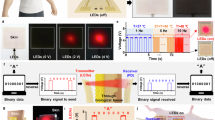
Abstract
Engineered microbes can be used for biomolecular sensing and therapeutic interventions. However, they cannot be monitored and controlled while in vivo. Here we combine optogenetically engineered Escherichia coli Nissle 1917, an ingestible optoelectronic capsule and a wireless smartphone to establish a bidirectional biological–optical–electronic signal processing chain for diagnostic or therapeutic capabilities under user control. As a proof of concept, we engineered E. coli Nissle 1917 to detect inflammation-associated nitric oxide in the pig gut and generate a bioluminescent signal for diagnosis of colitis. This signal is transduced by the optoelectronic capsule into a wireless electrical signal and remotely monitored by a smartphone. Smartphone wireless signals activate LED irradiation in the optoelectronic capsule, in turn activating the microbial expression and secretion of an anti-inflammatory nanobody to alleviate colitis in pigs. This approach highlights the potential for integrating synthetic biology and optoelectronics for digital health monitoring and controllable intervention.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Source data are provided with this paper. These data are also available via figshare at https://doi.org/10.6084/m9.figshare.29207006.v1 (ref. 50).
References
Riglar, D. T. & Silver, P. A. Engineering bacteria for diagnostic and therapeutic applications. Nat. Rev. Microbiol. 16, 214–225 (2018).
McNerney, M. P., Doiron, K. E., Ng, T. L., Chang, T. Z. & Silver, P. A. Theranostic cells: emerging clinical applications of synthetic biology. Nat. Rev. Genet. 22, 730–746 (2021).
Archer, E. J., Robinson, A. B. & Süel, G. M. Engineered E. coli that detect and respond to gut inflammation through nitric oxide sensing. ACS Synth. Biol. 1, 451–457 (2012).
Daeffler, K. N. et al. Engineering bacterial thiosulfate and tetrathionate sensors for detecting gut inflammation. Mol. Syst. Biol. 13, 923 (2017).
Scott, B. M. et al. Self-tunable engineered yeast probiotics for the treatment of inflammatory bowel disease. Nat. Med. 27, 1212–1222 (2021).
Lubkowicz, D. et al. Reprogramming probiotic Lactobacillus reuteri as a biosensor for Staphylococcus aureus derived AIP-I detection. ACS Synth. Biol. 7, 1229–1237 (2018).
Mao, N., Cubillos-Ruiz, A., Cameron, D. E. & Collins, J. J. Probiotic strains detect and suppress cholera in mice. Sci. Transl. Med. 10, eaao2586 (2018).
Zou, Z. P., Du, Y., Fang, T. T., Zhou, Y. & Ye, B. C. Biomarker-responsive engineered probiotic diagnoses, records, and ameliorates inflammatory bowel disease in mice. Cell Host Microbe 31, 199–212 (2021).
Canale, F. P. et al. Metabolic modulation of tumours with engineered bacteria for immunotherapy. Nature 598, 662–666 (2021).
Gurbatri, C. R., Arpaia, N. & Danino, T. Engineering bacteria as interactive cancer therapies. Science 378, 858–864 (2022).
Sun, D. et al. Utilizing engineered bacteria as ‘cell factories’ in vivo for intracellular RNA-loaded outer membrane vesicles’ self-assembly in tumor treatment. ACS Nano 18, 35296–35309 (2024).
Vockley, J. et al. Efficacy and safety of a synthetic biotic for treatment of phenylketonuria: a phase 2 clinical trial. Nat. Metab. 5, 1685–1690 (2023).
Puurunen, M. K. et al. Safety and pharmacodynamics of an engineered E. coli Nissle for the treatment of phenylketonuria: a first-in-human phase 1/2a study. Nat. Metab. 3, 1125–1132 (2021).
Kurtz, C. B. et al. An engineered E. coli Nissle improves hyperammonemia and survival in mice and shows dose-dependent exposure in healthy humans. Sci. Transl. Med. 11, eaau7975 (2019).
Hwang, I. Y. et al. Engineered probiotic Escherichia coli can eliminate and prevent Pseudomonas aeruginosa gut infection in animal models. Nat. Commun. 8, 15028 (2017).
Barger, N., Oren, I., Li, X., Hadib, M. & Daniel, R. A whole-cell bacterial biosensor for blood markers detection in urine. ACS Synth. Biol. 10, 1132–1142 (2021).
Cui, M. et al. NIR light-responsive bacteria with live bio-glue coatings for precise colonization in the gut. Cell Rep. 36, 109690 (2021).
Zhang, X. et al. A red light-controlled probiotic bio-system for in-situ gut-brain axis regulation. Biomaterials 294, 122005 (2023).
Cui, M. et al. Smartphone bioelectronic drug with visual colorimetric sensor and bulk nanoencapsulation optogenetic bacteria for chronic kidney disease theragnostics. Chem. Eng. J. 451, 138812 (2023).
Martínez, E. P. & Robles, E. P. Capsule endoscopy and deep enteroscopy. Endoscopy 46, 787–790 (2014).
Ding, Z. et al. Novel scheme for non-invasive gut bioinformation acquisition with a magnetically controlled sampling capsule endoscope. Gut 70, 2297–2306 (2021).
You, S. S. et al. An ingestible device for gastric electrophysiology. Nature 7, 497–508 (2024).
Hou, B. et al. A swallowable X-ray dosimeter for the real-time monitoring of radiotherapy. Nat. Biomed. Eng. 7, 1242–1251 (2023).
Ramadi, K. B. et al. Bioinspired, ingestible electroceutical capsules for hunger-regulating hormone modulation. Sci. Robot. 8, eade9676 (2023).
Srinivasan, S. S. et al. RoboCap: robotic mucus-clearing capsule for enhanced drug delivery in the gastrointestinal tract. Sci. Robot. 7, eabp9066 (2022).
De la Paz, E. et al. A self-powered ingestible wireless biosensing system for real-time in situ monitoring of gastrointestinal tract metabolites. Nat. Commun. 13, 7405 (2022).
Shi, J. et al. Active biointegrated living electronics for managing inflammation. Science 384, 1023–1030 (2024).
Deng, J. et al. Electrical bioadhesive interface for bioelectronics. Nat. Mater. 20, 229–236 (2021).
Mimee, M. et al. An ingestible bacterial-electronic system to monitor gastrointestinal health. Science 360, 915–918 (2018).
Inda-Webb, M. E. et al. Sub-1.4 cm3 capsule for detecting labile inflammatory biomarkers in situ. Nature 620, 386–392 (2023).
Ausländer, S. & Fussenegger, M. Synthetic biology: toehold gene switches make big footprints. Nature 516, 333–334 (2014).
Bernard, P. & Couturier, M. Cell killing by the F plasmid CcdB protein involves poisoning of DNA-topoisomerase II complexes. J. Mol. Biol. 226, 735–745 (1992).
Ong, N. T. & Tabor, J. J. A miniaturized Escherichia coli green light sensor with high dynamic range. ChemBioChem 19, 1255–1258 (2018).
Hartsough, L. A. et al. Optogenetic control of gut bacterial metabolism to promote longevity. Elife 9, e56849 (2020).
An, B. et al. Programming living glue systems to perform autonomous mechanical repairs. Matter 3, 2080–2092 (2020).
Lynch, J. P. et al. Engineered Escherichia coli for the in situ secretion of therapeutic nanobodies in the gut. Cell Host Microbe 31, 634–649 (2023).
Cosnes, J. et al. Adalimumab or infliximab as monotherapy, or in combination with an immunomodulator, in the treatment of Crohn’s disease. Aliment. Pharmacol. Ther. 44, 1102–1113 (2016).
Holland, I. et al. Type I protein secretion-deceptively simple yet with a wide range of mechanistic variability across the family. EcoSal Plus https://doi.org/10.1128/ecosalplus.ESP-0019-2015 (2016).
Yang, C. & Merlin, D. Unveiling colitis: a journey through the dextran sodium sulfate-induced model. Inflamm. Bowel Dis. 30, 844–853 (2024).
Saco, Y. & Bassols, A. Acute phase proteins in cattle and swine: a review. Vet. Clin. Pathol. 52, 50–63 (2023).
Waka, M. et al. Serum amyloid A is a better predictive biomarker of mucosal healing than C-reactive protein in ulcerative colitis in clinical remission. BMC Gastroenterol. 20, 85 (2020).
Cui, M. et al. Optotheranostic nanosystem with phone visual diagnosis and optogenetic microbial therapy for ulcerative colitis at-home care. ACS Nano 15, 7040–7052 (2021).
Cooper, R. M. et al. Engineered bacteria detect tumor DNA. Science 381, 682–686 (2023).
Fan, C., He, N. & Yuan, J. Cascaded amplifying circuit enables sensitive detection of fungal pathogens. Biosens. Bioelectron. 250, 116058 (2024).
Gurbatri, C. R. et al. Engineering tumor-colonizing E. coli Nissle 1917 for detection and treatment of colorectal neoplasia. Nat. Commun. 15, 646 (2024).
Centurion, F. et al. Nanoencapsulation for probiotic delivery. ACS Nano 15, 18653–18660 (2018).
Rebeck, O. N. et al. A yeast-based oral therapeutic delivers immune checkpoint inhibitors to reduce intestinal tumor burden. Cell Chem. Biol. 32, 98–110.e7 (2025).
Cheng, A. G. et al. Design, construction, and in vivo augmentation of a complex gut microbiome. Cell 185, 3617–3636.e19 (2022).
Abdigazy, A. et al. End-to-end design of ingestible electronics. Nat. Electron. 7, 102–118 (2024).
Wang, H. Source data of research Ingestible optoelectronic capsules enable bidirectional communication with engineered microbes for controllable therapeutic interventions. figshare https://doi.org/10.6084/m9.figshare.29207006.v1 (2025).
Acknowledgements
We thank the Laboratory Center of the School of Life Sciences (Tianjin University) and Experimental Animal Center of Northwest Agriculture and Forestry University for facility support. We thank C. Yang and X. Ma from Beijing Institute of Technology for assisting EcN genomic manipulation including gene knockout and integration. We also thank H. Yi, Z. Wang, W. Guo and Z. Jiang of the School of Life Sciences, Longyan University, and X. Sun and Y. Wei of the College of Veterinary, Northwest Agriculture and Forestry University, for their help in the animal experiments. This work is supported by the following fundings: National Key Research and Development Program of China (2021YFA0909300, D.L.), National Natural Science Foundation of China for Excellent Young Scholars (32122047, Hanjie Wang), Tianjin Natural Science Foundation for Distinguished Young Scholars (23JCJQJC00210, Hanjie Wang), Beijing-Tianjin-Hebei Basic Research Cooperation Project of Beijing Natural Science Foundation (23JCZXJC00370, Hanjie Wang) and Key Program of Tianjin Natural Science Foundation (22JCZDJC00230, Hanjie Wang).
Author information
Authors and Affiliations
Contributions
Hanjie Wang, D.L. and T.D. developed the key concept of this work and guided the whole research process. Z.F. designed and assembled the electronic capsules. X.Z., D.L. and J.L. designed the plasmids and strains. X.Z., Z.F., H.L., L.L., C.Z., H.X., Y.W., D.S., X.L., M.L., S.L. and J.L. constructed the plasmids and strains and performed in vitro synthetic biological experiments. X.Z. and H.L. designed and performed in vivo mouse experiments. X.Z., H.Y., P.D., T.D. and Hanjie Wang conceptualized and designed the ex vivo and in vivo pig experiments. X.Z., Z.F., H.Y., H.L., L.L., P.D., C.Z. and Hanxin Wang performed ex vivo and in vivo pig experiments. Z.F. and H.L. did the intestinal visualization experiments. X.Z., D.L. and Hanjie Wang wrote the paper.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Microbiology thanks David Carreño and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Citation diversity statement
The authors have taken local and regional research relevant to this study into account in citations.
Extended data
Extended Data Fig. 1 Illustration of monitoring and control of intestinal engineered microbes via optoelectronic capsules.
The optoelectronic capsule and the optogenetically engineered microbes are designed to establish the bidirectional signal processing chain. One direction from microbes to the optoelectronic capsule and the ex vivo smartphone is for monitoring microbial status through bioluminescent signals generated by engineered microbes in response to specific biomarkers. Another direction from the APP-controlled optoelectronic capsule to microbes is for activating the expression and secretion of therapeutic proteins. When applied in vivo, the whole process can be used for in-situ diagnose and intervention of intestinal disease. Figure created with BioRender.com.
Extended Data Fig. 2 Basic properties of the optoelectronic capsule used in this study.
(a) The composition and the whole size of optoelectronic capsule. (b) The user interface of the APP on smartphone for recording photocurrent signal. (c) The irradiating characterization of LED loaded in the optoelectronic capsule during 36 h. (d) The processing of raw photocurrent data through a sliding average filtering algorithm for smooth output when the optoelectronic capsule was placed under different conditions of luminescence Source data.
Extended Data Fig. 3 Monitoring of EcN-Lux in pig’s daily feed via optoelectronic capsule.
(a) Scheme of photocurrent measurement at particular time intervals after pig’s oral administration of EcN-Lux. (b) Mixing of the engineered EcN into daily feed for pig administration. (c) X-ray photos of the process of placing the optoelectronic capsule inside pig rectum to the 35 cm depth from anus with the assistance of a moderately soft pipeline. (d) Measurement of the photocurrents derived from EcN-Lux at 16 h interval after the strains were orally administrated Source data. Panels a and d created with BioRender.com.
Extended Data Fig. 4 Simulation experiments to test bioluminescence of EcN-NO-Lux.
(a) Scheme of monitoring the bioluminescence of EcN-NO-Lux when sensing NO/nitrate. (b) Imaging of EcN-NO-Lux in mice model within 4 hours after the administration of strains and nitrate. (c) Recording of photocurrents for monitoring the microbial dynamic sensing of nitrate within 2 h. (d), (f) The in vitro simulation experiment to mimic in vivo intestinal conditions without or with feeds. (e), (g) Detection of EcN-NO-Lux performance in within 24 h in the intestinal simulation conditions in (d) or (f) using four different methods. (h) Monitoring of the nitrate biosensing via the optoelectronic capsule while passing through an intestinal tissue segment containing evenly distributed EcN-NO-Lux. (i) Imaging of the real pig intestinal section in (h). The capsule was pulled from the ‘Start’ point to the ‘End’ point slowly to simulate the peristalsis in the gut. (j) Changes of photocurrents along with the optoelectronic capsule passing through the intestinal section. All the measurements in the above experiments were taken from distinct biological replicates. Data are presented as the mean ± SD. n = 3. Significance was determined by two-way ANOVA with Bonferroni’s post-test for (e) and (g). The significance labelled as ‘ns’ meant p > 0.05 Source data. Panel b created with BioRender.com.
Extended Data Fig. 5 Simulation experiments to test bioluminescence of EcN-Ara-Lux.
(a) Schematic diagram of the arabinose-sensing genetic circuit. (b) Measurement of the bioluminescence densities of EcN-Ara-Lux when sensing different arabinose concentrations. (c) Measurement of the bioluminescence changes of EcN-Ara-Lux when sensing 10 mM arabinose within 4 h. (d) Imaging of EcN-Ara-Lux in mice model within 4 hours after the administration of strains and arabinose. (e), (f) Photocurrent measurement from the EcN-Ara-Lux when responding to different concentrations of arabinose. (g) Monitoring of arabinose distribution via the optoelectronic capsule while passing through an intestinal tissue segment containing evenly distributed EcN-Ara-Lux. (h) Imaging of the real pig intestinal section in (g). The capsule was pulled from the ‘Start’ point to the ‘End’ point slowly. (i) Changes of photocurrents along with the optoelectronic capsule passing through the intestinal section. (j) Schematic diagram of monitoring the arabinose-sensing EcN via optoelectronic capsule in the surgery-exposed pig colon sections. (k), (l) Comparison of the photocurrents measured from the EcN-Ara-Lux when adding arabinose or not. All the measurements in the above experiments were taken from distinct biological replicates. Data are presented as the mean ± SD. n = 3. Significance was determined by two-way ANOVA with Bonferroni’s post-test for (l) Source data. Panel j created with BioRender.com.
Extended Data Fig. 6 Characterization of EcN equipped with CcaS-CcaR optogenetic circuit.
(a) Construction of the CcaS-CcaR two-component optogenetic circuit in EcN. (b) The activation of GFP expression after 2 h irradiation of green light on the EcN tubes. (c) Comparison of activated GFP expression in different strains, 1: EcN-MUT1-GFP; 2: EcN-MUT2-GFP; 3: EcN-SC101-GFP; 4: EcN-RSF-GFP; 5: EcN-Ara-GFP (after 8 hours of 0.2% arabinose induction); 6: EcN-Opto-GFP (after 8 hours of green light induction). (d) Test of the effect of ON-OFF switch after 1 h green-light irradiation on the EcN-Opto-GFP performance. (e) Comparison of the luminescence changes of EcN-Opto-GFP luciferase under different conditions. (f) Imaging of EcN-Opto-Lux in mice model irradiated by LED array loaded on the cage within 4 hours. (g), (h) Imaging and measurement of luciferase expression activated by the green light of capsule-LED at a distance from 0 to 70 mm. All the measurements were taken on the same mouse at different time points. All the measurements in the above experiments were taken from distinct biological replicates. Data are presented as the mean ± SD. n = 3.
Extended Data Fig. 7 Characterization of optoelectronic capsule orally administrated by live pig.
(a) Remote control of LED equipped in the optoelectronic capsule in vivo by APP (left) and the LED switch images under colonoscopy (right). (b) X-ray imaging of placing the electronic capsule into the esophagus via a gastric catheter. (c) X-ray imaging of the optoelectronic capsule in live pig at different hours after administration. (d) Confirmation of the connectivity of optoelectronic capsule in the whole period in pig gastrointestinal tract. (e) X-ray imaging of the optoelectronic capsule at three different positions in live pig. (f) The Bluetooth signal intensities of the optoelectronic capsule in vivo fluctuated at different positions. All the measurements in the above experiments were taken from distinct biological replicates. Data are presented as the mean ± SD. n = 3 Source data. Panels a and d created with BioRender.com.
Extended Data Fig. 8 Biosafety accessing of optoelectronic capsule.
(a) Detection of extra heat generated from optoelectronic capsule with LED switching on and off when placed in an incubator mimicking body temperature. (b) Scheme of biosafety accessing of optoelectronic capsule in pig body. Several blood indicators were detected before and after the two rounds of capsule ingestion for 4 days each round. (c) Comparison of pathological images of the pigs taking optoelectronic capsule or not. Sclar Bar=200μm. (d)–(g) Detection of the levels of C-reactive protein (CRP), hemoglobin (HGB), leukocyte, and platelet before and after the ingestion of optoelectronic capsule. The relatively normal parameter value range was shown in each figure by dashed line. All the measurements in the above experiments were taken from distinct biological replicates. Data are presented as the mean ± SD. n = 3. Significance was determined according to unpaired, two-tailed student’s t-test for (a). The significance labelled as ‘ns’ meant p > 0.05 Source data. Panel b created with BioRender.com.
Extended Data Fig. 9 Supplemented characterization of diagnosis and alleviation of colitis.
(a)–(d) Detection of photocurrent value by the capsule, serum CRP, SAA and fecal CalP on Day 4. (e)–(h) Detection of photocurrent value by the capsule, serum CRP, SAA and fecal CalP on Day 5. (i) Detection of EcN-NO-Lux density changes based on the quantification of LuxA DNA copies at Day 3, 4 and 5. (j) Detection of EcN-NO-Lux density changes based on the quantification of Nb-TNF DNA copies at Day 6, 10 and 14. All the measurements in the above experiments were taken from distinct biological replicates. Data are presented as the mean ± SD. n = 3. Significance was determined by one-way ANOVA with Tukey’s post-test. The significance labelled as ‘ns’ meant p > 0.05.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–9.
Supplementary Video 1 (download MP4 )
Remote control of electronic capsule-loaded LED in the intestine. To visualize the remote control of the electronic capsule in vivo and examine its functionality, we placed the capsule into pig’s rectum trans-anally at ~30 cm depth, repeatedly turned on and off the capsule-loaded LED under remote Bluetooth control on the smartphone APP and recorded the LED status via colonoscopy.
Supplementary Video 2 (download MP4 )
The simulated process of administering the electronic capsules into the pig’s oesophagus. The process of placing electronic capsules into the oesophagus of pigs was demonstrated through the simulation video. Using glycerol for lubrication, we inserted a soft catheter with an electronic capsule at one end into the pig’s oesophagus to a depth of ~40 cm and then used a guide rod to push the capsule deep into the oesophagus.
Supplementary Video 3 (download MP4 )
The wireless connectable time range when the electronic capsule was in the intestine of pigs. When the electronic capsule was at its position as shown in the right panel of Extended Data Fig. 7c (96 h after oral administration), the available range of the location of user’s smartphone to maintain Bluetooth connectivity was recorded.
Supplementary Tables (download XLSX )
Supplementary Tables 1–4 in an integrated Excel file.
Supplementary Data 1 (download XLSX )
Source data of all the supplementary figures in the form of Excel table.
Source data
Source Data Figs. 1–4 and Extended Data Figs. 2–9 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, X., Feng, Z., Li, H. et al. Ingestible optoelectronic capsules enable bidirectional communication with engineered microbes for controllable therapeutic interventions. Nat Microbiol 10, 1841–1853 (2025). https://doi.org/10.1038/s41564-025-02057-w
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41564-025-02057-w
This article is cited by
-
Switching bacteria on via a smartphone
Nature Reviews Microbiology (2025)
-
Remote control designer probiotics
Nature Microbiology (2025)