Abstract
The intercellular transmission of α-synuclein contributes to Parkinson’s disease pathology. Yet, the mechanisms of α-synuclein spread are not fully understood. Here we used live-cell microscopy to examine the impact of Parkinson’s disease associated lipid alterations on α-synuclein release. We discovered that increased glucosylceramides as a consequence of reduced β-glucocerebrosidase activity induce ectosome shedding from primary neurons and from dopaminergic neurons derived from induced pluripotent stem cells of a patient with Parkinson’s disease harbouring mutations in GBA1 (N370S, L444P and W378G) and LRRK2 (G2019S and R1441H) compared with their isogenic control. We show that elevated glucosylceramide and the pharmacological inhibition of β-glucocerebrosidase similarly increase vesicle release and uptake by other neurons in mouse brains. Finally, we show that ectosomes are loaded with α-synuclein and lead to the transmission of α-synuclein pathology to neighbouring neurons. These data reveal ectosomes as a major route for α-synuclein transmission in Parkinson’s disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
Data supporting this work are available in the Article. Uncropped blots (Source Data Fig. 1), raw data values (Source Data Table 1), statistics information (Supplemental Table 3) and light microscopy reporting table (Supplementary Table 4) are provided with the Article. All other data supporting the findings of this study, including imaging files, are available from the corresponding author upon request. Source data are provided with this paper.
References
Jaunmuktane, Z. & Brandner, S. Invited review: the role of prion-like mechanisms in neurodegenerative diseases. Neuropathol. Appl. Neurobiol. 46, 522–545 (2020).
Yoshida, S. & Hasegawa, T. Deciphering the prion-like behavior of pathogenic protein aggregates in neurodegenerative diseases. Neurochem. Int. 155, 105307 (2022).
Calabresi, P. et al. α-Synuclein in Parkinson’s disease and other synucleinopathies: from overt neurodegeneration back to early synaptic dysfunction. Cell. Death Dis. 14, 176 (2023).
Pan, X., Dutta, D., Lu, S. & Bellen, H. J. Sphingolipids in neurodegenerative diseases. Front. Neurosci. 17, 1137893 (2023).
Fanning, S., Selkoe, D. & Dettmer, U. Parkinson’s disease: proteinopathy or lipidopathy? NPJ Parkinsons Dis. 6, 3 (2020).
Alecu, I. & Bennett, S. A. L. Dysregulated lipid metabolism and its role in α-synucleinopathy in Parkinson’s disease. Front. Neurosci. 13, 1–22 (2019).
Fanning, S. & Selkoe, D. Parkinson disease is a fatty acidopathy. Nat. Rev. Neurol. 21, 642–655 (2025).
Sidransky, E. et al. Multicenter analysis of glucocerebrosidase mutations in Parkinson’s disease. N. Engl. J. Med. 361, 1651–1661 (2009).
Migdalska-Richards, A. & Schapira, A. H. V. The relationship between glucocerebrosidase mutations and Parkinson disease. J. Neurochem. 139, 77–90 (2016).
Nalls, M. A. et al. A multicenter study of glucocerebrosidase mutations in dementia with Lewy bodies. JAMA Neurol. 70, 727–735 (2013).
Brady, R., Kanfer, J. & Shapiro, D. The metabolism of glucocerebrosides. J. Biol. Chem. 240, 39–43 (1965).
Leyns, C. E. G. et al. Glucocerebrosidase activity and lipid levels are related to protein pathologies in Parkinson’s disease. NPJ Parkinsons Dis. 9, 74 (2023).
Mazzulli, J. R. et al. Activation of β-glucocerebrosidase reduces pathological α-synuclein and restores lysosomal function in Parkinson’s patient midbrain neurons. J. Neurosci. 36, 7693–7706 (2016).
Mazzulli, J. R. et al. Gaucher disease glucocerebrosidase and α-synuclein form a bidirectional pathogenic loop in synucleinopathies. Cell 146, 37–52 (2011).
Zunke, F. et al. Reversible conformational conversion of α-synuclein into toxic assemblies by glucosylceramide. Neuron 97, 92–107 (2018).
Burbulla, L. F. et al. A modulator of wild-type glucocerebrosidase improves pathogenic phenotypes in dopaminergic neuronal models of Parkinson’s disease. Sci. Transl. Med. 11, 1–11 (2019).
Schöndorf, D. C. et al. IPSC-derived neurons from GBA1-associated Parkinson’s disease patients show autophagic defects and impaired calcium homeostasis. Nat. Commun. 5, 4028 (2014).
Mahoney-Crane, C. L. et al. Neuronopathic GBA1L444P mutation accelerates glucosylsphingosine levels and formation of hippocampal α-synuclein inclusions. J. Neurosci. 43, 501–521 (2023).
Huebecker, M. et al. Reduced sphingolipid hydrolase activities, substrate accumulation and ganglioside decline in Parkinson’s disease. Mol. Neurodegener. 14, 40 (2019).
Esfandiary, A., Finkelstein, D. I., Voelcker, N. H. & Rudd, D. Clinical sphingolipids pathway in Parkinson’s disease: from GCase to integrated-biomarker discovery. Cells 11, 1353 (2022).
Mielke, M. M. et al. Plasma ceramide and glucosylceramide metabolism is altered in sporadic Parkinson’s disease and associated with cognitive impairment: a pilot study. PLoS ONE 8, e73094 (2013).
Nguyen, M. & Krainc, D. LRRK2 phosphorylation of auxilin mediates synaptic defects in dopaminergic neurons from patients with Parkinson’s disease. Proc. Natl Acad. Sci. USA 115, 5576–5581 (2018).
Ysselstein, D. et al. LRRK2 kinase activity regulates lysosomal glucocerebrosidase in neurons derived from Parkinson’s disease patients. Nat. Commun. 10, 5570 (2019).
Guedes, L. C. et al. Serum lipid alterations in GBA-associated Parkinson’s disease. Parkinsonism Relat. Disord. 44, 58–65 (2017).
Estes, R. E., Lin, B., Khera, A. & Davis, M. Y. Lipid metabolism influence on neurodegenerative disease progression: is the vehicle as important as the cargo? Front. Mol. Neurosci. 14, 788695 (2021).
Welsh, J. A. et al. Minimal information for studies of extracellular vesicles (MISEV2023): from basic to advanced approaches. J. Extracell. Vesicles 13, e12404 (2024).
Sapon, K., Manka, R., Janas, T. & Janas, T. The role of lipid rafts in vesicle formation. J. Cell Sci. 136, jcs260887 (2023).
Fortin, D. L. et al. Lipid rafts mediate the synaptic localization of α-synuclein. J. Neurosci. 24, 6715–6723 (2004).
Fantini, J. & Yahi, N. Molecular basis for the glycosphingolipid-binding specificity of α-synuclein: key role of tyrosine 39 in membrane insertion. J. Mol. Biol. 408, 654–669 (2011).
Lingwood, D. & Simons, K. Lipid rafts as a membrane-organizing principle. Science 327, 46–50 (2010).
Fantini, J., Carlus, D. & Yahi, N. The fusogenic tilted peptide (67-78) of α-synuclein is a cholesterol binding domain. Biochim. Biophys. Acta Biomembr. 1808, 2343–2351 (2011).
Taguchi, Y. V. et al. Glucosylsphingosine promotes α-synuclein pathology in mutant GBA-associated parkinson’s disease. J. Neurosci. 37, 9617–9631 (2017).
Wang, L. et al. Neuronal activity induces glucosylceramide that is secreted via exosomes for lysosomal degradation in glia. Sci. Adv. 8, eabn3326 (2022).
Braak, H. et al. Staging of brain pathology related to sporadic Parkinson’s disease. Neurobiol. Aging 24, 197–211 (2003).
Sugiyama, H., Hainfellner, J. A., Yoshimura, M. & Budka, H. Neocortical changes in Parkinson’s disease, revisited. Clin. Neuropathol. 13, 55–59 (1994).
Feliciano, D. et al. YAP1 nuclear efflux and transcriptional reprograming follow membrane diminution upon VSV-G-induced cell fusion. Nat. Commun. 12, 4502 (2021).
Gao, J., Liao, J. & Yang, G.-Y. Review article CAAX-box protein, prenylation process and carcinogenesis. Am. J. Transl. Res. 1, 312–325 (2009).
Broad, K. et al. Unraveling multilayered extracellular vesicles: speculation on cause. J. Extracell. Vesicles 12, e12309 (2023).
Matthies, D. et al. Microdomains form on the luminal face of neuronal extracellular vesicle membranes. Sci. Rep. 10, 11953 (2020).
Daniels, L. B., Glew, R. H., Radin, N. S. & Vunnam, R. R. A revised fluorometric assay for Gaucher’s disease using conduritol-beta-epoxide with livers as the source of beta-glucosidase. Clin. Chim. Acta 106, 155–163 (1980).
Zimmer, T. S., Orr, A. L. & Orr, A. G. Astrocytes in selective vulnerability to neurodegenerative disease. Trends Neurosci. 47, 289–302 (2024).
Wang, C., Yang, T., Liang, M., Xie, J. & Song, N. Astrocyte dysfunction in Parkinson’s disease: from the perspectives of transmitted α-synuclein and genetic modulation. Transl. Neurodegener. 10, 39 (2021).
Zhang, H. et al. Identification of distinct nanoparticles and subsets of extracellular vesicles by asymmetric flow field-flow fractionation. Nat. Cell Biol. 20, 332–343 (2018).
Dujardin, S. et al. Ectosomes: a new mechanism for non-exosomal secretion of Tau protein. PLoS ONE 9, e100760 (2014).
Chen, C. X.-Q. et al. Generation of patient-derived pluripotent stem cell-lines and CRISPR modified isogenic controls with mutations in the Parkinson’s associated GBA gene. Stem Cell Res. 64, 102919 (2022).
Deneault, E. et al. A streamlined CRISPR workflow to introduce mutations and generate isogenic iPSCs for modeling amyotrophic lateral sclerosis. Methods 203, 297–310 (2022).
Braak, H., Sandmann-Keil, D., Gai, W. & Braak, E. Extensive axonal Lewy neurites in Parkinson’s disease: a novel pathological feature revealed by α-synuclein immunocytochemistry. Neurosci. Lett. 265, 67–69 (1999).
O’Keeffe, G. W. & Sullivan, A. M. Evidence for dopaminergic axonal degeneration as an early pathological process in Parkinson’s disease. Parkinsonism Relat. Disord. 56, 9–15 (2018).
Mathieu, M. et al. Specificities of exosome versus small ectosome secretion revealed by live intracellular tracking of CD63 and CD9. Nat. Commun. 12, 4389 (2021).
Kvalvaag, A. et al. Clathrin mediates both internalization and vesicular release of triggered T cell receptor at the immunological synapse. Proc. Natl Acad. Sci. USA 120, e2211368120 (2023).
Shaver, M., Gomez, K., Kaiser, K. & Hutcheson, J. D. Mechanical stretch leads to increased caveolin-1 content and mineralization potential in extracellular vesicles from vascular smooth muscle cells. BMC Mol. Cell Biol. 25, 8 (2024).
Patnaik, S. et al. Discovery, structure–activity relationship, and biological evaluation of noninhibitory small molecule chaperones of glucocerebrosidase. J. Med. Chem. 55, 5734–5748 (2012).
Aflaki, E. et al. Macrophage models of gaucher disease for evaluating disease pathogenesis and candidate drugs. Sci. Transl. Med. 6, 240ra73 (2014).
Marano, M. et al. Increased glucosylsphingosine levels and Gaucher disease in GBA1-associated Parkinson’s disease. Parkinsonism Relat. Disord. 124, 107023 (2024).
Galvagnion, C. et al. Sphingolipid changes in Parkinson L444P GBA mutation fibroblasts promote α-synuclein aggregation. Brain 145, 1038–1051 (2022).
Samaddar, M. et al. Lysosomal polyamine storage upon ATP13A2 loss impairs β-glucocerebrosidase via altered lysosomal pH and electrostatic hydrolase-lipid interactions. Cell Rep. 44, 116179 (2025).
Realini, N. et al. Discovery of highly potent acid ceramidase inhibitors with in vitro tumor chemosensitizing activity. Sci. Rep. 3, 1035 (2013).
Dementiev, A. et al. Molecular mechanism of inhibition of acid ceramidase by carmofur. J. Med. Chem. 62, 987–992 (2019).
Viel, C. et al. Preclinical pharmacology of glucosylceramide synthase inhibitor venglustat in a GBA-related synucleinopathy model. Sci. Rep. 11, 20945 (2021).
Dai, L. et al. Lysosomal dysfunction in α-synuclein pathology: molecular mechanisms and therapeutic strategies. Cell. Mol. Life Sci. 81, 382 (2024).
Sanyal, A., Novis, H. S., Gasser, E., Lin, S. & LaVoie, M. J. LRRK2 kinase inhibition rescues deficits in lysosome function due to heterozygous GBA1 expression in human iPSC-derived neurons. Front. Neurosci. 14, 442 (2020).
Xian, X. et al. Reversal of ApoE4-induced recycling block as a novel prevention approach for Alzheimer’s disease. eLife 7, e40048 (2018).
Tejwani, L. et al. Lysosomes cell autonomously regulate myeloid cell states and immune responses. Preprint at bioRxiv https://doi.org/10.1101/2024.11.11.623074 (2024).
Fell, M. J. et al. MLi-2, a potent, selective, and centrally active compound for exploring the therapeutic potential and safety of LRRK2 kinase inhibition. J. Pharmacol. Exp. Ther. 355, 397–409 (2015).
Bayati, A. et al. Rapid macropinocytic transfer of α-synuclein to lysosomes. Cell. Rep. 40, 111102 (2022).
Xie, Y. X. et al. Lysosomal exocytosis releases pathogenic α-synuclein species from neurons in synucleinopathy models. Nat. Commun. 13, 4918 (2022).
Brás, I. C. et al. Molecular mechanisms mediating the transfer of disease-associated proteins and effects on neuronal activity. J. Parkinsons Dis. 12, 2397–2422 (2022).
Maroteaux, L., Campanelli, J. T. & Scheller, R. H. Synuclein: a neuron-specific protein localized to the nucleus and presynaptic nerve terminal. J. Neurosci. 8, 2804–15 (1988).
Volpicelli-Daley, L. A. et al. Exogenous α-synuclein fibrils induce Lewy body pathology leading to synaptic dysfunction and neuron death. Neuron 72, 57–71 (2011).
Volpicelli-Daley, L. A., Luk, K. C. & Lee, V. M. Y. Addition of exogenous α-synuclein preformed fibrils to primary neuronal cultures to seed recruitment of endogenous α-synuclein to Lewy body and Lewy neurite-like aggregates. Nat. Protoc. 9, 2135–2146 (2014).
Polinski, N. K. et al. Best practices for generating and using α-synuclein pre-formed fibrils to model Parkinson’s disease in rodents. J. Parkinsons Dis. 8, 303–322 (2018).
Ma, M. R., Hu, Z. W., Zhao, Y. F., Chen, Y. X. & Li, Y. M. Phosphorylation induces distinct α-synuclein strain formation. Sci. Rep. 6, 37130 (2016).
Anderson, J. P. et al. Phosphorylation of Ser-129 is the dominant pathological modification of α-synuclein in familial and sporadic lewy body disease. J. Biol. Chem. 281, 29739–29752 (2006).
Fujiwara, H. et al. Synuclein is phosphorylated in synucleinopathy lesions. Nat. Cell. Biol. 4, 160–164 (2002).
Luberto, C. et al. Inhibition of tumor necrosis factor-induced cell death in MCF7 by a novel inhibitor of neutral sphingomyelinase. J. Biol. Chem. 277, 41128–41139 (2002).
Trajkovic, K. et al. Ceramide triggers budding of exosome vesicles into multivesicular endosomes. Science 319, 1244–1247 (2008).
Kumar, A. et al. Role of extracellular vesicles secretion in paclitaxel resistance of prostate cancer cells. Cancer Drug Resist. 5, 612–624 (2022).
Menck, K. et al. Neutral sphingomyelinases control extracellular vesicles budding from the plasma membrane. J. Extracell. Vesicles 6, 1378056 (2017).
Rojas, C. et al. DPTIP, a newly identified potent brain penetrant neutral sphingomyelinase 2 inhibitor, regulates astrocyte-peripheral immune communication following brain inflammation. Sci. Rep. 8, 17715 (2018).
Gustafsson, G. et al. Secretion and uptake of α-synuclein via extracellular vesicles in cultured cells. Cell. Mol. Neurobiol. 38, 1539–1550 (2018).
Kurzawa-Akanbi, M. et al. Altered ceramide metabolism is a feature in the extracellular vesicle-mediated spread of α-synuclein in Lewy body disorders. Acta Neuropathol. 142, 961–984 (2021).
Duffield, A., Kamsteeg, E.-J., Brown, A. N., Pagel, P. & Caplan, M. J. The tetraspanin CD63 enhances the internalization of the H,K-ATPase-subunit. Proc. Natl Acad. Sci. USA 100, 15560–15565 (2003).
Iguchi, Y. et al. Exosome secretion is a key pathway for clearance of pathological TDP-43. Brain 139, 3187–3201 (2016).
Choi, Y. R. et al. Prion-like propagation of α-synuclein is regulated by the FcγRIIB-SHP-1/2 signaling pathway in neurons. Cell. Rep. 22, 136–148 (2018).
Zurzolo, C. Tunneling nanotubes: reshaping connectivity. Curr. Opin. Cell Biol. 71, 139–147 (2021).
Sáenz-De-Santa-María, I., Henderson, M., Pepe, A. & Zurzolo, C. Identification and characterization of tunneling nanotubes for intercellular trafficking. Curr. Protoc. 3, e939 (2023).
Senol, A. D. et al. A-synuclein fibrils subvert lysosome structure and function for the propagation of protein misfolding between cells through tunneling nanotubes. PLoS Biol. 19, e3001287 (2021).
Dieriks, B. V. et al. α-Synuclein transfer through tunneling nanotubes occurs in SH-SY5Y cells and primary brain pericytes from Parkinson’s disease patients. Sci. Rep. 7, 42984 (2017).
Chang, M. et al. Intercellular communication in the brain through a dendritic nanotubular network. Science 390, eadr7403 (2025).
Hein, L. K., Rozaklis, T., Adams, M. K., Hopwood, J. J. & Karageorgos, L. Lipid composition of microdomains is altered in neuronopathic Gaucher disease sheep brain and spleen. Mol. Genet. Metab. 121, 259–270 (2017).
Hattersley, K. J., Hein, L. K. & Fuller, M. Lipid composition of membrane rafts, isolated with and without detergent, from the spleen of a mouse model of Gaucher disease. Biochem. Biophys. Res. Commun. 442, 62–67 (2013).
Kim, M. J., Jeon, S., Burbulla, L. F. & Krainc, D. Acid ceramidase inhibition ameliorates α-synuclein accumulation upon loss of GBA1 function. Hum. Mol. Genet. 27, 1972–1988 (2018).
Varela, A. R. P. et al. Effect of glucosylceramide on the biophysical properties of fluid membranes. Biochim. Biophys. Acta Biomembr. 1828, 1122–1130 (2013).
Hurley, J. H. ESCRTs are everywhere. EMBO J. 34, 2398–2407 (2015).
Lo, C. H. & Zeng, J. Defective lysosomal acidification: a new prognostic marker and therapeutic target for neurodegenerative diseases. Transl. Neurodegener. 12, 29 (2023).
Colacurcio, D. J. & Nixon, R. A. Disorders of lysosomal acidification—the emerging role of v-ATPase in aging and neurodegenerative disease. Ageing Res. Rev. 32, 75–88 (2016).
Madeira, A. et al. Caveolin-1 interacts with α-synuclein and mediates toxic actions of cellular α-synuclein overexpression. Neurochem. Int. 59, 280–289 (2011).
Vargas, K. J. et al. α-Synuclein colocalizes with AP180 and affects the size of clathrin lattices. J. Biol. Chem. 299, 105091 (2023).
Henderson, M. X. et al. Glucocerebrosidase activity modulates neuronal susceptibility to pathological α-synuclein insult. Neuron 105, 822–836 (2020).
Malfertheiner, K., Stefanova, N. & Heras-Garvin, A. The concept of α-synuclein strains and how different conformations may explain distinct neurodegenerative disorders. Front. Neurol. 12, 737195 (2021).
Gegg, M. E. et al. Glucocerebrosidase deficiency in substantia nigra of parkinson disease brains. Ann. Neurol. 72, 455–463 (2012).
Lin, G. et al. Phospholipase PLA2G6, a parkinsonism-associated gene, affects Vps26 and Vps35, retromer function, and ceramide levels, similar to α-synuclein gain. Cell Metab. 28, 605–618 (2018).
Vos, M. et al. Ceramide accumulation induces mitophagy and impairs β-oxidation in PINK1 deficiency. Proc. Natl Acad. Sci. USA 118, e2025347118 (2021).
Bassil, F. et al. α-Synuclein modulates tau spreading in mouse brains. J. Exp. Med. 218, e20192193 (2021).
Ioannou, M. S., Liu, Z. & Lippincott-Schwartz, J. A neuron-glia co-culture system for studying intercellular lipid transport. Curr. Protoc. Cell. Biol. 84, e95 (2019).
Chen, C. X. Q. et al. A multistep workflow to evaluate newly generated ipscs and their ability to generate different cell types. Methods Protoc. 4, 50 (2021).
Ritter, B., Ferguson, S. M., De Camilli, P. & Mcpherson, P. S. A lentiviral system for efficient knockdown of proteins in neuronal cultures [version 1; referees: 2 approved]. MNI Open Res. 1, 12766.1 (2017).
Sasaki, A., Arawaka, S., Sato, H. & Kato, T. Sensitive western blotting for detection of endogenous Ser129-phosphorylated α-synuclein in intracellular and extracellular spaces. Sci. Rep. 5, 14211 (2015).
Tervo, D. G. R. et al. A designer AAV variant permits efficient retrograde access to projection neurons. Neuron 92, 372–382 (2016).
Xu, Y. H. et al. Dependence of reversibility and progression of mouse neuronopathic Gaucher disease on acid β-glucosidase residual activity levels. Mol. Genet. Metab. 94, 190–203 (2008).
Roed Brekk, O., Honey, J. R., Lee, S., Hallett, P. J. & Isacson, O. Cell type-specific lipid storage changes in Parkinson’s disease patient brains are recapitulated by experimental glycolipid disturbance. Proc. Natl Acad. Sci. USA 117, 27646–27654 (2020).
Ollion, J., Cochennec, J., Loll, F., Escudé, C. & Boudier, T. TANGO: a generic tool for high-throughput 3D image analysis for studying nuclear organization. Bioinformatics 29, 1840–1841 (2013).
Ioannou, M. S. et al. Neuron-astrocyte metabolic coupling protects against activity-induced fatty acid toxicity. Cell 177, 1522–1535 (2019).
Rubio-Atonal, L. F. et al. Glutamate decreases oxidative stress and lipid droplet formation in astrocytes. J. Cell Sci. 138, jcs263983 (2025).
Ralhan, I. et al. Protective ApoE variants support neuronal function by effluxing oxidized phospholipids. Neuron. 25, 00847–5 (2025).
Acknowledgements
We thank P. McPherson, H. Bellen, G. Lin, X. Pan, M. Gu, M. Tyrlik, A. Walimbe, S. Kaduskar and S.-A. Mok for helpful comments on the manuscript. We thank X. Sun, P. Gao and S. van Baarle for assistance with electron microscopy and A. Simmonds for the use of Imaris software. We thank J. Han and his team of the Uvic-Genome BC Proteomics Centre for the sphingolipid analysis and K. Jacquemyn for the support related to serialEM software. Experiments were performed at the Faculty of Medicine and Dentistry Cell Imaging Core RRID: SCR_019200. We thank E. Tayler and L. Kravitz for vector art distributed on SciDraw (https://doi.org/10.5281/zenodo.3925901). This work was supported by the Canadian Institutes of Health Research (grant nos. 173321 and 191990), the Canada Research Chairs Programme (grant no. 2021-00027) and a Future Leaders in Canadian Brain Research from Brain Canada (grant no. 5946). T.D. was supported by GBA1 Canada (G-CAN) research and tools and development funding. J. Jacquemyn was supported by a Dr. Rowland and Muriel Haryett Neuroscience Fellowship and an EMBO Postdoctoral Fellowship (grant no. ALTF 120-2022).
Author information
Authors and Affiliations
Contributions
Conceptualization: M.S.I., J. Jacquemyn. Methodology: M.S.I., J. Jacquemyn, B.M., J. Jackson, C.X.Q.C., T.D. Investigation: J. Jackson. Technical assistance: J.C., E.I., K.C., N.Y.L., L.F.R.A., C.G., J.W., C.A.M. Resources: N.A., M.N., E.D., Z.Y. Visualization: M.S.I., J. Jacquemyn. Funding acquisition: M.S.I., J. Jacquemyn, T.D. Supervision: M.S.I., J. Jackson, T.D. Writing—original and revised draft: M.S.I., J. Jacquemyn. Writing—review and editing: M.S.I., J. Jacquemyn, J. Jackson, C.X.Q.C., T.D.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Cell Biology thanks Dan Li and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Increased glucosylceramide and reduced GCase activity induces ectosome formation in neurons.
a, Live-cell image of cortical neuron expressing mCh-GPI treated with NBD-glucosylceramide (NBD-GlcCer). Boxed area highlighting vesicles budding from soma and magnified on the left. b, Intensity of ethidium homodimer-1 (EthD-1) staining of neurons treated with NBD-GlcCer relative to control. n = 4 independent experiments; mean ± SEM; Two-tailed one sample t-test. c, Cytotoxicity assessment of NBD-GlcCer on cortical neurons by MTT assay. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; Two-tailed Mann-Whitney test. d, Time-lapse imaging showing mCh-GPI positive ectosome biogenesis with NBD-GlcCer treatment. e, Images of attached and detached ectosomes containing intraluminal vesicles labeled with mCh-GPI and NBD-GlcCer from the neuron-conditioned media. f, GCase activity of cortical neurons treated with CBE. The plotted line is a non-linear fit of values. n = 4 independent experiments. g, Heatmap showing fold change of significantly altered HexCer (galactosylceramides and glucosylceramides) of neuron treated with 100 µM CBE. n = 5 independent experiments. h, Average fold change of HexCer species with CBE treatment. n = 5 independent experiments. P values calculated using unpaired two-tailed t-test with false discovery rate adjusted using Benjamini-Hockberg method. i, Live-cell image of cortical neuron expressing mVenus-CAAX treated with 3 µM CBE, to induce 50% activity reduction. The arrows highlight vesicles budding from the plasma membrane along the neurites. Boxes show magnified images on the right. j, Amount of mVenus-CAAX ectosome buds per cell area relative to control. n = 4 independent experiments; 10 cells/coverslip/treatment; mean ± SEM; Two-tailed one sample t-test. k-m, Size distribution of attached and detached mCh-GPI and mVenus-CAAX-positive vesicles from neurons treated with 3 and 100 µM CBE. n = 4 independent experiments; for 3 and 100 µM CBE a total of 111 attached, 231 attached and 64 detached were analyzed respectively; mean ± SEM. n-o, Neurons were treated with CBE and stained with ethidium homodimer-1 (EthD-1) and assessed for cytotoxicity by MTT assay. n = 4 independent experiments; mean ± SEM; One-way ANOVA with Dunn’s multiple comparisons test. p, GCase activity of cortical astrocytes treated with CBE. The plotted line is a non-linear fit of values. n = 4 independent experiments; mean ± SEM. q, Live-cell image of cortical astrocytes expressing mVenus-CAAX treated with 100 µM CBE. Boxes show magnified images with orthogonal views on the right confirming ectosome budding from plasma membrane. r, Amount of mVenus-CAAX ectosome buds per astrocyte area relative to control. n = 4 independent experiments; 10 cells/coverslip/treatment; mean ± SEM; Two-tailed one sample t-test. s, Representative image of detached mVenus-CAAX positive ectosome from the astrocyte-conditioned media. 4 independent experiments were performed. Graphs are depicted as superplots where biological replicates are shown in large shapes, and technical replicates are shown as small shapes.
Extended Data Fig. 2 Ectosomes containing tdTomato fluorophore are released by cortical neurons treated with glucosylceramide in vivo.
a, Percentage of NBD-GlcCer positive buds containing the tdTomato fluorophore. n = 4 independent experiments; mean ± SEM. b, Coronal section of mouse brain with saline or NBD-GlcCer injection. Tiled widefield and fluorescent images are displayed. Boxes indicate the saline or NBD-GlcCer injection site. c, Maximum intensity projection of fixed tissue showing a cortical neuron expressing tdTomato after saline administration. Boxed area shows magnification of tdTomato positive vesicle, ≥ 2 µM from neuron, with orthogonal views on the right confirming it is fully detached. d, Number of tdTomato positive puncta in NeuN positive cells. n = 3 animals; 5 images per n; mean ± SEM; Two-tailed unpaired t-test. e, Number of NBD-GlcCer positive puncta in NeuN positive cells. n = 3 animals; 5 images per n; mean ± SEM; Two-tailed unpaired t-test. f, Percentage of tdTomato positive puncta with NBD-GlcCer in Neun positive cells neighboring the tdTomato positive cortical neuron. n = 3 animals; 5 images per n; mean ± SEM; Two-tailed unpaired t-test. Graphs are depicted as superplots where biological replicates are shown in large shapes, and where applicable technical replicates are shown as small shapes.
Extended Data Fig. 3 Increased mVenus-CAAX-positive ectosomes from GBA1 and LRRK2 patient-derived dopaminergic neurons.
a, iPS cell-derived dopaminergic neurons fixed and immunostained for β3-tubulin and tyrosine hydroxylase. b, Percentage of tyrosine hydroxylase positive dopaminergic neurons in GBA1 and LRRK2 mutant lines and their isogenic controls. n = 4 independent inductions; mean ± SEM. c, Live-cell maximum intensity projection of LRRK2-G2019S and isogenic corrected control (corr) iPS cell-derived dopaminergic neurons expressing mVenus-CAAX. Boxed areas highlighting ectosomes forming at the plasma membrane are magnified below. d, Number of mVenus-CAAX positive buds per cell area in LRRK2-G2019S and control iPS cell-derived dopaminergic neuron. n = 3 independent experiments; mean ± SEM; Two-tailed one sample t-test. e-f, Size distribution of attached mVenus-CAAX-positive vesicles in GBA1 and LRRK2 mutant lines. independent experiments: n = 3 for GBA1 (total 118 vesicles) and n = 4 for LRRK2 (total 239 vesicles); mean ± SEM. g-h Cytotoxicity assessment of mVenus-CAAX expression in GBA1 and LRRK2 mutant lines and their isogenic controls with MTT assay. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. Graphs are depicted as superplots where biological replicates are shown in large shapes, and where applicable technical replicates are shown as small shapes.
Extended Data Fig. 4 Increased tubulin-positive ectosomes from GBA1 and LRRK2 patient-derived dopaminergic neurons.
a, Schematic of differential ultracentrifugation procedure used to separate extracellular vesicles (EVs). b-d, Western blotting of centrifugation fractions prepared from the GBA1-N370S mutant and its isogenic control as in A. CL, cell lysate; S, supernatant; P, pellet. Quantification of CD81 and tubulin present in 10,000 x g fractionated pellet normalized to the cell lysate and relative to the isogenic control. n = 3 independent experiments; mean ± SEM; Two-tailed one sample t-test. Graph shows the experimental averages. e, Percentage mVenus-CAAX positive buds containing tubulin induced by CBE. n = 3 independent experiments; mean ± SEM; Two-tailed unpaired t-test. f, Live-cell image of cortical neurons expressing mVenus-CAAX treated with SiR-tubulin and CBE. Boxed areas highlighting an ectosome containing tubulin is magnified below. g, Size distribution of mVenus-CAAX-positive ectosome buds from neurons treated with SiR-tubulin and 100 µM CBE. n = 4 independent experiments and a total of 77 attached vesicles were analyzed. h, Live-cell image of the GBA1-N370S neuron expressing mVenus-CAAX treated with SiR-tubulin. Boxed areas highlighting an ectosome containing tubulin is magnified below. i, Percentage mVenus-CAAX positive buds containing tubulin in GBA1-N370S mutant and its isogenic corrected control. n = 3 independent experiments; mean ± SEM; Two-tailed unpaired t-test. A total of 113 attached vesicles were analyzed. j, Size distribution of mVenus-CAAX-positive ectosome buds in SiR-tubulin treated GBA1-N370S neurons. k-m, Cytotoxicity assessment of SiR-tubulin treatment on mVenus-CAAX expressing GBA1-N370S and control neurons, as well as in NBD-glucosylceramide (NBD-GlcCer) or CBE treated cortical neurons by MTT assay. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison and Two-tailed Mann-Whitney test. n-q, Maximum intensity projection of GBA1-N370S iPS cell-derived dopaminergic neurons with Tubb3 and caveolin-1 or clathrin. Boxed areas showing single plane magnified tubulin-positive buds containing clathrin and caveolin-1. Percentage tubulin positive buds containing clathrin and caveolin-1 in GBA1-N370S neuron. n = 4 independent experiments; mean ± SEM. r, Maximum intensity projections of GBA1-N370S dopaminergic neurons and isogenic controls +/- Pitstop 2 treatment, stained for β3-tubulin. Arrows indicate ectosomes budding from the neurite and soma. s, Quantification of tubulin-positive buds relative to control with Pitstop 2 treatment. n = 4 independent experiments; 8 cells per experiment; Mean ± SEM; One-way ANOVA with Šidák’s correction for multiple comparisons. Graphs are depicted as superplots where biological replicates are shown in large shapes, and technical replicates are shown as small shapes.
Extended Data Fig. 5 Validation of hGCase/eGFP expression in dopaminergic neurons.
a-b, Cytotoxicity assessment of the GCase modulator 758 on GBA1-N370S, LRRK2-G2019S and corresponding isogenic control neurons by MTT. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. c-h, Western blotting of GBA1-N370S, LRRK2-G2019S and corresponding isogenic control neurons expressing hGCase/eGFP or eGFP and immunoblotted for hGCase, GFP and β-actin. Expression of hGCase and eGFP normalized against β-actin. n = 4 independent experiments, mean ± SEM. i-j, Percentage of eGFP positive dopaminergic neurons in GBA1-N370S, LRRK2-G2019S and corresponding isogenic controls. n = 4 independent experiments; 4 tile scans /2 coverslip/experiment; mean ± SEM. k-l, Cytotoxicity assessment of hGCase/eGFP expression on GBA1-N370S, LRRK2-G2019S and corresponding isogenic control neurons by MTT. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. Graphs are depicted as superplots where biological replicates are shown in large shapes, and where applicable technical replicates are shown as small shapes.
Extended Data Fig. 6 Sphingolipid profiling of lysates and large vesicle fraction containing ectosomes in dopaminergic neurons.
a, Schematic overview of sphingolipid pathway, species are color coded, GCase and important lipids in bold. b-q, Abundance of lipids in lysates and large extracellular vesicle fractions (lEV) from GBA1-N370S neurons and isogenic corrected controls. n = 5 independent experiments from separate inductions; Mean ± SEM; Two-tailed unpaired t-tests with Šidák’s correction for multiple comparisons. Graphs are depicted as superplots where biological replicates are shown in large shapes, and technical replicates are shown as small shapes.
Extended Data Fig. 7 Ectosome formation is inhibited by ibiglustat and MLi-2, but unaffected by glucosylsphingosine or carmofur.
a, Live-cell images of cortical neurons expressing mVenus-CAAX +/- glucosylsphingosine (GlcSph). Magnified images of neurites on the right. b, Amount of mVenus-CAAX ectosome buds per cell area relative to control with GlcSph treatment. n = 4 independent experiments; 10 cells/coverslip/treatment; mean ± SEM; Two-tailed one sample t-test. c, Cytotoxicity assessment of GlcSph on cortical neurons by MTT assay. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; Two-tailed Mann-Whitney test. d, Quantification of average diameter of attached tubulin-positive vesicles in GBA1-N370S +/- carmofur. n = 4 independent experiments; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. e, Cytotoxicity assessment of carmofur (Car) treatment on dopaminergic neurons by MTT assay. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Šidák’s correction for multiple comparisons. f-g, Quantification of average diameter of attached tubulin-positive vesicles in GBA1-N370S +/- ibiglustat. n = 4 independent experiments; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. h-i, Cytotoxicity assessment of ibiglustat on GBA1-N370S, LRRK2-G2019S and corresponding isogenic control neurons by MTT. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. j-k, Cytotoxicity assessment of rimeporide on GBA1-N370S, LRRK2-G2019S and corresponding isogenic control neurons by MTT. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. l, Cytotoxicity assessment of LRRK2 kinase inhibitor, MLi-2, on LRRK2-G2019S and corresponding isogenic control neurons by MTT. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. Graphs are depicted as superplots where biological replicates are shown in large shapes, and where applicable technical replicates are shown as small shapes.
Extended Data Fig. 8 Validation of α-synuclein pre-formed fibrils.
a-b, Thioflavin T increases emission at 482 nm with pre-formed fibrils (PFFs) generated from monomeric α-synuclein indicating the presence of beta sheet-rich structures which are absent from monomeric or PBS controls. 2x, double concentration. n = 6-8 independent experiments; mean ± SEM. c, Transmission electron micrographs of α-synuclein PFFs before and after sonication. d-e, Length distribution of fluorescently labeled and unlabeled PFFs after sonication. f, Percent area of pS129-α-synuclein relative to β3-tubulin in primary cortical neurons over time. n = 4 independent experiments; mean ± SEM; two-way ANOVA with Dunn’s multiple comparison test. g-h, Images of cortical cultures at DIV14 treated with PBS, monomeric α-synuclein, tagged PFFs (Atto-594) or untagged PFFs stained for β3-tubulin and pS129-α-synuclein. Boxed areas are magnified below and on the right. i, Representative live-cell image of ATTO594-α-synuclein fibrils in ectosomes formed upon addition of NBD-GlcCer in cortical neurons. Boxed areas are magnified on the left. 4 independent experiments were performed.
Extended Data Fig. 9 Ectosomes as a vehicle for the transmission of pathogenic α-.
a-b, Cytotoxicity assessment of PFFs on NBD-GlcCer and CBE treated cortical neurons expressing mVenus-CAAX by MTT assay. n = 4 independent experiments; 3 technical replicates/experiment; mean ± SEM; Two-tailed Mann-Whitney test and One-way ANOVA with Dunn’s multiple comparison. c, Number of mVenus-CAAX positive vesicles per area in GBA1 isogenic control neurons +/-PFFs. Independent experiments: n = 3 control and n = 4 for PFF-ATTO594 treatment; 10 cells/coverslip/treatment; mean ± SEM; Two-tailed one sample t-test. d, Cytotoxicity assessment of PFFs on mVenus-CAAX expressing GBA1-N370S and isogenic control neurons by MTT. n = 6 independent experiments; 3 technical replicates/experiment; mean ± SEM; One-way ANOVA with Dunn’s multiple comparison. e-f, Western blotting of centrifugation fractions derived from GBA1-N370S and control neurons treated with PFFs and immunoblotted for flotilin-1, pS129-α-synuclein and synuclein. CL, cell lysate; S, Supernatant; P, Pellet. 2 K, 2000 x g; 10 K, 10,000 x g; 100k, 100,000 x g. n = 3 independent experiments. g, Procedures used to separate vesicles and low molecular weight α-synuclein released by GBA1-N370S neurons by differential centrifugation. h, Image of fixed GBA1-N370S dopaminergic showing pS129-α-synuclein in tubulin-positive ectosomal bud. Boxed areas are magnified on the right. i, Percentage of tubulin-positive buds containing pS129-α-synuclein in PFFs60-treated GBA1-N370S dopaminergic. n = 4 independent experiments; 10 cells/experiment. Mean ± SEM. Graphs are depicted as superplots where biological replicates are shown in large shapes, and technical replicates are shown as small shapes.
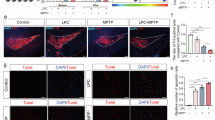
Extended Data Fig. 10 Ectosome-mediated propagation of phosphorylated α-synuclein in GBA1-N370S neurons.
a-b, Maximum intensity projections of GBA1-N370S and control neurons incubated with large vesicle fraction derived from GBA1-N370S + /- PFF for 14 days and stained with β3-tubulin and pS129-α-synuclein. Quantifications of the amount of intracellular pS129-α-synuclein normalized to the neuron area in GBA1-N370S and control neurons treated with different centrifugation fractions after 14 days of incubation. n = 3 independent experiments; 6 cells/experiment. Mean ± SEM, One-way ANOVA with uncorrected Fisher’s LSD. c, Schematic representation of the multi-step propagation of pathogenic α-synuclein through large vesicles/ectosomes. d-e, Quantifications of the amount of intracellular pS129-α-synuclein normalized to the neuron area in GBA1-N370S and control neurons treated with large vesicle fraction from multi-step process depicted in (C) after 7 days of incubation. n = 4 independent experiments; 8 cells/experiment. Mean ± SEM; One-way ANOVA with uncorrected Fisher’s LSD. Max intensity projections of GBA1-N370S and control neurons incubated with the second-generation large vesicle fractions derived from GBA1-N370S. f-h, Size distribution of attached β3-tubulin-positive vesicles from GBA1-N370S + /- GW4869 and corresponding isogenic control neurons. n = 4 independent experiments; a total of 138, 199 and 234 attached vesicles for isogenic control and GBA1-N370S + /- GW4869 were analyzed; bars show mean ± SEM. i, Average size of vesicle diameter of GBA1-N370S + /- GW4869. n = 4 independent experiments; bars show mean ± SEM; Two-tailed Mann-Whitney test. j, Cytotoxicity assessment of GW4869 on GBA1-N370S and isogenic control neurons by MTT assay. n = 4 independent experiments; 3 technical replicates per experiment; bars show mean ± SEM; One-way ANOVA, with Dunn’s multiple correction test. k, Percentage of Iba1 positive microglia neighboring the tdTomato-expressing neuron containing tdTomato puncta. n = 5 animals; 6 images per n; mean ± SEM; Two-tailed unpaired t-test. l, Maximum intensity projection showing a cortical neuron expressing tdTomato after saline or CBE administration and immunostained for Iba1 and DAPI. Boxed area, maximum intensity projection, single slice magnification and orthogonal views (YZ; scale bar 2 µm) showing tdTomato-positive puncta internalized by neighboring microglia stained with Iba1. Graphs are depicted as superplots where biological replicates are shown in large shapes, and where applicable technical replicates are shown as small shapes.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–10. List and availability information for all iPS cell lines used in the study. Validation and characterization of new lines generated.
Supplementary Video (download MP4 )
Supplementary Video 1. A super-resolution microscopy image of a coronal brain section showing detached tdTomato-NBD-GlcCer positive puncta in three dimensions.
Supplementary Table (download XLSX )
Supplementary Table 1. List of significantly altered HexCer species, as determined by lipidomics, in CBE-treated cortical neurons versus control neurons.
Supplementary Table (download XLSX )
Supplementary Table 2. List of sphingolipid species in ectosome fraction and cell lysates of GBA1-N370S dopaminergic neurons and corresponding isogenic control.
Supplementary Table (download XLSX )
Supplementary Table 3. Statistics used for the study.
Supplementary Table (download XLSX )
Supplementary Table 4. Light microscopy reporting table.
Source data
Source Data Fig. 1 (download PDF )
Source Data Fig. 1 uncropped blots. Unprocessed western blots.
Source Data Extended Data Table 1 (download XLSX )
Source Data Table 1 source data. Raw data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jacquemyn, J., Marriott, B., Chang, J. et al. Glucosylceramide-induced ectosomes propagate pathogenic α-synuclein in Parkinson’s disease. Nat Cell Biol 28, 492–506 (2026). https://doi.org/10.1038/s41556-026-01871-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41556-026-01871-6
This article is cited by
-
Ectosomes step forward in α-synuclein spread
Nature Cell Biology (2026)