineered HCT116 cells in which both endogenous G3BP1 and endogenous G3BP2 were homozygously tagged with RFP and an auxin-inducible degron (AID) allowing their rapid and efficient depletion (>90% within 3 h) upon addition of indole-3-acetic acid (IAA)47,48. By assessing another SG marker protein, PABPC1 labelled with GFP, we confirmed that our depletion of G3BPs before stress imparted minimal effects on PABPC1 stability and largely prevented the formation of SGs in response to both sodium arsenite and heat shock (Fig. 2a–f).
a, The depletion of endogenous G3BPs fused to AID. Immunoblots probed for the indicated proteins show specific depletion of AID fusion proteins after treatment with 500 µM IAA for 3 h. b, Quantification of protein depletion in a, normalizing to levels of GAPDH. The points show values for two biological replicates. c, The prevention of SG formation during arsenite stress. HCT116 cells were either treated with 500 µM IAA to deplete G3BPs (right) or not treated (left) before arsenite stress (500 µM sodium arsenite for 1 h). The images show either G3BPs (red) or SG marker protein PABPC1 (green) (scale bar, 10 µm). d, The quantification of SG formation in c. The points show values for three biological replicates (***P = 6.5 × 10−5; Welch’s two-sample, two-tailed t-test). e,f, Images (e) and quantification (f) of the prevention of SG formation during heat shock (45 °C for 25 min; ***P = 0.001), otherwise as in c and d. g, A schematic representation of the G3BP-depletion experiment. Ribosome profiling was performed on stressed HCT116 cells to determine the effects of stress on translation, G3BP-depleted unstressed HCT116 cells were used to determine the effect of G3BPs on translation and G3BP-depleted stressed HCT116 cells were used to determine the effects of G3BPs/SGs on translation. The effects of stress were then compared with the effects of G3BPs and SGs. h, The relationship between G3BP-dependent translation during arsenite stress and the ISR translation programme. The plots indicate the log2 FC in TE owing to the presence of G3BPs in cells treated with arsenite as a function of the log2 FC in TE owing to arsenite. Points for stress-enhanced, stress-resistant and stress-hypersensitive mRNAs are indicated by colours. n indicates the number of unique mRNAs. i, The relationship between G3BP-dependent translation in the absence of stress and the ISR translation programme, otherwise as in h. j, The relationship between G3BP-dependent translation during heat shock and the heat shock ISR translation programme, otherwise as in h. k, The relationship between G3BP-dependent translation in the absence of stress and the ISR translation programme, otherwise as in h.
Having generated these degron lines, we used ribosome profiling with the ribo-spike to compare translation in cells with and without the ability to form SGs (Fig. 2g). Compared with G3BP-depleted cells, normal cells underwent a somewhat stronger inhibition of global translation during arsenite stress (1.4 fold; Extended Data Fig. 2a), consistent with reports that G3BPs are largely dispensable for the global reduction in translation caused by the ISR16.
We were intrigued to find that G3BPs, and perhaps SGs, did seem to contribute, if only modestly, to the massive reduction in translation that occurs during the ISR. To further examine this contribution, we compared the translation changes caused by presence of G3BPs with those caused by activation of the ISR. Here, we observed a clear positive correlation (RS of 0.33; Fig. 2h and Extended Data Fig. 2b). This implied that G3BPs, and perhaps SGs, tend to translationally upregulate the same mRNAs that the ISR upregulates and tend to translationally downregulate the same mRNAs that the ISR downregulates, thereby reinforcing the ISR translation programme.
To determine whether these G3BP-dependent changes to TE were driven by effects on RPFs or RNA, we examined these two measurements separately. Interestingly, changes to RPFs and mRNA levels both contributed to the overall TE effects, with the presence of G3BPs leading to more RPFs of ISR-enhanced transcripts despite a reduction in the levels of these transcripts (Extended Data Fig. 2c,d). Although these G3BP-dependent decreases in mRNA might represent an independent function of G3BPs, they are perhaps more parsimoniously explained as the translation-dependent destabilization of mRNA, which has been previously reported49,50,51,52,53,54. Indeed, we also observed evidence of such mRNA stabilization occurring transcriptome-wide in response to arsenite stress (Extended Data Fig. 2e). However, the previously observed correlation between translation change upon stress and unstressed mRNA half-lives might complicate this interpretation (Extended Data Fig. 1e).
Given that the ISR translation programmes differed between arsenite and heat shock, we examined whether G3BPs also reinforced the heat shock ISR translation programme and found similar results (Fig. 2j and Extended Data Fig. 2f–j). Furthermore, analyses of published measurements of G3BP-dependent translation in U2OS cells treated with the endoplasmic reticulum stressor thapsigargin, also yielded similar results55 (Extended Data Fig. 3a–e). Taken together, these results indicated that G3BPs, and perhaps SGs, reinforce the ISR translation programme across multiple stresses and cell types.
We next asked whether the factors correlating with ISR translation—uORF presence and estimated mRNA half-lives—also correlated with G3BP-dependent translation during both arsenite stress and heat shock. However, we found no strong relationship between either of these factors and G3BP-dependent translation (Extended Data Fig. 4a–d).
To determine whether the G3BP-dependent translation was constitutive or stress specific—potentially through the formation of SGs—we performed analogous ribosome profiling experiments, comparing translation in cells with and without G3BPs, but this time in the absence of stress. Interestingly, in unstressed cells, G3BP-dependent translation changes showed no positive correlation to the translation changes of arsenite or the heat-shock ISRs (RS values of –0.033 and –0.12, respectively; Fig. 2i,k). Similar results were observed when analysing published data from G3BP-KO U2OS cells55 (Extended Data Fig. 3b). Together, these results argued that G3BPs reinforce the ISR translation programme in a stress-dependent manner, consistent with a model in which this is a function of G3BP through its nucleation of SGs.
As G3BPs are required for SG formation, we could not say whether the effects we observed upon depleting G3BPs were a consequence of losing SGs or whether they were a consequence of losing some other stress-specific G3BP function. We sought to disambiguate G3BP and SG function by targeting CAPRIN1 or UBAP2L, two other proteins reported to be required for SG formation16,19,56,57. However, no reduction in SG formation was detected upon AID depletion of either of these proteins (Extended Data Fig. 4e–m). These results concurred with findings that, in many cell types, G3BPs are uniquely required for SG formation, and illustrated the difficulties in disentangling the functions of SGs from those of their required G3BP components12.
SGs are enriched for mRNAs that are favoured by the ISR translation programme
After difficulty using this genetic approach to define the role of SGs, we turned to other approaches. One attractive hypothesis was that mRNAs that are differentially regulated by the ISR translation programme are also differentially localized to SGs and that this localization to SGs determines the effect that G3BPs have on the translation of those mRNAs during stress.
To assess the relationships between regulation by the ISR, regulation by G3BPs and SG localization, we purified SG cores from HCT116 cells treated with 500 µM sodium arsenite for 1 h18,58. Sequencing identified 488 transcripts enriched in SG cores and 453 that were depleted compared with total cytoplasm (>2-fold change, adjusted P value <0.05; Fig. 3a). Overall, our SG enrichments observed in HCT116 cells resembled those previously reported from U2OS cells18, including an enrichment for longer mRNAs and mRNAs that were poorly translated before stress. This indicated that we had successfully purified SG cores (Extended Data Fig. 5a–d).
a, mRNAs enriched and depleted in SGs. Plotted for each mRNA is its abundance in the SG fraction as a function of its abundance in the cytoplasmic fraction. Abundance is reported in reads per million (RPM). Points for SG-enriched and SG-depleted mRNAs, as determined by DESeq2 (log2 FC >1, adjusted P value <0.05), are indicated in red and blue, respectively, with their numbers in parentheses. n indicates the total number of unique mRNAs. b, The relationship between SG enrichment and ISR translation. Plotted are the SG enrichments of mRNAs in each of three ISR translation categories: stress-enhanced/resistant, bulk behaviour and stress-hypersensitive (lines represent the median, notches represent the 95% confidence interval, boxes represent quartiles and whiskers represent 1.5× interquartile ranges (IQRs); ***P = 7 × 10−26, 9 × 10−56 and 6 × 10−26 (from left to right) Welch’s two-sample, two-tailed t-test; n = 10,383 unique mRNAs). c, The relationship between G3BP-dependent translation and SG enrichment. The plots indicate the log2 FC in TE owing to presence of G3BPs in stressed cells for mRNAs in each of three SG-enrichment categories: SG enriched, SG depleted and neither (***P = 2.6 × 10−5, 1.2 × 10−15 and 5.6 × 10−15 (from left to right); otherwise as in b). All stresses were 500 µM sodium arsenite for 1 h.
We next examined the relationship between ISR regulation and SG enrichment and found that mRNAs that retained or enhanced their translation during stress tended to be more enriched in SGs, whereas hypersensitive mRNAs tended to be depleted (Fig. 3b). Furthermore, analyses of SG enrichment in U2OS cells treated with sodium arsenite18, stress-dependent RNA-granule enrichment in NIH 3T3 cells treated with thapsigargin59 and stress-dependent G3BP proximity labelling in HEK293T cells treated with sodium arsenite60 yielded similar, albeit sometimes weaker, trends (Extended Data Fig. 5f–j). Together, these results supported a model in which mRNAs that are favoured by the ISR translation response preferentially localize to SGs and suggest that this model might be generalizable across multiple stresses and cell types.
SG-enriched mRNAs are favoured by G3BPs during stress
We next examined the relationship between G3BP regulation and SG enrichment in our HCT116 cells. Surprisingly, the presence of G3BPs tended to enhance the TE of SG-enriched mRNAs during arsenite stress, whereas the presence of G3BPs tended to repress the TE of mRNAs depleted from SGs during arsenite stress (Fig. 3c). Moreover, repeating this analysis with SG enrichment data from arsenite-treated U2OS cells18, RNA-granule enrichment data from heat-shocked NIH 3T3 cells59 and G3BP proximity labelling data from arsenite-stressed HEK293T cells60 all yielded analogous results (Extended Data Fig. 4k–m). Thus, in a variety of stresses and cell types, the G3BPs appeared to favour the translation of SG-enriched mRNAs or disfavour the translation of SG-depleted mRNAs, suggesting a model in which those transcripts that are most strongly upregulated by the ISR translation programme localize to SGs, leading to further upregulation by G3BPs.
SG-like OGs drive global reduction in translation without a full ISR
Our global analyses showed that G3BPs, while not required to establish the ISR translation programme, do reinforce it. To determine whether SG formation was indeed sufficient to drive these translation changes in the absence of exogenous stress, we set out to form ectopic SGs in the absence of exogenous stress. Overexpressing reported SG-nucleating proteins UBAP2L and G3BP1 (refs. 61,62) failed to induce authentic SGs in a majority of cells. UBAP2L-induced puncta lacked the canonical SG marker eIF4G, and, while puncta induced by G3BP1 overexpression did resemble authentic SGs, they formed in only ~20% of cells (Extended Data Fig. 6a–d).
To more robustly form ectopic SGs, we adapted a published CRY2 optogenetic system63. We replaced the N-terminal NTF2L dimerization domain of G3BP1 with the blue light-dependent cryptochrome 2 photolyase homology region oligomerization domain (CRY2), tagged with GFP (GFP::CRY2::G3BPΔN) (Fig. 4a). Even in the absence of blue light, doxycycline-induced expression of this construct caused formation of ectopic puncta in ~20% of cells, similar to overexpressing GFP::G3BP1. However, exposing these cells to 488-nm blue light enhanced puncta formation, such that they were observed in ~80% of cells within 3 h. Consistent with previous findings63, our light-induced granules were also reversible (disassembling to basal levels within 2 h of blue light removal), positive for canonical SG markers eIF4G and CAPRIN1 and exhibited photobleaching dynamics resembling those of canonical SGs (Fig. 4a,b and Extended Data Fig. 6e–g). Together, these results showed that our OptoGranules (OGs) were dynamic, reversible, SG-like condensates formed in the absence of exogenous stress.
a, A schematic representation of the OG system. HCT116 cells expressing endogenous G3BP containing an NTF2L dimerization domain were edited to also express doxycycline (dox)-inducible ectopic G3BP in which the NTF2L domain had been replaced with a fusion of GFP and the light-inducible CRY2 dimerization domain (GFP::CRY2::G3BPΔN). After doxycycline induction, a minority of cells formed ectopic granules. After exposure to blue light (488 nm), most cells formed ectopic granules. b, SG marker localization to OGs. HCT116 cells expressing wild-type GFP::G3BP1 were either stressed with 500 µM sodium arsenite for 1 h (top) or treated with 1 µM doxycycline to induce expression of GFP::CRY2::G3BPΔN and exposed to blue light for 3 h (bottom). Fluorescent or immunostained proteins are as indicated (scale bars, 10 µm). White boxes highlight insets that are expanded on the right. n = 3 replicates with similar results. c, The comparison of TE in OG-induced cells to that in blue-light control cells. Plotted are the TE distributions in HCT116 cells in which GFP::CRY2::G3BPΔN was expressed and exposed to blue light to form OGs (right) and in control cells expressing only wild-type G3BP exposed to blue light, in which no OGs were formed (left) (lines represent medians, notches represent 95% confidence intervals, boxes represent quartiles and whiskers represent 1.5x IQRs; n = 10,129 unique mRNAs). Significance was determined using ribo-spike values as in Extended Data Fig. 1b (*P = 0.026 with Welch’s two-sample, two-tailed t-test). d, The comparison of OG and ISR translation programmes. Plotted is TE change caused by induction of OGs as a function of TE change caused by arsenite. n indicates the total number of unique mRNAs. e, A comparison of OG and G3BP-dependent translation programmes. Plotted is TE change caused by OGs as a function of TE change caused by the presence of G3BPs in arsenite-treated cells, otherwise as in c. f, A comparison of OG translation programme and SG enrichment. The plots indicate the log2 FC in TE caused by the induction of OGs for mRNAs in each of three SG enrichment categories: SG enriched, SG depleted and neither (lines represent medians, notches represent 95% confidence intervals, boxes represent quartiles and whiskers represent 1.5× IQRs; n = 10,129 unique mRNAs. ns, not significant; ***P = 6.9 × 10−9, 2.3 × 10−5 and 0.067 (left to right) Welch’s two-sample, two-tailed t-tests).
If SG-like condensation alone was sufficient to drive an ISR-like response, then we would predict that induction of OGs would yield the following results: first, cells with OGs would undergo a global reduction in translation. Second, these OG-induced translation changes would positively correlate with ISR-induced translation changes. Third, OG-induced translation changes would also positively correlate with G3BP-dependent translation during stress. Fourth, transcripts enriched in SGs would be resistant to any translation shutdown caused by the formation of these SG-like condensates.
To test these predictions, we induced OGs in HCT116 cells and used ribosome profiling with our ribo-spike to compare them with cells exposed to blue light but lacking GFP::CRY2::G3BPΔN, and therefore lacking OGs. We observed a modest global TE reduction in OG-induced cells when compared to blue-light controls (Fig. 4c). This modest reduction (1.6 fold) agreed with our earlier result that G3BPs were responsible for a small fraction of the ISR translation shutdown (1.4 fold for arsenite stress) (Extended Data Fig. 2a) and was consistent with the first of our four predictions. However, analyses of the TE results failed to confirm any of the remaining three predictions. OG translation changes did not correlate with either the ISR translation programme or G3BP-dependent translation changes in stressed cells (RS values of 0.019 and –0.054, respectively; Fig. 4d,e), and mRNAs that were enriched in SGs did not perform any better than those that were neither enriched nor depleted (Fig. 4f). Considered together, these results argued that condensation of the SG-like OGs, while sufficient to drive a modest reduction in global translation, was not sufficient to specify the ISR translation programme.
OGs require cellular stress to recruit an SG transcriptome
One caveat of our previous analysis was the assumption that spontaneously formed OGs contained the same transcriptome as SGs and could therefore regulate those transcripts in the same manner. This assumption aligned with evidence that G3BP was the central scaffold of the SGs complex protein–RNA interaction network and was further supported by the observation that OGs were positive for many canonical SG markers12,13,14,63 (Extended Data Fig. 6e). Interestingly, this assumption was also consistent with the observation that the established determinants of the SG transcriptome (transcript length and TE before stress) are parameters that are independent of stress18. However, the SG transcriptome is also thought to be largely determined by the transcriptome RNA-binding landscape (the landscape created by proteins bound to the transcriptome)10,12,64,65. Stress can dramatically remodel this RNA-binding landscape, shifting the RNA content of other biological condensates, such as P bodies and P granules66,67. Thus, SGs formed in the presence of stress and OGs formed in the absence of stress might recruit different sets of mRNAs, despite their general compositional, morphological and biophysical similarities.
To test this hypothesis, we purified OGs formed in the presence of stress. To do this, we integrated the doxycycline-inducible GFP::CRY2::G3BPΔN construct into cells expressing endogenously edited AID::RFP::G3BPs and doxycycline-inducible OsTIR1, the E3 ligase responsible for the turnover of the AID. Upon treatment with doxycycline and IAA, these cells degraded their endogenous G3BPs and replaced them with GFP::CRY2::G3BPΔN, rendering them incapable of forming SGs in the absence of blue light, even when stressed. However, when these cells were stressed and exposed to blue light, they readily formed granules (Fig. 5a,b). We refer to these OGs formed in the presence of stress as StressOptoGranules (SOGs).
a, The replacement of G3BP with GFP::CRY2::G3BPΔN to induce SOGs. HCT116 cells expressing endogenously tagged AID::RFP::G3BPs were either untreated or treated with 1 µM doxycycline (dox) and 500 µM IAA for 18 h, leading to turnover of endogenous G3BP (red) and expression of GFP::CRY2::G3BPΔN (green) (left). Cells were treated as on the left, but were subsequently stressed with 500 µM sodium arsenite for 1 h in the absence of blue light (middle). Cells were treated as in the middle, but were also exposed to blue light, beginning 30 min after the start of the arsenite treatment (right) (scale bars, 10 µm). White boxes highlight insets that are expanded on the right. b, The quantification of granule formation in a. Error bars represent the s.d. for three biological replicates (n.s., P = 0.48; ***P = 5.9 x 10−9; and n.s., P = 0.071 (left to right) Welch’s two-sample, two-tailed t-test). c, A schematic representation of the granule purification experiment. Three cell lines were used: the parental HCT116 line expressing endogenous G3BPs; an OG HCT116 line expressing endogenous G3BPs and doxycycline-inducible GFP::CRY2::G3BPΔN; and an SOG line expressing endogenous G3BP fused to AID and RFP (RFP::AID::G3BP), doxycycline-inducible OsTIR1 and doxycycline-inducible GFP::CRY2::G3BPΔN. Control SGs were purified from each cell line by treating them with 500 µM sodium arsenite for 1 h. In addition, OGs were purified from the OG line treated with doxycycline for 18 h, followed by 3 h of blue light, and SOGs were purified from the SOG line treated with both doxycycline and IAA for 18 h, followed by exposure to arsenite stress for 30 min in the absence of blue light and then an additional 30 min of stress in the presence of blue light. d, mRNAs enriched and depleted in the indicated types of granules, otherwise as in Fig. 3a. e, Comparisons of granule enrichments. The heat map depicts the pairwise correlations (RS values) observed between the enrichments of granules from Figs. 3a and 5d. f, Comparisons of cytoplasmic transcriptomes of cells used to determine granule enrichments in e. g, A comparison of OG-dependent translation and OG enrichment. Plotted are distributions of TE changes caused by the induction of OGs for mRNAs in each of three OG enrichment categories, ***P = 9.5 × 10−9, 1.5 × 10−17 and 9.5 x 10−11 (left to right), otherwise as in Fig. 4f.
We then purified and sequenced the transcripts associated with both OGs and SOGs. We compared transcripts across OGs, SOGs and control SGs from each cell line, including our original SG enrichment data acquired from the parental line (Fig. 5c,d). Control SGs correlated strongly with each other (RS values of 0.72–0.75). OGs formed in the absence of stress, correlated substantially less with control SGs (RS values of 0.35–0.45), indicating a distinct transcriptome (Fig. 5e and Extended Data Fig. 7a). Interestingly, we found that transcripts enriched in SOGs strongly resembled those enriched in SG controls (RS values of 0.69–0.70), indicating that the difference between SG and OG transcriptomes was because of a lack of cellular stress, as opposed to some intrinsic bias of OG formation (Fig. 5e and Extended Data Fig. 7a). These differences in granule composition did not appear to be driven by differences in gene expression, as the cytoplasmic mRNA levels correlated quite well between these groups (RS values of 0.84–0.96) (Fig. 5f and Extended Data Fig. 7b). Taken together, our results indicated that establishing the SG transcriptome requires not only the interaction network established by G3BPs but also the RNA-binding landscape established during cellular stress.
These OG enrichment data also allowed us to confirm that the transcripts enriched in OGs did indeed better retain their translation (Fig. 5g). Taken together, our OG ribosome profiling and purification results support a model in which SGs are sufficient to drive a modest decrease in global translation while prioritizing the translation of their enriched mRNAs. However, SGs required stress to induce an ISR-like translation programme, presumably because the sorting of SG mRNAs is dictated by the cellular RNA-binding landscape, which is drastically remodelled upon activation of the ISR.
Tethering to SGs imparts resistance to ISR translation shutdown
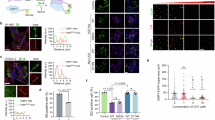
Our findings suggested that localization to an SG grants an mRNA prioritized translation during the ISR. To test this model, we examined whether tethering a reporter to SGs influenced its translation. The reporter-encoded nanoluciferase (nLuc) fused to the Escherichia coli dihydrofolate reductase (ecDHFR) destabilizing domain, which causes rapid turnover of its protein fusions, ensuring reporting on recently translated nLuc68. The 3′-UTR of the reporter mRNA included an array of 24 bacteriophage MS2 hairpins, which bind MS2 coat protein (MCP), thereby providing a means to tether the reporter to the SG30,69 (Fig. 6a).
a, A schematic representation of G3BP1 tethering. b, Tethering a luciferase reporter to SGs via G3BP1. HCT116 cells expressing a NanoLuc reporter bearing 5′ and 3′ UTRs from CNOT10 and endogenous G3BP1, tagged with either GFP or MCP::GFP, were stressed with 500 µM sodium arsenite for 90 min. The images show G3BP1 (green) and smFISH of NanoLuc reporter molecules (magenta) (scale bars, 1 µm). White boxes highlight insets on the right. n = 3 replicates with similar results. c, The G3BP1 tethering efficiency. The percentage of SG-localized reporter molecules in either untethered cells (green) or tethered cells (pink) are shown. The points show values for biological replicates (***P = 3.6 × 10−6 Welch’s two-sample, two-tailed t-test; n = 3 biological replicates). d, G3BP1-tethered ISR translation. Plotted is the TE of the reporter in untethered cells (green) or tethered cells (pink) during a time course of arsenite stress (500 µM sodium arsenite). The bold line shows average values from at least three biological replicates (shown as thin lines). Light-coloured ribbons report the s.e.m. TE is reported relative to that of untethered, unstressed cells from the matched biological replicate. e, A schematic representation of CAPRIN1 tethering. f, The quantification of SG formation in cells treated as depicted in e. Error bars represent the s.d. for three biological replicates (n.s., P = 1; ***P = 5.9 × 10−8 Welch’s two-sample, two-tailed t-test). g, CAPRIN1-tethered ISR translation. Plotted is the relative TE of the reporter in untethered cells (green), tethered cells with SGs (+SGs; pink), and tethered cells without SGs (–SGs; red), otherwise as in d. h, A schematic representation of the polysome profiling experiment. We performed polysome fractionation on cells treated with 500 µM sodium arsenite, added an in vitro transcribed RNA standard to each fraction, isolated the total RNA and performed RT–qPCR to measure the abundance of the reporter relative to the standard. i, The polysome profile analysis of MCP::GFP::G3BP1-expressing cells. Plotted are the polysome traces produced by the workflow depicted in h. The x axis shows 12 equal-volume fractions that were taken with boundary points between fractions marked as ticks. j, Reporter distributions across G3BP1-tethered polysome profile fractions. Plotted are the proportions of total reporter mRNA in each polysome fraction as measured in h and i by RT–qPCR, otherwise as in i. k, The relative ribosome loading of the G3BP1-tethered reporter. Plotted is the total ribosome load as determine by polysome profiling of the G3BP1-tethered reporter relative to its untethered counterpart. l, The polysome profile analysis of MCP::GFP::CAPRIN1-expressing cells. Plotted are the polysome traces for untethered cells expressing endogenous G3BPs (green), tethered cells expressing MCP::GFP::CAPRIN1 along with endogenous G3BPs (pink) and tethered cells expressing MCP::GFP::CAPRIN1 with endogenous G3BPs depleted (red), otherwise as in i. m, Reporter distributions across CAPRIN1-tethered polysome profile fractions. Plotted are the proportions of total reporter mRNA in each polysome fraction of conditions shown in l. Plots are shown for stress timepoints of 0 and 15 min, otherwise as in j. n, The relative ribosome loading of the CAPRIN1-tethered reporter. Plotted is the total ribosome load as determine by polysome profiling of the CAPRIN1-tethered reporter in the presence of G3BPs (pink) and in their absence (red) relative to their untethered counterpart (green), before and during arsenite stress, otherwise as in k. o, A plausible model of SG translation control: (1) ISR-enhanced transcripts are preferentially recruited to SGs between cycles of translation, when they lack ribosomes; (2) as a consequence of being recruited to SGs, these transcripts are licensed for translation, possibly through the recruitment of G3BPs; and (3) after this licensing, this model is agnostic to whether the transcript diffuses out to the cytosol or remains inside the SG to be translated. Once these transcripts complete their translation, they would be available to, once again, be preferentially recruited to (or retained in) the granule.
G3BPs strongly partition into SGs16,62,70; almost half of our endogenous G3BP1 fusion protein (45.9%) localized to SGs after 90 min of arsenite stress (Extended Data Fig. 8a,b). Considering this high partitioning coefficient, we chose an MCP fusion to endogenous G3BP1 as our SG-tethering protein. Our reporter was stably integrated into HCT116 cells in which all endogenous G3BP1 alleles were tagged with either GFP (GFP::G3BP1) or MCP and GFP (MCP::GFP::G3BP1) (Fig. 6a). We also explored the λN–BoxB aptamer system71,72, but found that it perturbed G3BP localization and SGs (Extended Data Fig. 8c).
To assess the effects of SG tethering on an mRNA not normally recruited to SGs, we created a reporter using the 5′ and 3′ UTR of CNOT10, an mRNA that was not enriched in SGs in either our data or published datasets18, and was translationally repressed both by the ISR and by G3BPs during stress (Supplementary Table 1). Single-molecule fluorescence in situ hybridization (smFISH) with probes targeting the nLuc sequence indicated that the reporter was recruited to SGs at a basal level of 38% in cells expressing only GFP::G3BP (Fig. 6b,c). In cells expressing MCP::GFP::G3BP, the reporter was more robustly recruited to SGs (92%) (Fig. 6b,c), a higher fraction than that observed for 99% of endogenous mRNAs18.
Next, we measured how the translation of this reporter responded to arsenite stress. Cells were treated with 500 µM sodium arsenite for timepoints ranging from 0 to 90 min, and protein production was monitored by measuring the luciferase signal. Reporter mRNA levels were then monitored by reverse transcription–quantitative PCR (RT–qPCR) to calculate a TE for each timepoint. For the untethered reporter, we observed a 95% reduction in reporter TE over the 90 min period of stress (Fig. 6d). This aligned with the 20-fold average decrease in TE observed by ribosome profiling, indicating that our assay accurately reported on stress-induced translation changes.
We then examined what happens to the translation of this reporter when tethered to MCP::GFP::G3BP1. Strikingly, even when tethered to G3BP1 in unstressed cells, our reporter was more efficiently translated, with an average threefold higher TE. This observation was consistent with previous reports that G3BPs can act as translational regulators, even outside the context of stress16,21,22,23,73,74 (Fig. 6d). When stressed, we saw that our G3BP1 tethering continued to enhance the translation of our reporter. Interestingly, this translation enhancement peaked early in stress, as translation both in tethered and untethered cells diminished at later timepoints. However, even at our latest timepoints, our tethered reporter was translated significantly better than its untethered counterpart (Fig. 6d).
To test whether these effects on translation were specific to this reporter, we created two additional reporters using 5′ and 3′ UTRs from the genes DDIT4L and RB1, which represented a range of endogenous translational responses to stress and to G3BPs (Supplementary Table 1). The results for both of these reporters resembled those observed for CNOT10, with the tethered reporter showing enhanced SG recruitment and translation across all stressed timepoints, peaking early in stress and then decreasing at later timepoints (Extended Data Fig. 8e–g).
We next wanted to determine whether, as in our ribosome profiling experiment, changes to mRNA levels contributed to these changes in TE. Examining protein and RNA separately, we saw that, although the rate of luciferase production was modestly higher for some tethered reporters, as evidenced by the slower rate of luciferase disappearance upon stress (Extended Data Fig. 8h,i), the untethered reporter mRNA levels consistently increased across all reporter constructs during the stress treatment, by an average of 5.2 fold, whereas G3BP1-tethered mRNA levels remained steady or slightly decreased, with an average change of 0.9 fold (Extended Data Fig. 8j,k). This pattern was consistent with a model in which translation destabilizes the reporter mRNA, the stress-dependent translation shutdown stabilizes it and recruitment to SGs, in rescuing translation, destabilizes the reporter yet again49,50,53,54.
To test this model, we treated cells expressing our untethered CNOT10 reporter with two different translation inhibitors—cycloheximide and puromycin (puro)—and monitored protein and mRNA levels. As expected, both drugs inhibited translation by 98% over a 4 h time course (Extended Data Fig. 8l,m). Both drugs also caused dramatic stabilization of reporter mRNA (cycloheximide by tenfold and puro by 22 fold; Extended Data Fig. 8n). These results indicated that our reporters are subject to translation-dependent destabilization, and are consistent with a model in which the translation shutdown during stress contributes to the stabilization of our reporters.
To further distinguish between SG and G3BP function, we examined whether the effect of tethering was specific to G3BPs. To do this, we tethered our CNOT10 reporter to SGs using another SG protein, CAPRIN1. CAPRIN1-mediated recruitment to SGs was robust, resembling that observed with G3BP1 tethering (Extended Data Fig. 9a,b). To measure the translation effects of this alternative tethering strategy, we integrated both the reporter and doxycycline-inducible, MCP-tagged CAPRIN1 into our AID::G3BP cell line. With this combination, we could assess the effects of tethering the reporter to SGs via CAPRIN1 and also deplete G3BPs to control for any effects that did not require formation of SGs (Fig. 6e).
We first expressed the reporter in untreated cells without the induction of any MCP-tagged protein (Fig. 6f and Extended Data Fig. 9c). In this condition, our reporter underwent a ~75% reduction in TE, similar to our previous experiments (Fig. 6g). We then induced MCP-tagged CAPRIN1 (MCP::GFP::CAPRIN1) without depleting the AID-tagged G3BPs by treating cells with both 1 µg ml−1 doxycycline and 200 µM auxinole, a small-molecule inhibitor of OsTIR1 activity (Fig. 6f and Extended Data Fig. 9c). Under these conditions, the CAPRIN1-tethered reporter was more efficiently translated following arsenite treatment, indicating that tethering effects do not require tethering via G3BP (Fig. 6g). However, these benefits to translation were abolished when cells expressing MCP::GFP::CAPRIN1 were depleted of G3BPs by treating cells with both 1 µg ml−1 doxycycline and 500 µM IAA (Fig. 6f,g and Extended Data Fig. 9c), indicating that this effect of tethering required the presence of G3BPs and perhaps SGs.
As with our G3BP1-tethering experiments, we then replicated these CAPRIN1-tethering experiments using both the DDIT4L and RB1 reporters. We observed results similar to those obtained with CNOT10 at the TE, protein and RNA levels, consistent with the idea that the reporter was better translated only when recruited to SGs, and thus underwent translation-dependent destabilization (Extended Data Fig. 9d–i). Interestingly, the average effect size observed with CAPRIN1 tethering was smaller than that observed with G3BP1 tethering and, unlike our G3BP1-tethered reporters, CAPRIN1-tethered reporters showed only modestly improved translation in unstressed cells (Extended Data Fig. 9d,e). This difference may suggest that, while heavily G3BP-bound mRNAs are subject to enhanced translation, even in unstressed conditions, other mRNAs only benefit once they are recruited to the SG and brought into proximity with G3BPs.
To further measure the effect of SG association on translation by an orthogonal method less likely to be affected by mRNA stability or the kinetics of luciferase production and degradation, we monitored the ribosome association of our CNOT10 reporter using polysome profiling RT–qPCR (Fig. 6h). As expected, untethered reporter mRNA shifted gradually from heavy polysomes to lighter polysomes, and finally to monosomes and free RNA fractions over a 60 min arsenite treatment time course. However, G3BP-tethered reporter mRNA was associated with heavier polysome fractions compared with its untethered counterpart, even in unstressed cells, where SGs were not formed. In addition, this preferential association peaked early in stress and diminished by the 60-min timepoint, when SGs were still fully formed but translation had largely shut down (Fig. 6j,k). Both the preferential polysome association of our G3BP-tethered reporter in the absence of SGs, and the collapse in this preference at later timepoints suggested that this shift was not due to the inclusion of the reporter in heavy-sedimenting granules, but due to more efficient loading into actual ribosomes. These results indicated that tethering to G3BP enhanced reporter translation in both unstressed and stressed conditions, and were consistent with the results of our luciferase assays.
To further evaluate the results of our luciferase assays, we performed polysome profiling of the CNOT10 reporter in CAPRIN1-tethered cells under unstressed and early stress (15 min) conditions. Consistent with results of our luciferase assay, CAPRIN1 tethering enhanced ribosome association only during stress and in a G3BP-dependent manner (Fig. 6l–n). The G3BP dependence of this polysome shift further supported the idea that it was not caused simply by CAPRIN1 protein sedimenting farther into the gradient. Collectively, the results of these tethering experiments indicated that tethering the 3′ UTR of an mRNA to either G3BP1 or CAPRIN1 increased recruitment to SGs and imparted preferential translation during the ISR—consistent with a model in which localization to SGs grants an mRNA prioritized translation during stress.
Discussion
SGs were once thought to be required for the global repression of translation observed during the ISR1,11,20,24,25. Later studies hypothesized that SGs might have no function at all and are incidental by-products of increased RNA availability owing to translation repression32,33. Here, we propose a model in which SGs are neither required for the ISR nor functionless condensates, but instead measurably enhance and reinforce the ISR translation programme, thereby leading to a widespread and statistically significant, albeit relatively modest, impact on global translation.
This widespread yet subtle reinforcement of the ISR is consistent with several observations of SG biology. SGs recruit a wide array of mRNAs and proteins and yet contain only ~10% of cytoplasmic mRNAs and protein molecules10,17,18,59. This implies that they would be more likely to subtly tune widespread gene expression than to have dramatic impacts. Indeed, both our ribosome profiling and reporter experiments demonstrate that, in addition to translational effects, SGs modestly regulate mRNA levels. Whether this additional example of SGs imparting subtle, global tuning effects on gene expression was indirectly caused by translation-dependent destabilization of mRNAs or illustrates an independent function of SGs will be an interesting question for further investigation.
Consistent with the idea that SGs impart global tuning effects, a recent study investigating the impact of SGs during viral infection reports that SGs play a role in tuning the innate immune response to viral infection57. However, instead of amplifying the ISR translation response, here SGs are reported to dampen the ISR transcriptional response57. This difference in results might be explained by differences in the nature of the stressors or composition of the SGs19,37. Compared with SGs formed by arsenite, heat shock or endoplasmic reticulum stress, the granules formed in response to viral infection tend to be smaller, only weakly recruit PABPC1 and polyadenylated mRNAs, do not require the phosphorylation of eIF2α for their formation and can include RNAse L bodies, which share many of the same markers with SGs and also form in response to viral infection57,75,76. Moreover, the response to viral infection might differ between cells or viruses, as this negative tuning role has been absent in some other reports on viral stress77. Thus, although our results seem likely to be generalizable across canonical SGs that require eIF2α phosphorylation, granules formed under viral infection appear to have distinct effects.
In presenting a model in which SGs reinforce the ISR translation programme, we propose that SGs help to promote the translation of a subset of RNAs. Several observations have led to the hypothesis that SGs play strictly repressive roles in translation. However, none of these observations actually preclude SGs from promoting translation. Although SGs are nucleated by translationally silent mRNAs, implying that the mRNAs localized to them are non-translating, those mRNAs also dynamically exchange with the cytosol and could therefore transit out of the SG after their translation was initiated, maintaining enrichment for translationally silent mRNAs inside the SGs25,29,30,36,78. Indeed, translation initiation itself might even expel the mRNA from the SG, as the presence of even a single ribosome has been reported to prevent SG localization79. Similarly, although SGs are enriched for 40S but not 60S ribosomal subunits, implying that 60S subunits are not stoichiometrically available to assemble translating 80S ribosomes, 60S subunits are not depleted from SGs and appear to be equally available to form translating ribosomes as in the cytosol20,28. Indeed, a recent single-molecule study reports that translation of an ATF4 reporter inside the SG is neither impossible nor rare30. Thus, the prior observations, although originally interpreted as evidence of SGs being incompatible with translation, are also consistent with the possibility that they could be sites of privileged translation for a subset of mRNAs.
Although a model of mRNAs being preferentially translated while inside the SG is perhaps the simplest interpretation of our results, it is not the only interpretation. Alternatively, mRNAs could be licensed for translation while transiting through an SG. In this scenario, pre-initiation complexes or translational activators such as UBAP2L, which are known to be enriched in SGs1,10,20,28,56,80, could preferentially interact with mRNAs transiting through the SG. Then, having acquired this license, the mRNAs could go on to be translated independently of their localization inside or outside of the SG (Fig. 6o). Such a model, in which licensed mRNAs are translated inside or outside the SG, could reconcile the previous observation that mRNAs devoid of ribosomes are preferentially recruited to SGs with our observation that mRNAs that are translationally enhanced during the ISR are enriched in SGs, further enhancing their translation. Such a model could also explain why a single-molecule reporter based on ATF4 mRNA is no more efficiently translated in the SG than in the cytosol30. This being said, we found that ATF4 mRNA was neither enriched in SGs nor influenced by the presence of SGs, indicating that other transcripts might better represent mRNAs influenced by SGs.
The difficulty of distinguishing the function of SGs from the function of the SG-nucleating G3BPs must be acknowledged. One approach to inferring SG function is to consider shared phenotypes of depleting proteins reported to be required for SG formation. However, many of these proteins are thought to also form complexes with each other, independent of SGs, obscuring the interpretation of shared phenotypes19,32,33. Moreover, we and others found that the requirement for reported non-G3BP nucleators appears to be stress or cell-type specific, which limits the utility of depleting these factors.
Although we have not conclusively distinguished between these models, five lines of reasoning support the proposal that the ISR translation programme is reinforced by the formation of SGs rather than only by G3BPs. First, both our ribosome profiling experiments and our tethering experiments demonstrated stress-specific effects. Although G3BP1 tethering enhanced translation in unstressed cells, this might reflect the effect of our 24× MS2/MCP system artificially recruiting many copies of G3BP simultaneously, and thereby simulating the SG environment. Second, although our SG purifications relied on immunoprecipitation (IP) of G3BP, these were IPs on SG-enriched fractions depleted of cytosolic G3BP17,18,58,81. Thus, our data should reflect the SG transcriptome and not simply the G3BP–RNA interactome. Furthermore, we identified similar trends in SG-enrichment data that do not require IP of G3BPs59. Third, the process of forming OGs, which simply manipulates G3BPs condensation, was sufficient to trigger a translation response. Fourth, tethering reporter mRNAs to SGs, whether by G3BP1 or CAPRIN1, was sufficient to impart resistance to stress-induced translation shut down. Although G3BPs and CAPRIN1 are binding partners16, this result indicates that this effect does not require direct recruitment via G3BPs.
Last, in distinguishing between SG- and G3BP-centric models, we should not dismiss the possibility that both G3BPs and SGs contribute to this translation reinforcement. G3BPs have been observed to regulate translation and exhibit exceptionally high partitioning into SGs16,21,22,23,71,73,74. Indeed, we observed almost half of G3BP1 localizing to SGs and that tethering to the protein enhanced translation, even in unstressed cells. If G3BPs license translation, perhaps recruitment of mRNAs to SGs is a molecular strategy to increase their interaction with G3BPs, reinforcing their translation during stress (Fig. 6o). Although these considerations do not conclusively distinguish between SG and G3BP function, they do support the proposal that SGs are a functional condensate responsible for reinforcing the ISR translation programme.
Methods
Cell culture
All cells were cultured at 37 °C with 5% CO2 using McCoy’s 5 A medium supplemented with 10% foetal bovine serum and 2 mM l-glutamine. HCT116 (CCL-247, ATCC) were obtained from the Young laboratory.
Cell line construction
Cas9-mediated editing
Fusion proteins were introduced into the endogenous locus of genes using the Cas9 genome editing system. The 5′ and 3′ homology arms (each 200–700 bp) flanking an insertion site were either amplified from genomic DNA of the parental cell line or from gblock gene fragments (IDT) cloned into a rescue template plasmid, flanking the fusion protein sequence. This plasmid, along with pX330 plasmid expressing a Cas9 guide RNA, was reverse transfected into the parental cell line using Lipofectamine 3000 transfection reagent in a 6-well dish at 100% confluency. After 48 h, the cells of each well were expanded to a 10-cm dish and cultured for an additional 24 h. Cells with a strong signal from the fusion protein fluorescence were single-cell sorted into 96-well plates using flow cytometry. Colonies derived from single cells were grown, expanded and screened by PCR genotyping and western blotting. Unless otherwise stated, all endogenously edited cell lines were edited homozygously.
PiggyBac editing
Exogenous genes were introduced into cells using the PiggyBac transposon system. The exogenous gene was either amplified from genomic DNA of the parental cell line or from gblock gene fragments (IDT) and cloned into a plasmid expressing the PiggyBac inverted-repeat sequences along with a selection marker for either hygromycin, puro or blasticidin. This plasmid, along with the super PiggyBac transposase expression vector (System Biosciences, PB210PA-1), was reverse transfected into the parental cell line using Lipofectamine 3000 transfection reagent (Thermo, L3000015) in a 6-well dish at 100% confluency. After 48 h, cells of each well were expanded to a 10-cm dish, and successfully edited cells were selected using the appropriate antibiotic. For cases in which a fluorescent protein had been transposed into the genome, cells expressing the protein at the appropriate level were single-cell sorted into 96-well plates by flow cytometry. Colonies derived from single cells were grown, expanded and screened by either microscopy, western blotting or both.
Ribo-spike production
In vitro transcription
Ribo-spike mRNA was generated using the mMessage mMachine kit (Thermo, AM1344) following the standard T7 protocol. The DNA transcription template was generated by PCR of a plasmid template encoding the firefly luciferase coding region (fLuc) preceded by the T7 promotor and followed by an encoded poly(A) tail of 30 nt length. The resulting PCR product was purified using phenol–chloroform extraction followed by ethanol precipitation, and was size selected and purified from an agarose gel using the ‘freeze and squeeze’ method, in which the gel slice was frozen at –80 °C for 30 min, followed by centrifugation at 21,000g for 5 min (ref. 82). The eluted DNA was then concentrated by an additional ethanol precipitation and added to the mMessage mMachine in vitro transcription reaction, incubated for at 37 °C for 2 h and then treated with TURBO DNAse for 15 min at 37 °C. RNA from the in vitro transcription reaction was recovered by passing the reaction over a Tris-buffered micro bio-spin p30 gel column (Bio-Rad, 7326223) and centrifuging at 1,000g for 4 min. The resulting flow through was then purified by phenol–chloroform extraction and ethanol precipitation, and then resuspended in water. RNA was examined by running an aliquot on a 4% urea–polyacrylamide denaturing gel and staining with Sybr Gold (Thermo, S11494).
In vitro translation
The fLuc mRNA was in vitro translated using a rabbit reticulocyte lysate system (Promega, L4151) following the standard non-nuclease treated protocol, with the exception of using a shorter incubation time to maximize ribosome occupancy. The reaction was assembled by combining 0.06 µM fLuc mRNA with reticulocyte lysate, 0.02 mM complete amino acid mixture, 0.8 units µl−1 RNAsin ribonuclease inhibitor, 0.01 M creatine phosphate, 0.05 mg ml−1 creatine phosphokinase, 2 mM dithiothreitol (DTT), 0.05 mg ml−1 tRNA, 80 mM potassium acetate and 0.5 mM magnesium acetate in 23 117 µl reactions. The reactions were incubated at 30 °C for 15 min, cooled on ice and treated with 0.1 mg ml−1 cycloheximide to block ribosome translocation. Reactions were then combined, mixed, aliquoted, snap frozen in liquid nitrogen and stored at –80 °C.
IAA-induced protein depletion
HCT116 cells were engineered using the PiggyBac system to express multiple copies of doxycycline-inducible OsTIR1. These cells were then also engineered using Cas9 to expressed AID-tagged fusion proteins edited at their endogenous loci. Cells expressing fusion proteins were first doxycycline-induced for 4 h and were then treated with 500 µM IAA (Gold Bio, I-110-25) to induce rapid (<3 h) depletion of the AID fusion protein. Depletion was confirmed by microscopy, western blot or both. When confirming by western blot, as was the case with G3BP1, G3BP2 and CAPRIN1 proteins, antibodies raised directly against the target proteins were used to enable detection of untagged alleles, if present. Where indicated, non-depleted controls were treated with 200 µM auxinole (Aobious, AOB8812) to minimize background protein degradation83. Unless otherwise stated, all other non-depleted control cells were treated only with ethanol, the solvent used for IAA. Previous work has shown that the addition of IAA alone does not affect translation84.
Stress conditions
For oxidative stress, unless otherwise stated, cells were treated with media containing 500 µM sodium arsenite (Sigma, 93289) for 1 h. For heat shock, media was separately heated to 45 °C. Media were removed from cells previously grown in a 37 °C, rapidly replaced with 45 °C media and then cells were quickly placed in a 45 °C incubator for 25 min.
Doxycycline induction
For all doxycycline inductions, cells were treated with 1 µg ml−1 doxycycline (Clonetech Takara Bio, 631311) for the indicated amount of time.
Tethered reporter TE experiments
To measure changes in TE for the nanoluciferase reporters (NanoLuc), cells expressing a reporter containing selected 5′ and 3′ UTRs, NanoLuc fused to an ecDHFR domain and 24× MS2 stem loops, were washed with cold PBS, lysed directly in their dish in buffer containing 10 mM Tris HCl pH 7.5, 5 mM MgCl2, 100 mM KCl, 1% Triton, 1× cOmplete mini tablet (EDTA free) and 0.02 U µl−1 SUPERase-In RNase inhibitor, and depleted of nuclei by centrifuging at 1,300g for 10 min at 4 °C. Nuclear-depleted supernatant was then transferred to a new tube and flash frozen using liquid nitrogen and stored at –80 °C. Cells were then assayed for total protein using bicinchoninic acid, luciferase production and reporter mRNA using RT–qPCR by comparison to GAPDH
mRNA, which did not change in abundance during the treatment with sodium arsenite (Extended Data Fig. 8c). TE was then calculated according to the following equation:
In G3BP-tethering experiments, the reporter was integrated into in two different HCT116-derived cell lines: a line expressing endogenous G3BP1 tagged with GFP (GFP::G3BP1) and a line expressing endogenous G3BP1 tagged with MCP fused to GFP (MCP::GFP::G3BP1) (Fig. 6a). In CAPRIN1-tethering experiments, the reporter was expressed in an HCT116-derived cell line under three different conditions: untethered, in which the cell line was untreated and expressed endogenous G3BPs tagged with AID and RFP (AID::RFP::G3BPs); tethered (+SGs), in which the cell line was treated with 1 µM doxycycline and 200 µM auxinole for 10 h, leading to retention of G3BPs and expression of MCP::GFP::CAPRIN1; and tethered (–SGs), in which the cell line was treated with 1 µM doxycycline and 500 µM IAA for 10 h, leading to turnover of endogenous G3BPs, which prevented SG formation, and expression of MCP::GFP::CAPRIN1 (Fig. 6e).
Luciferase assays
Luciferase assays were conducted using the standard protocol of the Luciferase Reporter Assay System (Promega, E1910). Cell lysates were analysed in a 96-well microplate format at room temperature using a GloMax microplate reader and luminometer device (Promega, GM3000). Each sample was run in technical duplicate.
RT–qPCR
RNA was extracted using TRI reagent (Sigma, 93289), precipitated using ethanol and linear acrylamide (Thermo, J67830-XF) and resuspended in water. Reverse transcription (RT) reactions were performed using a Quantitect RT kit following kit protocols. Genomic DNA was first eliminated using 7× gDNA wipeout buffer and incubating at 42 °C for 2–5 min. Reactions were then placed on ice, RT reagents were added and then RT was performed at 42 °C for 30 min. The RT enzyme was heat inactivated by incubating at 95 °C for 3 min. The cDNA product was then diluted 1:2 in water and stored at –20 °C. qPCR was then performed using the PowerUP SYBR Green qPCR master mix with primers targeting either the NanoLuc or GAPDH sequences. Each sample was run in technical triplicate PCR using a QuantStudio 6 Pro RT–PCR System (Thermo).
Blue-light LED treatment
Cells were exposed to blue light for indicated amounts of time using a 12 inch square light-emitting diode (LED) Grow Light System with 225× 14 W blue LEDs (HQRP, 884667106091218) mounted inside of their cell culture incubator.
FRAP
Cells were bleached using a blue light (488 nm) laser, followed by green channel imaging every second for 3 min. Imaging was performed using an Andor Revolution Spinning Disk Confocal with FRAPPA system. Fluorescence recovery after photobleaching (FRAP) analysis was performed using Fiji (ImageJ).
Immunofluoresence and live imaging
Cells were grown in plastic Nunc Cell Culture Treated Multidishes (Thermo, 140675). For live imaging, cells were aspirated of their cell growth media and replaced with PBS. For immunofluorescence, cells were fixed using 3.7% formaldehyde in PBS at room temperature for 15 min, washed with PBS for 5 min three times, blocked using normal goat serum diluted in PBS with 0.3% Triton for at least 1 h, then treated with primary antibody diluted in 1% normal goat serum, 0.3% Triton and PBS. Primary antibody treatments were performed overnight at 4 °C. Cells were then washed in PBS for 5 min three times and treated with secondary antibody using the same dilution buffer for 1–2 h at room temperature. Cells were washed a final time with PBS and mounted using mounting media containing DAPI (Vectashield, H-2000-10). Imaging was performed on a Nikon Ti automated inverted microscope with incubation enclosure. Images were analysed using Fiji (ImageJ).
smFISH
For smFISH, cells were plated in glass-bottomed, 10-mm, 24-well plates (MatTek Corporation, P24G-1.5-10-F). Cells were prepared following the standard Stellaris RNA FISH protocol for adherent cells (Biosearch Technologies). Cells were fixed using 3.7% formaldehyde in PBS at room temperature for 10 min. Cells were then permeabilized using 70% ethanol for at least 1 h at 4 °C. Cells were washed with Wash Buffer A and incubated at room temperature for 5 min. Wash Buffer A was aspirated and cells were treated with approximately 30 µl of 125 nM FISH probe diluted in hybridization buffer. The plate was sealed with Parafilm and stored in a dark humid chamber overnight at 37 °C. Cells were then washed with Wash Buffer A for 30 min at 37 °C, then washed with Wash Buffer B for 5 min and mounted with mounting media containing DAPI. smFISH imaging was performed using a Zeiss LSM 980 with Airyscan 2 Laser Scanning Confocal imaging system with a Zeiss AxioObserver motorized inverted microscope stand, DIC optics and a motorized XYZ stage. Images were analysed using IMARIS.
Western blots
Samples used for western blots were lysed by boiling in NuPAGE LDS Sample Buffer (Thermo, NP0007) diluted to a 1× concentration and supplemented with 1 mM DTT. Samples were run on NuPAGE 4–12% Bis–Tris 1.0 mm miniprotein gels (Thermo, NP0321BOX), then transferred to 0.2 µM PVDF transfer membranes (Thermo, 22860). All protein gels and transfers were run using a mini gel tank (Thermo, A25977) with protein gels run at 200 V for 80 min and transfers run at 30 V for 60 min. Transferred membranes were blocked in PBS with 1% Triton and 5% milk for at least 1 h at room temperature before they were transferred to primary antibody diluted in the same buffer at 4 °C. Primary antibodies were applied overnight followed by at least three 5 min washes with the same blocking buffer. Secondary antibodies were diluted in the same buffer for 30 min at room temperature, and three 5 min washes in the same buffer. Western blots were imaged using an Odyssey CLx imaging system by LI-COR. All western blot quantifications were done using Fiji/ImageJ software.
Polysome profiling
HCT116 cells were cultured in a 15-cm dish, washed twice with cold PBS, scraped into lysis buffer containing 10 mM Tris HCl pH 7.5, 5 mM MgCl2, 100 mM KCl, 1% Triton, 2 mM DTT, 1× cOmplete mini tablet (EDTA free) and 0.3 U µl−1 RNasin Plus RNase inhibitor. Cells were lysed by passing through a 26 G 0.5-inch needle at least seven times. Lysates were depleted of nuclei by centrifugation at 1,300g for 10 min at 4 °C, and snap frozen using liquid nitrogen and stored at –80 °C until use. Samples were loaded onto a 10–50% sucrose gradient and centrifuged at 36,000 rpm for 2 h at 4 °C. Polysome profiling was then performed using a BioComp Gradient Fractionator. Twelve fractions were collected across approximately 90% of the volume of the sucrose gradient, with approximately 800 µl collected per fraction. Each fraction was spiked with 0.1 fmol of an in vitro transcribed RNA standard and mixed thoroughly. Samples were then snap frozen using liquid nitrogen and stored at –80 °C until later use. RNA extraction and RT–qPCR were then performed on each fraction as described above. Total ribosome loads reported in Fig. 6k,n were calculated by multiplying the proportion of reporter RNA from the time course by the mean estimated number of ribosomes found on transcripts in each polysome fraction and then summing those values across all fractions.
Ribosome profiling and matched RNA sequencing
HCT116 cells were cultured in a 15-cm dish, washed twice with cold PBS, scraped into lysis buffer containing 10 mM Tris HCl pH 7.5, 5 mM MgCl2, 100 mM KCl, 1% Triton, 2 mM DTT, 1× cOmplete mini tablet (EDTA free), 0.3 U µl−1 RNasin Plus RNase inhibitor and 1:60 dilution of ribo-spike. Cells were lysed by passing through 26 G 0.5-inch needle at least seven times. Lysates were depleted of nuclei by centrifugation at 1,300g for 10 min at 4 °C, divided into aliquots for ribosome profiling and matched RNA libraries and snap frozen using liquid nitrogen. Ribosome profiling and matched RNA sequencing libraries were then prepared according to previously established protocol85, a detailed version of which is available at http://bartellab.wi.mit.edu/protocols.html. Sequencing was performed on an Illumina HiSeq 2500 using 50 cycles. Only reads mapping to ORFs of GRCh38-annotated genes, fluorescent protein-coding genes, the OsTIR1 gene or the fLuc gene were used. Reads mapping to the first 50 nt of each ORF were excluded. Reads mapping to heme-related genes from the rabbit genome (Broad/oryCun2) were also excluded. TE changes were calculated and count depth normalization was performed using DESeq286.
Isolation of RNA from HCT116 cells and SG cores for RNA sequencing
The Isolation and sequencing of SG cores was adapted from an established protocol18,58. HCT116 cells were cultured in a 24.5-cm square dish, washed with PBS, scraped into PBS, centrifuged at 1,500g for 3 min, aspirated to remove the liquid and snap frozen in liquid nitrogen. Frozen cell pellets were thawed in lysis buffer (50 mM Tris HCl pH 7.4, 2 mM MgOAc, 10 mM MgCl2, 100 mM KOAc, 0.1% NP40, 0.5 mM DTT, 1× cOmplete mini tablet (EDTA free) and 1 U µl−1 RNasin Plus RNase inhibitor). Cells were lysed by passing through a 26 G 0.5-inch needle at least seven times. Lysates were depleted of their nuclei by centrifugation at 1,000g for 5 min at 4 °C. Then, 5% of this nuclear-depleted sample was aliquoted and snap frozen in liquid nitrogen and stored at –80 °C as the cytoplasmic fraction. The remaining 95% was used to isolate the SG fraction. Samples were centrifuged at 18,000g for 20 min at 4 °C. Pellets were resuspended in 1 ml lysis buffer and allowed to turn end-over-end for at least 10 min at 4 °C. Once resuspended, samples were centrifuged again at 18,000g for 20 min at 4 °C. The pellet was resuspended in 300 µl lysis buffer and centrifuged at 850g for 2 min at 4 °C. The supernatant was taken and incubated with 6 µg of G3BP2 antibody (Bethyl Laboratories, A302-040A) overnight at 4 °C, rotating end-over-end. Samples were centrifuged at 18,000g for 20 min at 4 °C to remove unbound antibody, and pellets were resuspended in 500 µl lysis buffer. Then, 625 µl of M-280 sheep, anti-rabbit IgG dynabeads (Thermo, 11204D) were added to the sample and incubated for at least 3 h at 4 °C, rotating end-over-end. Beads were washed with buffer containing 20 mM Tris HCl pH 8.0 and 50 mM NaCl and eluted using a high-salt buffer (10 mM Tris HCl pH 7.5, 1 M NaCl, 5% 1,6 hexanediol, 5 mM MgCl2, 100 mM KCl, 1% Triton, 2 mM DTT, 1× cOmplete mini tablet (EDTA free) and 0.3 U µl−1 RNasin Plus RNase inhibitor), turning end-over-end for at least 4 h. The eluate was snap frozen using liquid nitrogen and stored at –80 °C as the SG fraction. RNA-sequencing libraries were then prepared from the cytoplasmic and SG fractions in parallel, following the protocol used for the RNA-sequencing libraries that matched the ribosome profiling. SG enrichment values were calculated using DESeq286.
Statistical analysis and comparison with published datasets
Plots and statistical analyses were performed using R. No statistical methods were used to predetermine sample sizes but our sample sizes are similar to those reported in previous publications18,55,59. Statistical tests and parameters are shown in figure legends. Data distribution was assumed to be normal but this was not formally tested. Data collection and analysis were not performed blind to the conditions of the experiments. Additional data were obtained from publicly available datasets associated with published work and re-analysed using R18,42,55,59,60.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
Raw sequencing data for all ribo-seq and SG purification experiments of this study have been deposited in the Gene Expression Omnibus under accession code GSE256237. Previously published sequencing data were re-analysed using R and are available under accession codes GSE99304, GSE131650, GSE254636, GSE254637, GSE254638, GSE90869, GSE103667 and GSE223295 (refs. 18,42,55,59,60). All other data supporting the findings of this study are available from the corresponding author on reasonable request. Source data are provided with this paper.
References
Buchan, J. R. & Parker, R. Eukaryotic stress granules: the ins and outs of translation. Mol. Cell 36, 932–941 (2009).
Advani, V. M. & Ivanov, P. Translational control under stress: reshaping the translatome. Bioessays 41, e1900009 (2019).
Pakos-Zebrucka, K. et al. The integrated stress response. EMBO Rep. 17, 1374–1395 (2016).
Harding, H. P. et al. Regulated translation initiation controls stress-induced gene expression in mammalian cells. Mol. Cell 6, 1099–1108 (2000).
Harding, H. P., Zhang, Y. & Ron, D. Protein translation and folding are coupled by an endoplasmic-reticulum-resident kinase. Nature 397, 271–274 (1999).
Yamasaki, S. & Anderson, P. Reprogramming mRNA translation during stress. Curr. Opin. Cell Biol. 20, 222–226 (2008).
Starck, S. R. et al. Translation from the 5′ untranslated region shapes the integrated stress response. Science 351, aad3867 (2016).
Sidrauski, C., McGeachy, A. M., Ingolia, N. T. & Walter, P. The small molecule ISRIB reverses the effects of eIF2alpha phosphorylation on translation and stress granule assembly. eLife 4, e05033 (2015).
Vattem, K. M. & Wek, R. C. Reinitiation involving upstream ORFs regulates ATF4 mRNA translation in mammalian cells. Proc. Natl Acad. Sci. USA 101, 11269–11274 (2004).
Protter, D. S. W. & Parker, R. Principles and properties of stress granules. Trends Cell Biol. 26, 668–679 (2016).
Kedersha, N. L., Gupta, M., Li, W., Miller, I. & Anderson, P. RNA-binding proteins TIA-1 and TIAR link the phosphorylation of eIF-2 to the assembly of mammalian stress granules. J. Cell Biol. 147, 1431–1441 (1999).
Yang, P. et al. G3BP1 is a tunable switch that triggers phase separation to assemble stress granules. Cell 181, 325–345 (2020).
Guillen-Boixet, J. et al. RNA-induced conformational switching and clustering of G3BP drive stress granule assembly by condensation. Cell 181, 346–361 (2020).
Sanders, D. W. et al. Competing protein–RNA interaction networks control multiphase intracellular organization. Cell 181, 306–324 (2020).
Molliex, A. et al. Phase separation by low complexity domains promotes stress granule assembly and drives pathological fibrillization. Cell 163, 123–133 (2015).
Kedersha, N. et al. G3BP–Caprin1–USP10 complexes mediate stress granule condensation and associate with 40S subunits. J. Cell Biol. 212, 845–860 (2016).
Jain, S. et al. ATPase-modulated stress granules contain a diverse proteome and substructure. Cell 164, 487–498 (2016).
Khong, A. et al. The stress granule transcriptome reveals principles of mRNA accumulation in stress granules. Mol. Cell 68, 808–820 (2017).
Markmiller, S. et al. Context-dependent and disease-specific diversity in protein interactions within stress granules. Cell 172, 590–604 (2018).
Kedersha, N. et al. Evidence that ternary complex (eIF2–GTP–tRNAiMet)-deficient preinitiation complexes are core constituents of mammalian stress granules. Mol. Biol. Cell 13, 195–210 (2002).
Laver, J. D. et al. The RNA-binding protein Rasputin/G3BP enhances the stability and translation of its target mRNAs. Cell Rep. 30, 3353–3367 (2020).
Alam, U. & Kennedy, D. Rasputin a decade on and more promiscuous than ever? A review of G3BPs. Biochim. Biophys. Acta Mol. Cell Res. 1866, 360–370 (2019).
Meyer, C., Garzia, A., Morozov, P., Molina, H. & Tuschl, T. The G3BP1-family-USP10 deubiquitinase complex rescues ubiquitinated 40S subunits of ribosomes stalled in translation from lysosomal degradation. Mol. Cell 77, 1193–1205 (2020).
Anderson, P. & Kedersha, N. RNA granules: post-transcriptional and epigenetic modulators of gene expression. Nat. Rev. Mol. Cell Biol. 10, 430–436 (2009).
Kedersha, N. et al. Dynamic shuttling of TIA-1 accompanies the recruitment of mRNA to mammalian stress granules. J. Cell Biol. 151, 1257–1268 (2000).
Kwon, S., Zhang, Y. & Matthias, P. The deacetylase HDAC6 is a novel critical component of stress granules involved in the stress response. Genes Dev. 21, 3381–3394 (2007).
Youn, J. Y. et al. Properties of stress granule and P-body proteomes. Mol. Cell 76, 286–294 (2019).
Kimball, S. R., Horetsky, R. L., Ron, D., Jefferson, L. S. & Harding, H. P. Mammalian stress granules represent sites of accumulation of stalled translation initiation complexes. Am. J. Physiol. Cell Physiol. 284, C273–C284 (2003).
Moon, S. L. et al. Multicolour single-molecule tracking of mRNA interactions with RNP granules. Nat. Cell Biol. 21, 162–168 (2019).
Mateju, D. et al. Single-molecule imaging reveals translation of mRNAs localized to stress granules. Cell 183, 1801–1812 (2020).
Baymiller, M. & Moon, S. L. Stress granules as causes and consequences of translation suppression. Antioxid. Redox Signal. 39, 390–409 (2023).
Putnam, A., Thomas, L. & Seydoux, G. RNA granules: functional compartments or incidental condensates?. Genes Dev. 37, 354–376 (2023).
Mateju, D. & Chao, J. A. Stress granules: regulators or by-products? FEBS J. 289, 363–373 (2022).
Ingolia, N. T. Ribosome footprint profiling of translation throughout the genome. Cell 165, 22–33 (2016).
Iwasaki, S., Floor, S. N. & Ingolia, N. T. Rocaglates convert DEAD-box protein eIF4A into a sequence-selective translational repressor. Nature 534, 558–561 (2016).
Wilbertz, J. H. et al. Single-molecule imaging of mRNA localization and regulation during the integrated stress response. Mol. Cell 73, 946–958 (2019).
Aulas, A. et al. Stress-specific differences in assembly and composition of stress granules and related foci. J. Cell Sci. 130, 927–937 (2017).
Sonenberg, N. & Hinnebusch, A. G. Regulation of translation initiation in eukaryotes: mechanisms and biological targets. Cell 136, 731–745 (2009).
Holcik, M. & Sonenberg, N. Translational control in stress and apoptosis. Nat. Rev. Mol. Cell Biol. 6, 318–327 (2005).
Andreev, D. E. et al. Translation of 5′ leaders is pervasive in genes resistant to eIF2 repression. eLife 4, e03971 (2015).
Tomuro, K. et al. Calibrated ribosome profiling assesses the dynamics of ribosomal flux on transcripts. Nat. Commun. 15, 7061 (2024).
Chen, J., Brunner, A.-D., Nunez, J. K. & Weissman, J. Pervasive functional translation of noncanonical human open reading frames. Science 367, 1135–1140 (2020).
Glauninger, H. et al. Transcriptome-wide mRNA condensation precedes stress granule formation and excludes new mRNAs. Mol. Cell 85, 4393–4409.e11 (2025).
Zedan, M. et al. Timing of transcription controls the selective translation of newly synthesized mRNAs during acute environmental stress. Mol. Cell 85, 4379–4392.e5 (2025).
Agarwal, V. & Kelley, D. R. The genetic and biochemical determinants of mRNA degradation rates in mammals. Genome Biol. 23, 245 (2022).
Pizzinga, M. et al. The cell stress response: extreme times call for post-transcriptional measures. Wiley Interdiscip. Rev. RNA 11, e1578 (2020).
Natsume, T., Kiyomitsu, T., Saga, Y. & Kanemaki, M. T. Rapid protein depletion in human cells by auxin-inducible degron tagging with short homology donors. Cell Rep. 15, 210–218 (2016).
Nishimura, K., Fukagawa, T., Takisawa, H., Kakimoto, T. & Kanemaki, M. An auxin-based degron system for the rapid depletion of proteins in nonplant cells. Nat. Methods 6, 917–922 (2009).
Dave, P. et al. Single-molecule imaging reveals translation-dependent destabilization of mRNAs. Mol. Cell 83, 589–606 (2023).
Tuck, A. C. et al. Mammalian RNA decay pathways are highly specialized and widely linked to translation. Mol. Cell 77, 1222–1236 (2020).
Laird-Offringa, I. A., de Wit, C. L., Elfferich, P. & van der Eb, A. J. Poly(A) tail shortening is the translation-dependent step in c-myc mRNA degradation. Mol. Cell. Biol. 10, 6132–6140 (1990).
Bicknell, A. A. et al. Attenuating ribosome load improves protein output from mRNA by limiting translation-dependent mRNA decay. Cell Rep. 43, 114098 (2024).
Horvathova, I. et al. The dynamics of mRNA turnover revealed by single-molecule imaging in single cells. Mol. Cell 68, 615–625 (2017).
Rosa-Mercado, N. A., Buskirk, A. R. & Green, R. Translation elongation inhibitors stabilize select short-lived transcripts. RNA 30, 1572–1585 (2024).
Liboy-Lugo, J. M. et al. G3BP isoforms differentially affect stress granule assembly and gene expression during cellular stress. Mol. Biol. Cell 35, ar140 (2024).
Cirillo, L. et al. UBAP2L forms distinct cores that act in nucleating stress granules upstream of G3BP1. Curr. Biol. 30, 698–707 (2020).
Paget, M. et al. Stress granules are shock absorbers that prevent excessive innate immune responses to dsRNA. Mol. Cell 83, 1180–1196 (2023).
Khong, A., Jain, S., Matheny, T., Wheeler, J. R. & Parker, R. Isolation of mammalian stress granule cores for RNA-seq analysis. Methods 137, 49–54 (2018).
Namkoong, S., Ho, A., Woo, Y. M., Kwak, H. & Lee, J. H. Systematic characterization of stress-induced RNA granulation. Mol. Cell 70, 175–187 (2018).
Ren, Z., Tang, W., Peng, L. & Zou, P. Profiling stress-triggered RNA condensation with photocatalytic proximity labeling. Nat. Commun. 14, 7390 (2023).
Huang, C. et al. UBAP2L arginine methylation by PRMT1 modulates stress granule assembly. Cell Death Differ. 27, 227–241 (2020).
Tourriere, H. et al. The RasGAP-associated endoribonuclease G3BP assembles stress granules. J. Cell Biol. 160, 823–831 (2003).
Zhang, P. et al. Chronic optogenetic induction of stress granules is cytotoxic and reveals the evolution of ALS-FTD pathology. eLife 8, e39578 (2019).
Panas, M. D., Ivanov, P. & Anderson, P. Mechanistic insights into mammalian stress granule dynamics. J. Cell Biol. 215, 313–323 (2016).
Ivanov, P., Kedersha, N. & Anderson, P. Stress granules and processing bodies in translational control. Cold Spring Harb. Perspect. Biol. https://doi.org/10.1101/cshperspect.a032813 (2019).
Matheny, T., Rao, B. S. & Parker, R. Transcriptome-wide comparison of stress granules and p-bodies reveals that translation plays a major role in RNA partitioning. Mol. Cell Biol. https://doi.org/10.1128/MCB.00313-19 (2019).
Lee, C. S. et al. Recruitment of mRNAs to P granules by condensation with intrinsically-disordered proteins. eLife 9, e52896 (2020).
Ramadurgum, P. & Hulleman, J. D. Protocol for designing small-molecule-regulated destabilizing domains for in vitro use. STAR Protoc. 1, 100069 (2020).
Bertrand, E. et al. Localization of ASH1 mRNA particles in living yeast. Mol. Cell 2, 437–445 (1998).
Wheeler, J. R., Jain, S., Khong, A. & Parker, R. Isolation of yeast and mammalian stress granule cores. Methods 126, 12–17 (2017).
Matheny, T., Van Treeck, B., Huynh, T. N. & Parker, R. RNA partitioning into stress granules is based on the summation of multiple interactions. RNA 27, 174–189 (2021).
Khong, A., Matheny, T., Huynh, T. N., Babl, V. & Parker, R. Limited effects of m6A modification on mRNA partitioning into stress granules. Nat. Commun. 13, 3735 (2022).
Bidet, K., Dadlani, D. & Garcia-Blanco, M. A. G3BP1, G3BP2 and CAPRIN1 are required for translation of interferon stimulated mRNAs and are targeted by a dengue virus non-coding RNA. PLoS Pathog. 10, e1004242 (2014).
Ortega, A. D., Willers, I. M., Sala, S. & Cuezva, J. M. Human G3BP1 interacts with β-F1-ATPase mRNA and inhibits its translation. J. Cell Sci. 123, 2685–2696 (2010).
Burke, J. M., Moon, S. L., Matheny, T. & Parker, R. RNase L reprograms translation by widespread mRNA turnover escaped by antiviral mRNAs. Mol. Cell 75, 1203–1217 (2019).
Burke, J. M., Lester, E. T., Tauber, D. & Parker, R. RNase L promotes the formation of unique ribonucleoprotein granules distinct from stress granules. J. Biol. Chem. 295, 1426–1438 (2020).
Burke, J. M., Ratnayake, O. C., Watkins, J. M., Perera, R. & Parker, R. G3BP1-dependent condensation of translationally inactive viral RNAs antagonizes infection. Sci. Adv. 10, eadk8152 (2024).
Buchan, J. R., Muhlrad, D. & Parker, R. P bodies promote stress granule assembly in Saccharomyces cerevisiae. J. Cell Biol. 183, 441–455 (2008).
Fedorovskiy, A. G. et al. A solitary stalled 80S ribosome prevents mRNA recruitment to stress granules. Biochem. (Mosc.) 88, 1786–1799 (2023).
Luo, E. C. et al. Large-scale tethered function assays identify factors that regulate mRNA stability and translation. Nat. Struct. Mol. Biol. 27, 989–1000 (2020).
Wheeler, J. R., Matheny, T., Jain, S., Abrisch, R. & Parker, R. Distinct stages in stress granule assembly and disassembly. eLife 5, e18413 (2016).
Downey, N. Extraction of DNA from agarose gels. Methods Mol. Biol. 235, 137–139 (2003).
Yesbolatova, A., Natsume, T., Hayashi, K. I. & Kanemaki, M. T. Generation of conditional auxin-inducible degron (AID) cells and tight control of degron-fused proteins using the degradation inhibitor auxinole. Methods 164–165, 73–80 (2019).
Xiang, K. & Bartel, D. P. The molecular basis of coupling between poly(A)-tail length and translational efficiency. eLife 10, e66493 (2021).
Subtelny, A. O., Eichhorn, S. W., Chen, G. R., Sive, H. & Bartel, D. P. Poly(A)-tail profiling reveals an embryonic switch in translational control. Nature 508, 66–71 (2014).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Acknowledgements
We thank members of the Bartel laboratory for helpful discussions, R. Young at the Whitehead Institute for providing HCT116 cells, the Whitehead Genome Technology Core for high-throughput sequencing and the Whitehead Flow Cytometry Core for cell sorting. J.S. is a Hanna Gray Fellow of the Howard Hughes Medical Institute and D.B. is an investigator in the Howard Hughes Medical Institute.
Author information
Authors and Affiliations
Contributions
J.S. conceived the project and designed the experiments. J.S. performed the experiments and analyses. J.S. and D.P.B. designed the figures and wrote the paper. The paper was read and approved by all authors.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cell Biology thanks the anonymous reviewers for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Heat shock and oxidative stress illicit similar but distinct ISR translation programs.
(A) Quantification of ribo-spike. To confirm that ribo-spikes functioned as intended, cytoplasmic extract from HCT116 cells was serially diluted to achieve the indicated relative concentrations, and then a constant amount of ribo-spike was added to each sample before ribosome profiling and RNA-seq analyses. Plotted are the relative concentrations of total mRNA (dashed line) and RPFs (solid line) as calculated from the 1000 most highly expressed genes, using no-spike controls (blue) and ribo-spike normalized samples (red) as a function of the actual relative concentrations. Spike-normalized RPF and mRNA concentrations closely matched the known dilutions. (B) Relative TE measurements before adjusting for ribo-spike. Plotted with circles are the average log2 TE values (line, median; notch, 95% confidence interval; box, quartiles; whiskers, 1.5 x IQR) calculated for mRNAs from either untreated cells (black, three replicates), cells treated with 500 µM sodium arsenite for 1 h (green, four replicates), or cells treated with heat shock at 45 °C for 25 min (pink, 2 replicates). Red Xs denote the ribo-spike TE values (one for each replicate). Statistical significance was calculated based on ribo-spike values alone (***, p \(\le \,\)0.001; Welch’s two-sample t-test). (C) TE measurements of panel B after adjusting for ribo-spike; otherwise, as in (B). Note that statistical significance is determined in panel B, using ribo-spike values prior to adjustment (***, p \(\le \,\)0.001). (D) Preferential translation of mRNAs containing uORFs during arsenite stress. The plots indicate log2 FC in TE caused by arsenite for mRNAs containing at least one uORF and those containing no uORFs, according to annotations from Chen et al.42 (line, median; notch, 95% confidence interval; box, quartiles; whiskers, 1.5 x IQR); ***, p \(\le \,\)0.001; Welch’s two-sample t-test). (E) Relationship between translation during arsenite stress and mRNA half-lives. The plots indicate the log2 FC in TE due to arsenite as a function of the mean-normalized half-life (PC1) of mRNAs as calculated from consensus of multiple human cell data sets by Agarwal et al.45 n indicates number of unique mRNAs. (F) Preferential translation of mRNAs containing uORFs during heat shock. Plots indicate log2 FC in TE caused by treatment with 45 °C heat shock; otherwise, as in (D). (G) Relationship between translation during heat shock and mRNA half-lives. The plots indicate the log2 FC in TE due to heat shock as a function of the mean-normalized half-life of mRNAs; otherwise, as in (E). (H – J) Comparison of arsenite stress and heat shock ISR translation programs. Plotted for each mRNA are changes in TE caused by arsenite as a function of changes in TE caused by heat shock. Points for mRNAs classified as either stress-enhanced, stress-resistant, or stress-hypersensitive mRNAs in either arsenite stress (D), heat shock (E), or both (F) are coloured. n indicates number of unique mRNAs; Rs indicates Spearman coefficient.
Extended Data Fig. 2 G3BPs reinforce the ISR translation program across multiple cell types and stresses.
(A) The ISR translation response to arsenite stress, either with or without G3BPs. The plots indicate the log2 FC in TE caused by arsenite stress in either untreated HCT116 cells or cells depleted of their G3BPs by addition of 500 µM IAA (line, median; notch, 95% confidence interval; box, quartiles; whiskers, 1.5 x IQR). Significance was determined using ribo-spike values, as in Extended Data Fig. 1B (* p \(\le\) 0.05, ***, p \(\le \,\)0.001; Welch’s two-sample t-test). (B) Comparison of G3BP-dependent translation and ISR translation in HCT116 cells treated with arsenite stress. The plots indicate the log2 FC in TE due to presence of G3BPs in stressed cells in each of three ISR translation categories: stress-enhanced/resistant, bulk behaviour, and stress-hypersensitive (line, median; notch, 95% confidence interval; box, quartiles; whiskers, 1.5 x IQR; ***, p \(\le \,\)0.001; Welch’s two-sample t-test). (C) Comparison of G3BP-dependent RPF changes in HCT116 cells treated with arsenite stress. The plots indicate the fold change in RPFs due to presence of G3BPs in stressed cells; otherwise, as in (B). (D) Comparison of G3BP-dependent RNA changes in HCT116 cells undergoing arsenite stress. The plots indicate the fold change in RNA due to presence of G3BPs in stressed cells; otherwise, as in (B). (E) Comparison of stress-dependent RNA changes in HCT116 cells undergoing arsenite stress. The plots indicate the fold change in RNA due to treatment with 500 µM sodium arsenite for 1 h; otherwise, as in (B). (F) The ISR translation response to heat shock, either with or without G3BPs. ns, not significant; otherwise, as in (A). (G) Comparison of G3BP-dependent translation and ISR translation in HCT116 cells undergoing heat shock stress; otherwise, as in (B). (H) Comparison of G3BP-dependent RPF changes in HCT116 cells undergoing heat shock stress. The plots indicate the fold change in RPFs due to presence of G3BPs in stressed cells; otherwise, as in (B). (I) Comparison of G3BP-dependent RNA changes in HCT116 cells undergoing heat-shock stress. The plots indicate the fold change in RNA due to presence of G3BPs in stressed cells; otherwise, as in (B). (J) Comparison of stress-dependent RNA changes in HCT116 cells undergoing heat-shock stress. The plots indicate the fold change in RNA due to treatment with heat-shock stress; otherwise, as in (B).
Extended Data Fig. 3 G3BPs reinforce the ISR translation program across multiple cell types and stresses.
(A) Relationship between G3BP-dependent translation during thapsigargin stress and the ISR translation program as measured by Liboy-Lugo et al.55; otherwise as in Fig. 2h. (B) Relationship between G3BP-dependent translation in the absence of stress and the ISR translation program as measured by Liboy-Lugo et al.55; otherwise as in Fig. 2I. (C) Comparison of G3BP-dependent translation and ISR translation in U2OS cells undergoing thapsigargin stress as measured by Liboy-Lugo et al.55 otherwise as in (Extended Data Fig. 2B). (D) Comparison of G3BP-dependent RPF changes in U2OS cells undergoing thapsigargin stress as measured by Liboy-Lugo et al.55; otherwise as in (Extended Data Fig. 2C). (E) Comparison of G3BP-dependent RNA changes in U2OS cells undergoing thapsigargin stress as measured by Liboy-Lugo et al.55; otherwise as in (Extended Data Fig. 2D).
Extended Data Fig. 4 Neither CAPRIN1 nor UBAP2L are required for SG formation in HCT116 cells.
(A) G3BP-dependent translation of mRNAs containing uORFs during arsenite stress. The plots indicate the log2 FC in TE due to presence of G3BPs in arsenite-stressed cells for mRNAs containing at least one uORF and those containing no uORFs, according to annotations from Chen et al.42 (line, median; notch, 95% confidence interval; box, quartiles; whiskers, 1.5 x IQR); ***, p \(\le \,\)0.001; Welch’s two-sample t-test). (B) G3BP-dependent translation of mRNAs containing uORFs during heat stress. The plots indicate the log2 FC in TE due to presence of G3BPs in heat-stressed cells for mRNAs containing at least one uORF and those containing no uORFs. We observed that mRNAs containing one or more functional uORF were preferentially translated in the presence of heat stress. However, as this trend did not hold in arsenite-stressed cells, suggesting that uORFs might play only a minor or a stress-specific role in G3BP-dependent translation; otherwise as in (A). (C) Relationship between G3BP-dependent translation during arsenite stress and mRNA half-lives. We observed no correlation between the half-life of an mRNA and its G3BP-dependent translation, indicating that preferential translation of newly synthesized mRNAs might be stress-granule independent. The plots indicate the log2 FC in TE due to presence of G3BPs in arsenite-stressed cells as a function of the mean-normalized half-life (PC1) of mRNAs as calculated from consensus of multiple human cell data sets by Agarwal et al.45 n indicates number of unique mRNAs. (D) Relationship between G3BP-dependent translation during heat stress and mRNA half-lives. The plots indicate the log2 FC in TE due to presence of G3BPs in heat-stressed cells as a function of the mean-normalized half-life of mRNAs; otherwise, as in (C). (E) Depletion of endogenous CAPRIN1 fused to AID. Immunoblots probed for the indicated proteins show specific depletion of AID fusion protein after treatment with 500 µM IAA for 3 h. (F) Quantification of protein depletion in (E), normalizing to levels of GAPDH. Points show values for three biological replicates. (G) Colocalization of CAPRIN1 in SGs. Quantification of colocalization was performed using Mander’s coefficient between G3BP1 and indicated SG marker, comparing to a control image from an irrelevant field of cells (***, p \(\le \,\)0.001; Welch’s two-sample t-test). (H) SG formation despite CAPRIN1 depletion. HCT116 cells were either treated with 500 µM IAA to deplete CAPRIN1 (right) or not treated (left) prior to arsenite stress (500 µM sodium arsenite for 1 h). Images show either CAPRIN1 in red, SG marker protein G3BP1 in green, or SG marker protein eIF4G in magenta (scale bars, 10 µm). (I) Quantification of SG formation in (P). Points show values for three biological replicates (ns, not significant). (J) Depletion of endogenous UBAP2L fused to AID. Quantification was based on AID::RFP::UBAP2L and PABPC1::GFP fluorescence in homozygously tagged HCT116 cells (images not shown). Points show values for three biological replicates. (K) Colocalization of UBAP2L in SGs. Quantification of colocalization was performed using Mander’s coefficient between PABPC1 and UBAP2L, comparing to a control image from an irrelevant field of cells (***, p \(\le \,\)0.001; Welch’s two-sample t-test). (L) SG formation despite UBAP2L depletion. HCT116 cells were either treated with 500 µM IAA to deplete UBAP2L (right) or not treated (left) prior to arsenite stress (500 µM sodium arsenite for 1 h). Images show either UBAP2L in red or SG marker protein PABPC1 in green; otherwise, as in (P). (M) Quantification of SG formation in (L); otherwise, as in (I).
Extended Data Fig. 5 Conclusions regarding SG-enrichment are consistent across multiple cell types and stresses.
(A) Comparison of SG enrichments. Shown are SG enrichments calculated in Khong et al.18 plotted as a function of SG enrichments calculated in this study. n indicates total number of unique mRNAs. (B) Comparison of mRNA levels for the cells used in (A). Shown are relative mRNA abundances in U2OS cells, as measured by Khong et al.18, plotted as a function of relative abundance in HCT116 cells, as measured in this study. Otherwise as in (A). Our HCT116 cell SG enrichments resembled those previously reported in U2OS cells, especially when considering the differences in overall mRNA expression in the two cell types. (C – E) SG enrichment determinants. Shown is the relationship between either transcript length (C), unstressed TE (D), or stressed TE (E) and SG enrichment. The relationship between ISR translation change and SG enrichment was not simply a consequence of SGs being enriched for transcripts that were poorly translated prior to stress and therefore experienced a smaller decline, as SGs were also enriched for mRNAs that were translated relatively well during stress, as shown in E. otherwise as in Fig. 3c (* p \(\le\) 0.05, ** p \(\le\) 0.01, *** p \(\le\) 0.001). (F) Comparison of SG enrichment in arsenite-stressed U2OS cells as measured by Khong et al.18 and ISR translation in arsenite-stressed HCT116 cells; otherwise, as in Fig. 3b. (G) Comparison of arsenite-stressed TE in HCT116 cells and SG enrichment in arsenite-stressed U2OS cells as measured by Khong et al.18; otherwise, as in (E). (H) Comparison of Stress-Dependent RNP Granule (RG) enrichment and ISR translation in ER-stressed NIH 3T3 cells as measured by Namkoong et al.59; otherwise, as in (F). (I) Comparison of G3BP-depdenent proximity labelling in unstressed HEK293T cells as measured by Ren et al.60 and ISR translation in arsenite-stressed HCT116 cells. ns, not significant; otherwise, as in (F). (J) Comparison of G3BP-depdenent proximity labelling in arsenite-stressed HEK293T cells as measured by Ren et al.60 and ISR translation in arsenite-stressed HCT116 cells; otherwise, as in (I). (K) Comparison of G3BP-dependent translation in HCT116 cells and SG enrichment in U2OS cells as measured by Khong et al.18. All stresses were 500 µM sodium arsenite for 1 h; otherwise, as in (E). (L) Comparison of G3BP-dependent translation in HCT116 cells and RG enrichment in NIH 3T3 cells as measured by Namkoong et al.59. All stresses were heat shock; otherwise, as in (I). (M) Comparison of G3BP-dependent translation in HCT116 cells and G3BP-depdenent proximity labelling in stressed HEK293T cells as measured by Ren et al.60. All stresses were 500 µM sodium arsenite for 1 h; otherwise, as in (K).
Extended Data Fig. 6 OGs are SG-like condensates formed in the absence of stress.
(A) No induction of ectopic SGs caused by UBAP2L overexpression. Unstressed U2OS cells were either treated with 1 µM doxycycline (right) or not treated (left) to induce overexpression of UBAP2L. Images show either UBAP2L in red or SG marker protein eIF4G in magenta (scale bars, 10 µm). White boxes highlight insets that are expanded on the right. (B) Colocalization of UBAP2L puncta with SG marker. Quantification of colocalization was performed using Mander’s coefficient between UBAP2L and eIF4G as an SG marker, comparing to a control image from an irrelevant field of cells (***, p \(\le \,\)0.001; Welch’s two-sample t-test). (C) Induction of ectopic SGs caused by G3BP overexpression. Unstressed HCT116 cells were either treated with 1 µM doxycycline (right) or not treated (left) to induce overexpression of G3BP1. Images show either G3BP1 in green or SG marker protein eIF4G in magenta; otherwise, as in (A). (D) Quantification of ectopic granule formation in (C). Points indicate values for three biological replicates. (E) Kinetics of OG formation and dissolution. Plotted over time is the percent of cells with visible OGs. Cells were exposed to blue light starting at 0 min and removed from blue light at 360 min. Error bars report standard deviation for six biological replicates shown as points. (F) Colocalization of SG markers in OGs. Quantification of colocalization was performed using Mander’s coefficient between GFP::CRY2::G3BPΔN and indicated SG marker, comparing to a control image from an irrelevant field of cells (***, p \(\le \,\)0.001; Welch’s two-sample t-test). (G) FRAP of OGs. Plotted over time is the fluorescence intensity of GFP-labeled granules after photobleaching. SG (green) and OG (blue) recoveries are shown over a 180 s time course. Photobleaching occurred at 3 s. Error bars report standard deviation for at least six granules from at least three cells.
Extended Data Fig. 8 G3BP1-tethered preferential translation occurs across multiple reporters and correlates with translation-dependent mRNA destabilization.
(A) G3BP1 partitioning to SGs. HCT116 cells expressing G3BP1 endogenously tagged with GFP were stressed with 500 µM sodium arsenite for 90 min. Images show localization of G3BP in green. White outlines show cell boundaries as determined by Imaris software based on cytosolic fluorescence. Purple outlines show SG boundaries as determined by Imaris software (scale bars, 1 µm). (B) Quantification of G3BP1 partitioning to SGs. Points show the amount of fluorescence from SGs as a percent of total fluorescence, for eight independent replicates each containing at least three cells. (C) Effects of tagging G3BP with λN and MCP. HCT116 cells expressing endogenous G3BP tagged with the indicated GFP fusion protein were either unstressed (top) or treated with 500 µM sodium arsenite for 1 h (bottom) to assess SG formation (scale bar, 10 µm). White boxes highlight insets that are expanded on the right. (D) mRNA changes during stress. Plotted for each mRNA is its abundance in arsenite-stressed HCT116 cells (500 µM sodium arsenite for 1 h) as a function of its abundance in unstressed HCT116 cells. The GAPDH mRNA, which was used as a control for RT-qPCR, is highlighted in green. The red dashed line indicates unchanged abundance. n indicates total number of unique mRNAs. (E) SG localization for each reporter. Percent of cytoplasmic reporter molecules localized to SGs in untethered cells (green) and in tethered cells (pink) is shown for each reporter with 5’ and 3’ UTR sequences labeled below. Points show values for at least three biological replicates (** p \(\le\) 0.01, *** p \(\le\) 0.001, Welch’s two-sample t-test). The average SG tethering efficiency for the three different reporters was 30% for untethered cells and 82% for tethered cells, with standard deviations of 11% and 10%, respectively. (F) G3BP1-tethered ISR translation response for all reporters, plotted as in Fig. 6d. For comparison, results from Fig. 6d are replotted. (G) Average ISR translation response across all G3BP1-tethered reporters. Plotted are the average TE values across all three reporters shown in (F). (H) G3BP1-tethered ISR protein response for all reporters. Plotted is the fold change in luciferase protein in untethered cells (green) and tethered cells (pink) during a time course of treatment with arsenite stress (500 µM sodium arsenite). Luciferase levels are reported relative to those of unstressed cells from the same biological replicate; otherwise as in (F). (I) Average ISR protein response across all G3BP1-tethered reporters. Plotted are the average luciferase values across all three reporters shown in (H). (J) G3BP1-tethered ISR RNA response for all reporters. Plotted is the fold change in reporter mRNA in untethered cells (green) and tethered cells (pink) during a time course of treatment with arsenite stress (500 µM sodium arsenite). RNA levels are reported relative to those of unstressed cells from the same biological replicate; otherwise as in (F). (K) Average ISR RNA response across all G3BP1-tethered reporters. Plotted are the average mRNA fold changes across all reporters shown in (J). (L) Reporter translation response to translational inhibitors. Plotted is the TE of the CNOT10 reporter in untethered cells treated with 100 µg/ml cycloheximide (light green) or 100 µg/ml puromycin (dark green) in a time course. Time points were taken at 0, 15, 30, 60, 120, and 240 min; otherwise as in (F). (M) Reporter protein response to translational inhibitors. Plotted is the fold change in luciferase protein of the CNOT10 reporter during the time course shown in (L); otherwise as in (G). (N) Reporter mRNA response to translational inhibitors. Plotted is the mRNA fold change of the CNOT10 reporter during the time course shown in (L); otherwise as in (H).
Extended Data Fig. 9 CAPRIN1-tethered preferential translation occurs across multiple reporters and correlates with translation-dependent mRNA destabilization.
(A) Tethering a luciferase reporter to SGs via CAPRIN1. HCT116 cells expressing a NanoLuc reporter bearing 24x MS2 hairpins and the 5’ and 3’ UTRs from CNOT10 along with MCP::RFP::CAPRIN1 were stressed with 500 µM sodium arsenite for 90 min. Images show localization of CAPRIN1 in red and smFISH of NanoLuc reporter molecules in yellow (scale bars, 1 µm). White boxes highlight insets that are expanded on the right. (B) CAPRIN1 tethering efficiency. Percent of cytoplasmic reporter molecules localized to SGs in untethered cells (replotted from Extended Data Figure 8E in green) and to SGs in CAPRIN1-tethered cells (pink) are shown for reporters with 5’ and 3’ UTRs of the indicated genes. Points show values for at least three biological replicates (** p \(\le\) 0.01, *** p \(\le\) 0.001, Welch’s two-sample t-test). The average SG tethering efficiency for the four different reporters was 30% for untethered cells and 89% for tethered cells, with standard deviations of 11% and 4%, respectively. (C) Assessment of SG formation in cells expressing CAPRIN1 tether while depleted of G3BPs. HCT116 cells expressing endogenously tagged AID::RFP::G3BPs were either untreated (left), treated with 1 µM doxycycline and 200 µM auxinole for 10 h, leading to retention of G3BPs and expression of MCP::GFP::CAPRIN1 (middle), or treated with 1 µM doxycycline and 500 µM IAA for 10 h, leading to turnover of endogenous G3BPs and expression of MCP::GFP::CAPRIN1 (right). Cells were subsequently stressed with 500 µM sodium arsenite for 90 min. White boxes highlight insets that are expanded on the right. Scale bar, 10 µm. These results are quantified in Fig. 6f. (D) CAPRIN1-tethered ISR translation response for all reporters. Plotted as in Fig. 6g for all reporters. For comparison, results of Fig. 6g are replotted. (E) Average ISR translation response across all CAPRIN1-tethered reporters. Plotted are the average TE values across all reporters shown in (D). (F) CAPRIN1-tethered ISR protein response for all reporters. Plotted is the fold change in luciferase protein in untethered cells (green), tethered ( + SGs) cells (pink), and tethered (–SGs) cells (red) during a time course of treatment with arsenite stress (500 µM sodium arsenite). Luciferase levels are reported relative to that of unstressed cells from the same biological replicate; otherwise as in (D). (G) Average ISR protein response across all CAPRIN1-tethered reporters. Plotted are the average luciferase values across all three reporters shown in (F). (H) CAPRIN1-tethered ISR RNA response for all reporters. Plotted is the fold change in reporter mRNA in untethered cells (green), tethered ( + SGs) cells (pink), and tethered (–SGs) cells (red) during a time course of treatment with arsenite stress (500 µM sodium arsenite). RNA levels are reported relative to those of unstressed cells from the same biological replicate; otherwise as in (D). (I) Average ISR RNA response across all CAPRIN1-tethered reporters. Plotted are the average RNA fold changes across all reporters shown in (H).
Source data
Source Data Fig. 2 (download PDF )
Unprocessed western blots and images.
Source Data Fig. 4 (download PDF )
Unprocessed western blots and images.
Source Data Fig. 5 (download PDF )
Unprocessed western blots and images.
Source Data Fig. 6 (download PDF )
Unprocessed western blots and images.
Source Data Extended Data Fig. 4 and Table 4 (download PDF )
Unprocessed western blots and images.
Source Data Extended Data Fig. 6 and Table 6 (download PDF )
Unprocessed western blots and images.
Source Data Extended Data Fig. 8 and Table 8 (download PDF )
Unprocessed western blots and images.
Source Data Extended Data Fig. 9 and Table 9 (download PDF )
Unprocessed western blots and images.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Smith, J., Bartel, D.P. The G3BP stress-granule proteins reinforce the integrated stress response translation programme. Nat Cell Biol 28, 135–148 (2026). https://doi.org/10.1038/s41556-025-01834-3
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41556-025-01834-3