Abstract
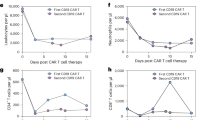
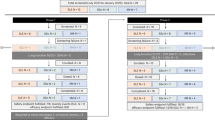
Systemic lupus erythematosus (SLE) remains refractory to conventional immunosuppression in a subset of patients. In treatment-refractory SLE, we show that peripheral CD19⁺ B cells and bone marrow CD19⁻BCMA⁺ long-lived plasma cells are dominant autoantibody sources, motivating dual CD19 and BCMA targeting. Here we report results from a cohort of patients (14 female, one male) in an ongoing phase 1 dose-escalation trial of co-infused autologous anti-CD19 and anti-BCMA chimeric antigen receptor (CAR) T cells after fludarabine/cyclophosphamide lymphodepletion. Primary endpoints were dose-limiting toxicities (DLTs) within 28 days and adverse events within 12 weeks; key secondary endpoints comprised attainment of Lupus Low Disease Activity State (LLDAS) and DORIS remission within 12 weeks and in vivo CAR-T persistence within 24 weeks. Exploratory endpoints were the duration of post-infusion B cell depletion and time to recovery, the kinetics of immune reconstitution and longitudinal changes in autoantibody titers and serum immunoglobulin concentrations after CAR-T therapy. Over a median 712-day follow-up (range, 613–1,134), no DLTs occurred. Grade 1 cytokine release syndrome developed in 86.7% of patients, with no neurotoxicity or treatment-related deaths. The most common grade 3 or higher adverse events were neutropenia (100%), thrombocytopenia (40%) and anemia (13.3%), all of which were reversible with supportive care. By week 12, 12 of 15 patients (80%) fulfilled both the LLDAS and DORIS remission criteria. Multiomic analyses confirmed elimination of autoreactive CD19⁺BCMA⁺ clones, reconstitution of naive IgM/IgD B cells and durable downregulation of interferon-stimulated and BAFF-dependent signatures, indicating improved immune homeostasis. Longitudinal monitoring of three patients for 1 year demonstrated sustained eradication of pathogenic clones, suggesting potential cure. Dual anti-CD19/anti-BCMA CAR-T cell therapy demonstrates good safety and promising clinical efficacy in treatment-refractory SLE. This study supports the further development of this treatment approach for patients with rSLE. ClinicalTrials.gov identifier: NCT05030779.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
Single-cell RNA sequencing, bulk BCR sequencing and single-cell BCR sequencing data that support the findings of this study have been deposited in the Genome Sequence Archive for Human of the China National Center for Bioinformation (CNCB) under accession number HRA009827. Proteomics raw files were uploaded to the OMIX platform of the CNCB under accession number OMIX008301. Original image datasets generated in the study can be found in Mendeley Data: https://doi.org/10.17632/y424dfbd78.1 Any additional requirement for further data sharing can be submitted via email to the lead contact: huanghe@zju.edu.cn. External data requests will be responded to within 60 business days.
References
Siegel, C. H. & Sammaritano, L. R. Systemic lupus erythematosus: a review. JAMA 331, 1480–1491 (2024).
Fanouriakis, A. et al. 2019 update of the EULAR recommendations for the management of systemic lupus erythematosus. Ann. Rheum. Dis. 78, 736–745 (2019).
Mougiakakos, D. et al. CD19-targeted CAR T cells in refractory systemic lupus erythematosus. N. Engl. J. Med. 385, 567–569 (2021).
Mackensen, A. et al. Anti-CD19 CAR T cell therapy for refractory systemic lupus erythematosus. Nat. Med. 28, 2124–2132 (2022).
Muller, F. et al. CD19 CAR T-cell therapy in autoimmune disease—a case series with follow-up. N. Engl. J. Med. 390, 687–700 (2024).
Zhang, W. et al. Treatment of systemic lupus erythematosus using BCMA-CD19 compound CAR. Stem Cell Rev. Rep. 17, 2120–2123 (2021).
Wang, W. et al. BCMA-CD19 compound CAR T cells for systemic lupus erythematosus: a phase 1 open-label clinical trial. Ann. Rheum. Dis. 83, 1304–1314 (2024).
Chen, W. et al. Distinct transcriptomes and autocrine cytokines underpin maturation and survival of antibody-secreting cells in systemic lupus erythematosus. Nat. Commun. 15, 1899 (2024).
Taddeo, A. et al. Long-lived plasma cells are early and constantly generated in New Zealand Black/New Zealand White F1 mice and their therapeutic depletion requires a combined targeting of autoreactive plasma cells and their precursors. Arthritis Res. Ther. 17, 39 (2015).
Hoyer, B. F. et al. Short-lived plasmablasts and long-lived plasma cells contribute to chronic humoral autoimmunity in NZB/W mice. J. Exp. Med. 199, 1577–1584 (2004).
Jacobi, A. M. et al. HLA-DRhigh/CD27high plasmablasts indicate active disease in patients with systemic lupus erythematosus. Ann. Rheum. Dis. 69, 305–308 (2010).
Anolik, J. H. et al. Rituximab improves peripheral B cell abnormalities in human systemic lupus erythematosus. Arthritis Rheum. 50, 3580–3590 (2004).
Halliley, J. L. et al. Long-lived plasma cells are contained within the CD19−CD38hiCD138+ subset in human bone marrow. Immunity 43, 132–145 (2015).
Qin, C. et al. Single-cell analysis of anti-BCMA CAR T cell therapy in patients with central nervous system autoimmunity. Sci. Immunol. 9, eadj9730 (2024).
Crow, M. K. Pathogenesis of systemic lupus erythematosus: risks, mechanisms and therapeutic targets. Ann. Rheum. Dis. 82, 999–1014 (2023).
Deng, Y. et al. Expression characteristics of interferon-stimulated genes and possible regulatory mechanisms in lupus patients using transcriptomics analyses. EBioMedicine 70, 103477 (2021).
Schoggins, J. W. et al. A diverse range of gene products are effectors of the type I interferon antiviral response. Nature 472, 481–485 (2011).
Vincent, F. B., Morand, E. F., Schneider, P. & Mackay, F. The BAFF/APRIL system in SLE pathogenesis. Nat. Rev. Rheumatol. 10, 365–373 (2014).
Yan, Z. et al. A combination of humanised anti-CD19 and anti-BCMA CAR T cells in patients with relapsed or refractory multiple myeloma: a single-arm, phase 2 trial. Lancet Haematol. 6, e521–e529 (2019).
Yang, C. et al. Allogeneic anti-CD19 CAR-T cells induce remission in refractory systemic lupus erythematosus. Cell Res. 35, 607–609 (2025).
Shu, J. et al. Safety and clinical efficacy of Relmacabtagene autoleucel (relma-cel) for systemic lupus erythematosus: a phase 1 open-label clinical trial. EClinicalMedicine 83, 103229 (2025).
An, L. et al. Humanized CD19 CAR-T cells in relapsed/refractory B-ALL patients who relapsed after or failed murine CD19 CAR-T therapy. BMC Cancer 22, 393 (2022).
Zhang, M. et al. Risk factors associated with durable progression-free survival in patients with relapsed or refractory multiple myeloma treated with anti-BCMA CAR T-cell therapy. Clin. Cancer Res. 27, 6384–6392 (2021).
Song, F. et al. Safety and efficacy of autologous and allogeneic humanized CD19-targeted CAR-T cell therapy for patients with relapsed/refractory B-ALL. J. Immunother. Cancer 11, e005701 (2023).
Lee, D. W. et al. ASTCT consensus grading for cytokine release syndrome and neurologic toxicity associated with immune effector cells. Biol. Blood Marrow Transpl. 25, 625–638 (2019).
Acknowledgements
We thank members of the H.C.B. and L.U. laboratories for insightful comments and discussions over the course of the study. We are grateful to Z. Liu (Zhejiang University) for providing critical comments on the manuscript and for helpful discussions. This study was supported by the National Natural Science Foundation of China (82341206, 82425002, 82270235, 82270234 and 82000179); the Key Project of Science and Technology Department of Zhejiang Province (2023C03060); and the Zhejiang Province Vanguard and Leading Goose Research Program (2024SSYS0025).
Author information
Authors and Affiliations
Contributions
H.H. conceptualized and supervised the project. A.C., Z.L., L.L., J.L. and Y.H. supervised the project. J.F., D.H., R.H, X.J. and R.W. performed experiments, analyzed the data, prepared the figures and wrote the manuscript. Clinical study implementation was conducted by J.F., H.C., S.F., M.Z., G.W., Z.L., J.L., Y.H. and H.H. M.S. did the clinical trial informatic collection and analysis. J.C. and S.H. did the flow cytometry experiment and analysis. Other authors provided assistance in molecular cloning, in vitro assay and library preparation. All authors commented on the figures and read and edited the manuscript. All authors critically reviewed and edited the manuscript. J.F., D.H. and H.H. had full access to all study data and verified the underlying results. All authors approved the final manuscript, agreed to its submission and take full responsibility for the content’s accuracy, adherence to the trial protocol (NCT05030779) and statistical analysis validity.
Corresponding authors
Ethics declarations
Competing interests
A.H.C. is a founding member of Shanghai YaKe Biotechnology, Ltd., a biotechnology company focusing on research and development of tumor cellular immunotherapy. The remaining authors declare no conflicts of interest.
Peer review
Peer review information
Nature Medicine thanks Jialiang Li, Sham Mailankody and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Saheli Sadanand, in collaboration with the Nature Medicine team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 In vitro antibody expression and validation.
(a) Feature plot illustrating the expression of CD19, BCMA, and CD138 in peripheral blood mononuclear cell (PBMC) and bone marrow mononuclear cell (BMMC) groups from three patients with SLE. (b) Quantitative analysis of anti-antinuclear antibody (ANA) immunofluorescence assay related to Fig. 1h.
Extended Data Fig. 2 Characteristics of CAR-T cells in vitro.
(a) In vitro expansion of CD3+ T cells between the beginning and the end of the production process (X varied from 8 to 14) of anti-CD19 CAR-T cells. (b) In vitro expansion of CD3+ T cells between the beginning and the end of the production process (X varied from 8 to 14) of anti-BCMA CAR-T cells. (c) Transduction rate (percentage of transfected cells of CD3+ T cells) of anti-CD19 CAR-T cells. (d) Transduction rate (percentage of transfected cells of CD3+ T cells) of anti-BCMA CAR-T cells. (e) Fraction of CD8+CD4- T cells, CD4+CD8- T cells, CD4+CD8+ or CD4-CD8- T cells (included in else) of anti-CD19 CAR-T cells. (f) Fraction of CD8+CD4- T cells, CD4+CD8- T cells, CD4+CD8+ or CD4-CD8- T cells (included in else) of anti-BCMA CAR-T cell. (g) Fractions of naïve (Tn), central memory (Tcm), effector memory (Tem) and effector (Te) cells of anti-CD19 CAR-T cells. (h) Fractions of Tn, Tcm, Tem and Te cells of anti-BCMA CAR-T cells.
Extended Data Fig. 3 Analysis of long-term clinical indicators, BCR, and proteomics dynamics in patients with SLE.
(a) Long-term follow-up of C3, C4, anti-antinuclear antibody (ANA), anti-double-stranded DNA (dsDNA) levels in patients’ peripheral blood. (b) Changes in 24-h urinary protein for patient 2. (c) Complementarity-Determining Region (H3CDRH3) sequencing results for patients at the time of B cell populations recovery. (d) Venn diagram illustrating the overlap of unique and shared proteins among four groups: control, before treatment, after treatment, and recovery. The numbers within each segment represent the count of proteins specific to each group or shared between groups. (e) Volcano plot displaying the log10(FOT) of protein abundance against the rank of quantified proteins. The plot highlights significant changes in protein expression, with points further from the center indicating greater fold changes. (f) Bar chart comparing the number of quantified proteins (GP Number) across various patient (p8, p10, p7, p9) and experimental conditions, including control, before treatment, after treatment, recovery. (g) Serum proteome analysis at the Before, After and Recover time points in patients with SLE. The color scale represents z-scores, indicating the deviation of signal intensity. (h) Gene Ontology (GO) enrichment analysis of differentially expressed proteins.
Extended Data Fig. 4 The expansion of anti-CD19 CAR-T cells and anti-BCMA CAR-T cells in the peripheral blood of each patient within one month by flow cytometry.
Red line, anti-CD19 CAR-T cells. Blue line, anti-BCMA CAR-T cells.
Extended Data Fig. 5 Expression of canonical markers on scRNA-seq in patients with SLE.
(a-f) Dot plots of gene expression for peripheral blood mononuclear cell (PBMC) clusters(a), bone marrow mononuclear cell (BMMC) clusters(b), T cell subclusters in PBMCs(c), T cell subclusters in BMMCs(d), B cell subclusters in PBMCs(e), B cell subclusters in BMMCs(f). The color scale represents the average log-normalized gene expression within each immune cell subset.
Extended Data Fig. 6 CAR-T cell therapy restores immune homeostasis.
(a) Violin plot shows the interferon-stimulated gene (ISG) scores in immune cells from PBMCs of three individual patients before treatment and at the time of recovery, based on an independent scRNA-seq experiment. Data are presented as median values with interquartile range (25th–75th percentiles); whiskers indicate minimum and maximum values. Statistical significance was determined using a two-sided Wilcoxon signed-rank test; **** indicates p < 0.0001. (b) Chord plot displays the interactions among various immune cell types. (c) Bar plot displays changes in immune cell interaction pathways at the Before and Recover time points. (d) Circle plot displays the intercellular interaction pathways of immune cells, highlighting B-cell activating factor (BAFF) signaling at the Before and Recover, as well as TNF-related apoptosis-inducing ligand (TRAIL) and Fas ligand (FASLG) signaling during recovery.
Extended Data Fig. 7 Changes in the interactions of immune cells in the bone marrow in Before and Recover group.
(a) Violin plot shows the interferon-stimulated gene (ISG) scores in immune cells from bone marrow mononuclear cells (BMMCs) of three individual patients before treatment and at the time of recovery, based on an independent scRNA-seq experiment. Data are presented as median values with interquartile range (25th–75th percentiles); whiskers indicate minimum and maximum values. Statistical significance was determined using a two-sided Wilcoxon signed-rank test; **** indicates p < 0.0001. (b) Bar plot displays changes in immune cell interaction pathways at the Before and Recover time points.
Extended Data Fig. 8 Comparison of memory B cell diversification Following CAR-T cell therapy.
(a) Uniform manifold approximation and projection (UMAP) plot of 40,232 (pre-treatment) and 31,597 (post-recovery) clones. UMAP plot colored by clone size. NA(X=0), Single (0 < X <= 1), Small (1 < X <= 3), Medium (3 < X <= 10), Large (10 < X <= 100). (b) Relative proportion composition of each clone size cluster, scaled by the total number of peripheral blood and tumor cells. (c) Percent of unique clones by total number of B cell receptor (BCRs) sequenced by patients. (d) Total abundance of clonotypes by sample. (e) Isotypes of heavy chain and light chain from patients with SLE. (f) Overlap of clonotypes between Before and Recover in each patient. (g-h) Gene Ontology (GO) Functional Enrichment analysis of upregulated (g) and downregulated (h) genes in Before and Recover group.
Extended Data Fig. 9 BCR Summarize from patients with SLE.
(a-c) Quantification of the proportion of V (a), D (b), or J (c) gene usage in the heavy chain from patients with SLE at the Before and Recover time points. The color scale denotes the percentage. (d–f) Quantification of the proportion of V (d), D (e), or J (f) gene usage in the light chain from patients with SLE at the Before and Recover time points. The color scale denotes the percentage.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Feng, J., Huo, D., Hong, R. et al. Co-infusion of CD19-targeting and BCMA-targeting CAR-T cells for treatment-refractory systemic lupus erythematosus: a phase 1 trial. Nat Med 31, 3725–3736 (2025). https://doi.org/10.1038/s41591-025-03937-8
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s41591-025-03937-8
This article is cited by
-
The dawn of ‘off-the-shelf’ B cell-depleting therapies for autoimmune diseases
Nature Reviews Rheumatology (2026)
-
Challenges and strategies in clinical applications of CAR-T therapy for autoimmune diseases
Journal of Hematology & Oncology (2025)
-
Chimeric antigen receptor (CAR) therapies for precise eradication of pathogenic cells in autoimmunity
Arthritis Research & Therapy (2025)
-
Developing cell therapies for lupus
Nature Medicine (2025)