Abstract
Preclinical mouse models are critical for understanding the pathophysiological response to infections and developing treatment strategies for sepsis. In keeping with ethical values, researchers follow guidelines to minimize the suffering of the mice. Weight loss is a criteria used as a humane end point, but there is no official recommendation for a maximum weight loss leading to euthanasia. To evaluate whether the thresholds used in daily practice are optimal, we performed a comprehensive retrospective analysis of data generated over 10 years with > 2300 mice used in models of infection with Listeria monocytogenes, Streptococcus pneumoniae, Candida albicans and H1N1 influenza virus. Weight loss segregated mice that survived from those that did not. Statistical analyses revealed that lowering the weight loss thresholds used (none, 30% or 20%) would have increased mortality rates due to the sacrifice of mice that survived infections (p < 0.01–0.001). Power calculations showed high variability and reduction of power as weight loss thresholds approached 20% for S. pneumoniae and L. monocytogenes models. Hence, weight loss thresholds need to be adapted to each model of infection used in a laboratory. Overall, weight loss is a valuable predictor of mortality that contributes to the robustness of composite scores. To our knowledge, this is the most extensive study exploring the relationship between weight loss threshold and sepsis outcome. It underscores the importance of the infection-model-specific evaluation of weight loss for use in clinical scores defining humane endpoints to minimize mouse suffering without compromising statistical power and scientific objectives.
Similar content being viewed by others
Introduction
Animal experimentation is a complementary pilar to in silico, in vitro and ex vivo analyses in advancing our understanding of the regulation of biological processes and the development of diseases. Rodents, mainly mice, account for around 95% of the animals that are utilized in biomedical research1. The use of mice has become widespread due to: (1) the relatively ease of obtaining large numbers, (2) the panel of congenic and genetically modified strains, (3) genetic similarities to humans, (4) the wealth of knowledge gained over decades of research, and (5) adaptability to controlled experimental conditions. Research using rodents has been instrumental to decipher host pathogen interactions and to advance therapeutics for human health such as antimicrobials and immunomodulatory therapies2.
A conscientious approach to preclinical research involves a commitment to the principles of the 3Rs (replacement, reduction, refinement). 3Rs advocate the ethical treatment of research animals and aim to minimize their suffering3. Scoring systems based on subjective and objective parameters such as behavior, external clinical signs, body weight and body temperature have been developed to scale rodent welfare and include a humane endpoint, i.e. a cut-off value at which animals are euthanized to avoid excessive suffering4,5. Scoring systems are established in collaboration with local veterinary and ethics authorities, adapted to the research theme (oncology, behavior, etc.) and updated to reflect changes in local legislation and ethical guidelines6. Body weight loss is one of the main criteria to evaluate the well-being of mice, but its usage either alone or in combination with other criteria is debated7,8,9,10,11,12. There is no official weight loss recommendation for euthanasia, but a threshold of 20% is often used13.
The innate immune system provides the first line of defense against infections, and a dysregulated host response underlies severe infections and sepsis14. Sepsis, defined as a life-threatening organ dysfunction caused by an infection15,16, is responsible for one in five deaths worldwide. Recently, the world health organization recognized sepsis as a global priority and antimicrobial resistance (a contributor to sepsis) as one of the top ten threats to global health. Following these initiatives, Switzerland launched the Swiss sepsis national action plan, identifying four priorities among which research development17. Participating to that effort, our laboratory has a long-standing expertise in studying host–pathogen interactions and testing biomarkers and innovative therapies for infected patients18,19,20. As an example, our work on the cytokine macrophage migration inhibitory factor (MIF) led to the identification of small molecules inhibitors of MIF21,22, and the development of humanized anti-MIF monoclonal antibodies that have been evaluated in phase 1/2a clinical trials23.
In daily practice, we use models of infections induced by bacteria, viruses and fungi inoculated through different routes to induce peritonitis, pneumonia and systemic infection. We adapt the inoculum to induce either a moderate or a severe infection, and we use genetic and pharmacological approaches to modulate host defenses. To take all these conditions into account, a weight loss of 20% or 30% was recently adopted as a humane endpoint in agreement with regulatory authorities. To assess whether these thresholds were optimal, we performed comprehensive retrospective analyses of data obtained over 10 years and with more than 2300 mice used in models of infections with Listeria monocytogenes, Streptococcus pneumoniae, Candida albicans and H1N1 influenza virus (H1N1). We observed that reducing the weight loss thresholds leading to euthanasia substantially increased mortality and decreased statistical stability and power in a model-dependent manner. Our data suggest that weight loss thresholds need to be adapted to each infectious model so that the composite clinical scores including weight loss minimize mouse suffering without jeopardizing statistical power and scientific objectives.
Methods
Mouse models of infection
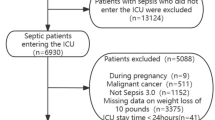
No mice were used for this study. Data were retrieved from experiments performed in 2014–2023 and approved by the Veterinary Affairs Department of the Direction Générale de l’Agriculture, de la Viticulture et des Affaires Vétérinaires of Etat de Vaud (Saint-Sulpice, Switzerland) (Refs.24,25,26,27,28,29,30,31,32,33,34,35,36 and unpublished data). Experiments were performed with Streptococcus pneumoniae American type collection (ATCC) 6303 and H1N1 PR08 (Porto Rico 08) IV inoculated intranasally under anesthesia with ketamine/xylazine, and Listeria monocytogenes 10403S and Candida albicans ATCC 90028 inoculated intravenously. Among 2400 used mice, 43 were excluded from our analyses (Fig. 1). Experiments were designed to determine lethal dose 50 (LD50) (n = 228 mice) or to compare at least two experimental conditions (n = 2129 mice). Altogether, 2024 C57BL/6J mice and 333 BALB/c mice were used (Fig. 1).
Number of mice used in statistical and power analyses. Experiments were conducted from June 2014 to March 2023. Exclusion criteria included death just following anaesthesia (n = 39) and incorrect injection of the infectious agent (n = 4). Statistical analyses were performed on 2'357 mice. Power analyses were performed on 1858 and 2199 mice for the in-house and simulation-based calculations. Experiments without an appropriate \({H}_{0}\) (n = 271) or determining LD50 (n = 228) were excluded from in-house power analysis.
Score sheets
In vivo experiments involved regular recording of the mice using preestablished score sheets. The experiments performed with authorizations covering 2014 to 2018 (cohort 1) had no limit on weight loss (Supplementary Table 1). Visual criteria were used to assess the well-being of the animals, with scores ranging from 0 (normal) to 4 (moribund). A score of 4 led to mouse euthanasia. The mice were observed at least once daily regardless the severity score. The weight was measured at the same time as the score was evaluated. The experiments performed with authorizations covering 2018 to 2023 (cohort 2) combined objective and subjective criteria (Supplementary Table 2). The combined score led to different actions: monitor mice 3 times a week, once a day or twice a day, and euthanasia. On its own, a weight loss ≥ 20% led to the euthanasia of mice infected with L. monocytogenes, and a weight loss ≥ 30% led to the euthanasia of mice infected with C. albicans, S. pneumoniae and H1N1. Score points were recorded on spreadsheets.
Time of event
The log-rank test was used to analyze mouse survival37. The time of event is the time of death with or without euthanasia (Supplementary Tables 1 and 2), measured in days post-infection. To explore the impact of the threshold at which weight loss leads to euthanasia, a new time of event was computed for each mouse regarding its maximum weight loss during the experiment. A time of event was calculated for thresholds varying from 5 to 30% weight loss with an increment of 1%. The computation was performed in python 3.10.6 and data are available on GitHub (https://github.com/mbrochut/weight_loss).
Statistical analysis
To evaluate the relationship between weight loss and health status, we used data from cohort 2, where both parameters were available. The maximum weight loss was plotted against the highest clinical score. The Pearson correlation coefficient and associated p values were calculated using pearsonr from the SciPy v1.12.0 library. To test the distribution of mice that did (survivors) and did not (non-survivors) survive infection for different thresholds, we used the Pearson’s chi-squared test (\({\chi }^{2}\)). A test of \({\chi }^{2}\) was applied for each model of infection, and for each threshold of interest: 10%, 15%, 20% and 25%. To analyze the difference in weight loss over time between mice that survived or not during the experiment, a Kruskal–Wallis test was used day-by-day and a mixed effects model was computed using statsmodels 0.14.0 Python package to evaluate the effect of weight loss on multiple days simultaneously. Due to the nature of the computation of mixed effect model that excludes data with premature death, data from the first 4–8 days of the experiments were fitted to include a maximum of mice and to avoid a survival bias. A complete model using data collected all days of the experiments was also fitted. p values < 0.05 were considered to be statistically significant.
Power analysis
Euthanasia based on weight loss influences statistical results. First, a mouse can end up in the dead or alive groups if we modify the threshold leading to sacrifice. Second, the survival time alters the results of the log-rank test. We defined the rejection of the null hypothesis (\({H}_{0}\), hypothesis of no difference between groups) in favor of the alternative hypothesis (\({H}_{1}\), hypothesis of an effect size between groups) when the p value of a test is lower than 0.0538. The rejection of the null hypothesis is directly related to the power (P), which is the probability to reject \({H}_{0}\) in favor of \({H}_{1}\) knowing there is a difference between groups, i.e. \({H}_{1}\) is true39:
We can express this probability in terms of a fraction, defined by the number of times we have rejected \({H}_{0}\) in favor of \({H}_{1}\) divided by the total number of p values (#p) that were calculated:
Power calculation is meaningful when there is an expected effect size to be tested between groups. Power was investigated using in-house data-based calculation or simulation-based calculation. The in-house data-based calculation used data from experiments carried out in the laboratory with a control and a treatment group of mice, excluding experiments to determine the LD50 (228 mice) and experiments for which no differences between groups were expected (271 mice)40. We performed log-rank tests using survival data with sacrifice thresholds varying from 1 to 1%, from 5 to 30%. The number of p values < 0.05 was used to run power calculation using Eq. (2). Each experiment was treated independently. The simulation-based calculation was carried out using the data from all the experiments performed within one specific model of infection. Since power is reflected by the number of significant p values, we created artificial experiments. First, we set the number (N) of mice used per group and chose a type of infection. Second, we randomly selected N mice from the control and the treatment groups. Finally, we compared the newly generated groups by computing p values with the log-rank test. The process was run 5000 times and repeated 20 times for a total of 100,000 tests to derive a theoretical power at N mice. We increased the number of mice in each group and fitted the data with a curve created with scipy 1.9.0 to calculate the number of mice corresponding to a power of 80%. With the number of mice thus obtained, we computed with the same principle 100,000 new p values with variable weight loss thresholds, incremented by 1% from 5 to 30% of weight loss.
Results
Data collection
To assess the relationship between weight loss and outcome of infected mice, and to define whether thresholds leading to euthanasia can be optimized, we performed a retrospective analysis of data recorded during experiments carried out between June 2014 and March 2023. We focused on models of infection used in more than 5 experiments and with over 200 mice. Among 2400 mice, 791 and 1609 mice were used under authorizations covering the years 2014–2018 (cohort 1) and 2018–2023 (cohort 2), respectively. We excluded 43 mice from the analyses because they died after anesthesia (39) or were incorrectly injected with the infectious agent (4) (Fig. 1). The models of infection included (number of experiments, number of mice) systemic infection with Candida albicans (6, 252) and Listeria monocytogenes (39, 1048), and intranasal infection with Streptococcus pneumoniae (32, 721) and H1N1 (19, 336) (Table 1). The infectious doses (median, interquartile range [IQR]) were 1.6 × 105 [0.8–2.0 × 105] colony forming units (CFU) C. albicans, 1.2 × 105 [0.9–1.6 × 105] CFU L. monocytogenes, 5.8 × 104 [0.2–2.9 × 105] CFU S. pneumoniae and 4.0 × 104 [0.2–4.0 × 104] plaque forming units H1N1. We compared groups of control mice (wild-type mice) and experimental mice (with immunomodulatory treatment in wild-type mice, or knockout mice). The mortality rate in the control groups was 40.2% for C. albicans, 67.9% for L. monocytogenes, 71.5% for S. pneumoniae and 51.5% for H1N1 infection. Over the period of data acquisition, the inoculum could vary depending on our working hypothesis. When our objective was to test an intervention likely to increase mortality, we chose a sublethal inoculum in control mice. Conversely, when our objective was to test an intervention likely to reduce mortality, we chose an inoculum that would induce high mortality in the control group.
Weight loss to predict outcome
Taking all the mice together, the proportion of mice that did not survive infection, either because they died or were euthanized, was higher for L. monocytogenes (47.0%) and S. pneumoniae (54.5%) than for C. albicans (34.9%) and H1N1 (39.6%) (Table 1 and Supplementary Fig. 1). The median survival time was 9 days for S. pneumoniae infection. In all models, independently of the conditions applied to the mice (control versus experimental group), weight loss segregated mice that survived from mice that did not survive. Differences were statistically significant as early as one day post-infection with L. monocytogenes and H1N1, and two days post-infection with C. albicans and S. pneumoniae (Fig. 2, left). Accordingly, the mean body weight loss over time showed a clear differentiation between surviving and non-surviving mice (Fig. 2, right). The percentage of initial weight (mean ± standard deviation) at day 6 post-infection reached 74.6 ± 3.2% (n = 7), 74.4 ± 6.9% (n = 66), 72.8 ± 7.9% (n = 51), and 91.9 ± 8.1% (n = 57) in mice dying from L. monocytogenes, H1N1, C. albicans and S. pneumoniae, respectively.
Percent of initial weight of mice infected with C. albicans, L. monocytogenes, S. pneumoniae and H1N1. (A) Day-by-day data were compared with the Kruskal–Wallis test. Each dot represents one mouse. *p < 0.05, ***p < 0.001; ****p < 0.0001 (survivors versus non-survivors at one specific day). (B) Median of percent weight loss with standard deviation as bandwidth. Blue: mice that survived; orange: mice that did not survive. Statistical significance of weight loss over time was computed by mixed effect model (p < 0.001).
To further compare weight loss and survival rate after infection, we performed a mixed-effects analysis to examine the relationship between weight loss and survival rate across multiple time points while considering individual differences among mice. The model was computed from day 0 to day 3 (S. pneumoniae and L. monocytogenes) and to day 5 (C. albicans and H1N1) to include as many data points as possible while minimizing bias due to missing values. Highly statistically significant differences (p < 0.001) were observed between groups of survivors and non-survivors (Fig. 2). Differences were also statistically significant when the mixed-effects analysis was performed using all data points (p < 0.001, data not shown).
Since data were collected from cohorts running from 2014 to 2018 without weight loss limit (cohort 1) and from 2018 to 2023 with specified weight loss leading to the euthanasia (cohort 2) (Supplementary Tables 1 and 2), we wondered whether we introduced a bias towards representing more non-survivors using the second cohort. To clarify this aspect, we compared the mortality rates of the first and second cohorts. The mortality rate was similar and not statistically significant for each of the models of infection (Supplementary Fig. 2). Moreover, the proportion of mice that reached more than 30% weight loss was similar in the cohorts 1 and 2 (Supplementary Table 3).
Importance of weight loss in clinical score to predict outcome
Weight loss (ranked 0–3) is one among other criteria to calculate the clinical score. Indeed, septic mice show external signs of illness affecting their mobility and aspect (behavior, ruffled coat, diarrhea, conjunctivitis) that are recorded on score sheets (Supplementary Tables 1 and 2). So, we could analyze the relationship between clinical scores and weight loss using the data collected in cohort 2 (i.e. without C. albicans) where both parameters were available. As shown in Fig. 3, the clinical score and the percentage of initial weight (reflecting weight loss) inversely correlated for L. monocytogenes (R = − 0.70, p < 0.0001), S. pneumoniae (R = − 0.64, p < 0.0001) and H1N1 (R = − 0.68, p < 0.0001) infections.
Correlation between weight loss and clinical scores. The maximum weight loss was plotted against the highest clinical score for non-survivors from cohort 2. The Pearson correlation coefficient (R) and p values were calculated using pearsonr.
We then further explored the relative contribution of weight loss and clinical score (which includes weight loss) in promoting euthanasia. Table 2 summarizes the data collected from euthanized mice in cohort 2. The proportion of mice reaching the weight loss limit for euthanasia (i.e. 3) while having a clinical score not requiring euthanasia (i.e. < 6) was 13.9%, 0.6% and 11.7% for L. monocytogenes, S. pneumoniae and H1N1 infection, respectively. The proportion of mice achieving a clinical score requiring euthanasia (i.e. ≥ 6) was much higher, at 86.1%, 99.4% and 88.3% for L. monocytogenes, S. pneumoniae and H1N1, respectively. However, within these groups, it is interesting to note that 54–56% of mice infected by L. monocytogenes and H1N1 also reached the maximum weight loss score of 3 imposing euthanasia. In addition, 43.0%, 95.3% and 42.6% of mice with a clinical score ≥ 6 reached a weight loss score of 2 when infected by L. monocytogenes, S. pneumoniae and H1N1.
Overall, these results indicate that most of the mice infected with L. monocytogenes and H1N1 reached the maximum weight loss threshold earlier or at the same time as the clinical score leading to euthanasia, while the clinical score appears to be reached just before maximum weight loss for S. pneumoniae-infected mice.
Impact of weight loss threshold on the mortality rate
Humane endpoints based on statistical and ethical considerations should quickly relieve the suffering of mice, without euthanizing animals that would otherwise have survived. Hence, we tested whether weight loss thresholds set at 25%, 20%, 15% and 10% increased the mortality rate in the four models of infection.
As expected, the death rate increased as weight loss thresholds were lowered, but from different thresholds onwards (Table 2). In the models of C. albicans and H1N1 infection, using a threshold of 25% increased the number of mice euthanized by 33 and 45 (over 252 and 336 mice), and the death rate from 34.9 to 48.0% and from 39.6 to 53.0% (p < 0.001), respectively. In the case of L. monocytogenes infection, statistical differences were observed from a threshold of 20%, which increased the number of mice euthanized by 79 (over 1048) and the death rate from 47.0 to 54.6% (p < 0.001). Finally, in the model of S. pneumoniae infection, thresholds of 25–15% had no influence on death rate, whereas a threshold of 10% increased the number of euthanized mice by 53 (over 721) and the death rate from 54.5 to 61.9% (p < 0.001). Therefore, the weight loss threshold influences the number of mice euthanized depending on the model of infection (Table 3).
In-house data-based power calculation
Power calculations are conducted to estimate the size of the samples to be used in the experiments. Numerous experimental conditions, including the strain, dose, and route of inoculation of the microorganism, as well as the treatment, genetic background, sex, and age of the mice influence computing power. As a first step, we assessed the power post hoc, based on the number of p < 0.05 reported to the total number of tests performed using data obtained in experiments comparing a control and a treatment group of mice (see “Methods”, “Power analysis”). Each model of infection was evaluated separately, using new survival data obtained with sacrifice thresholds varying from 1 to 1% (Fig. 4). We observed a decline in power from a weight loss of 21% in experiments using L. monocytogenes. In the model of S. pneumoniae infection, power was rather unstable between 22 and 14%. The number of p < 0.05 decreased rapidly after 14%. As anticipated, due to the limited number of experimental data, we did not observe noticeable changes in power in the models of infection with C. albicans and H1N1. More data should be collected to investigate power in these models.
Power analysis using the in-house approach. Power calculation was performed post hoc using the in-house approach (see “Methods”, “Power analysis”). Sacrifice thresholds were set from 1 to 1% to perform log-rank tests, count the number of p < 0.05, and run power calculation using Eq. (2). The orange and blue dotted lines mark the weight loss threshold after which the power decreases for L. monocytogenes and S. pneumoniae infections.
Simulation-based power calculation
Simulation is a powerful tool to increase the number of datapoints without the requirement of additional experimental mice. Hence, we computed experiments involving control and treated mice randomly selected from our dataset. First, we determined the number of mice, between 10 and 400, needed to achieve a power of 80% for a given type of infection (Fig. 5A). Simulation resulted in curves that reached a power of 80% with 35 and 56 mice in models of infection with L. monocytogenes and S. pneumoniae. Simulations with C. albicans and H1N1 were not conclusive due to an insufficient effect size to determine a power of 80% (Fig. 5A). We then selected 35 (L. monocytogenes) and 56 (S. pneumoniae) mice per group to run simulations varying the threshold of sacrifice (Fig. 5B). For L. monocytogenes, the power was stable between 30 and 20% weight loss and increased slightly to 87.3% until a weight loss of 18%. Power decreased rapidly to 40.0% from 13 to 5% weight loss. For S. pneumoniae, the power remained stable up to 13% weight loss and decreased rapidly thereafter, reaching 5.2% at 5% weight loss. Overall, in-house data-based and simulation-based power calculations revealed variability and/or reduction of power as weight loss thresholds approached 20% for S. pneumoniae and L. monocytogenes models.
Simulation-based power calculation. (A) Simulation to determine the number of mice needed to reach a power of 80%. (B) Simulation of power with 35 (L. monocytogenes) and 56 (S. pneumoniae) mice as a function of weight loss.
Discussion
The implementation of appropriate humane endpoints is an ethical foundation for experimental research using animals. We report here the most extensive study, to our knowledge, exploring the relationship between weight loss threshold and outcome of mice used in preclinical models of sepsis. Globally, our data indicate that weight loss is a valuable predictor of mortality, but that specific thresholds should be defined for each model of infection. Hence, weight loss is an objective, easy and powerful measure for generating composite clinical scores to minimize mouse suffering while preserving the statistical power of the experiments.
Our study is unique because it is based on an important data set collected in experiments testing working hypothesis linked to host response to infections. In this sense, it differs from tightly controlled studies specifically designed to test the predictive value of weight loss (with or without other criteria of wellbeing) during infections7,8,9,11,41,42,43,44. We analyzed data accumulated over 10 years, excluding those from experiments with a low number of data points, i.e. one experiment with Citrobacter rodentium (n = 15 mice), one with Staphylococcus aureus (n = 10), two with Pseudomonas aeruginosa (n = 68), seven with Escherichia coli (n = 116) and two with lipopolysaccharide (LPS, n = 37). Even for a specific model of infection, several factors varied including inoculum size, mouse characteristics (strain, sex, age, origin, microbiota, treatment), housing conditions (temperature, humidity, number of mice per cage, enrichment material), as well as animal keepers and experimenters and the criteria used to evaluate animal welfare. On the one hand, these factors may be confounding and influence our analyses. On the other hand, they reflect the diversity of biomedical research and, in our opinion, make this study all the more interesting.
A small number of mice achieved 30% weight loss while surviving (39 out of 2357 mice, 1.7%, Table 1), suggesting that an important weight loss is predictive of poor outcome11. Though, weight loss does not always predict death. Among the 4 models of infections, infections with C. albicans or H1N1 resulted in the greatest weight loss in both survivor and non-survivor groups, while they were the least lethal (35–40% mortality with C. albicans and H1N1 versus 47–54% mortality with L. monocytogenes and S. pneumoniae). This is because acute infections result in more rapid mortality and leave less time for substantial weight loss compared with subacute or chronic infections. Accordingly, mice that did not survive S. pneumoniae infection lost on average 15% of body weight, while those that survived lost 6%. Importantly, for a specific model of infection and according to our working hypothesis, we used an inoculum with mortality rates ranging from 0 to 100% in the control group. Hence, inoculum also has an impact on the timing and trajectory of weight loss.
Considering non-surviving mice in each of the four models of infection, the mean time to death was 8 and 7 days for C. albicans and H1N1 infections, while it was only 4 and 5 days for L. monocytogenes and S. pneumoniae infection (Table 1 and Supplementary Fig. 1). Sixteen of 64 (25.0%) and 17 of 68 (25.0%) mice infected with C. albicans and H1N1 that lost more than 30% of their initial weight at some point during the experiment finally survived. Fixing a maximum weight loss of 20% would have increase the proportion of non-survivors due to increased euthanasia by 24% for C. albicans and 22% for H1N1 infections.
Given our large dataset, we could analyze the effect size by performing post hoc in-house data-based power calculation. Unfortunately, for C. albicans and H1N1 infection, the number of p values calculated was not sufficient to draw a conclusion. For L. monocytogenes and S. pneumoniae, we observed a decrease in power at around 20% weight loss, but also variability in power. This is not surprising since calculations were based on log-rank tests with small samples (n = 7/group on average). To solve this issue, we employed simulation-based power calculation carried out with 100,000 tests instead of around 40 tests for in-house data-based power calculation, which greatly reduced power variability. In simulation-based power calculation, power decreased sharply at around 13% weight loss in models of infection with S. pneumoniae and L. monocytogenes. Consequently, the introduction of stringent thresholds would have important consequences on designing the size of the experimental groups.
Weight loss was not reflective of mortality in a study assessing novel methods to evaluate the outcome of mice subjected to intranasal infection with Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, Enterobacter cloacae and Escherichia coli11. Instead, the measurement of internal and external temperature using microchips and infrared thermometer revealed that internal temperature more precisely predicted mortality. Taken together with our data, these observations comfort the idea that weight loss recommendations for euthanasia cannot be universal and that a threshold of 20%, as is often used, is not applicable to any type of infection13. For instance, we now use a weight loss of ≥ 20%, instead of ≥ 30%, as a humane endpoint for S. pneumoniae infected mice. On the contrary, we increased the weight loss limit used to euthanize L. monocytogenes-infected mice from ≥ 20 to ≥ 25%.
Our composite clinical scores include weight loss in addition to mobility and external aspect (supplementary Tables 1 and 2). If we focus on euthanized animals from cohort 2, 68.2% of Listeria and 67.6% of H1N1-infected mice were euthanized reaching the weight loss humane endpoint (Table 2). On the contrary, 98.5% of S. pneumoniae-infected mice were euthanized because of their clinical score, while weight loss was almost invariably scored at 2. These observations support further our decision to reduce the weight loss limit leading to euthanasia of S. pneumoniae-infected mice. All these considerations imply that changes in the weight loss threshold are not trivial, and that the question of whether the clinical score requires euthanasia before a fixed weight loss limit must be assessed on a case-by-case basis according to the score sheet criteria (number of dimensions, scoring system, weight of each criterion). If the weight loss thresholds for euthanasia are very strict, then weight loss would be the main criterion for stopping the experiment. On the contrary, if the weight loss thresholds are permissive, the weight loss limit will probably never be reached in sacrificed mice. Based on our analyses, we recommend that thresholds should be defined in a model-dependent manner. Though, we recognize that it is challenging to collect a large amount of data points to perform statistical and power calculations. While using weight loss as a humane endpoint is debated11,12, weight loss is a valuable indicator of the health status of mice when combined with other criteria such as temperature, appearance, and behavior.
Several studies reported humane endpoints in preclinical mouse models of infection and sepsis. Body temperature was a predictor of outcome of bacterial and fungal infections, as well as cecal ligation and puncture-induced sepsis11,42,45,46,47. However, there are important variations in temperature cut-off values between studies10. A sepsis severity score based on grading appearance (smooth or ruffled fur), eye aspect (normal, with secretion), activity (active, lethargic, moribund), consciousness (alert, sleepy) and respiration (normal or slow breaths) had a good predictive value in mice infected intratracheally with K. pneumoniae48. A similar score based on subjective parameters (consciousness, activity, behavior, response to stimuli and breathing) was improved by adding objectives values (glycemia, temperature, body weight). The combinatorial score achieved high sensitivity and specificity for the early detection of sepsis in mice injected intraperitoneally with fecal solution43. In a machine learning approach using data from mice challenged with LPS, scores of sickness and temperature improved the accuracy and performance of the prediction model10. Given that the host response to live microorganisms differs from that to toxins such as LPS, a similar learning approach using data from infected mice could prove instructive. Generally, the scores reported in the literature have been established as part of specific studies. It is assumed that humane endpoints may vary according to the infectious model, disease intensity, the treatments administrated to the mice, and many additional factors including age, sex, and environmental conditions. Therefore, the robustness of the scores must be verified in independent experimental contexts so that they can be used as humane endpoint for rapidly terminating sepsis experiments while ensuring that the scientific objectives are achieved.
Our study has several limitations. We excluded from our analysis experiments with few data points, which could have been informative. We did not have groups of uninfected mice for each model to adhere to the 3Rs, but that raises the question of the influence of anesthesia and mouse handling on weight loss. In response, mice anesthetized with ketamine and xylazine and receiving a non-lethal inoculum of H1N1 did not lose weight (100.2% ± 2.7% of initial weight 24 h after treatment). Moreover, in all experiments, the control and treated groups were manipulated in the same way (injection of diluent for the control group). These observations suggest that, apart from deleterious infections, handling procedures had a relatively minor impact on weight loss. We included data collected during experiments carried out with different parameters, including the parameters used to monitor the wellbeing of the mice and to terminate the experiments. It is possible that we have overestimated the weight limits associated with mortality. Nonetheless, we believe that the diversity of our data reflects the situation encountered in many laboratories and is closer to reality than the studies specifically designed to test the predictive value of a predefined humane endpoint.
To conclude, we report the largest study analyzing weight loss in models of bacterial, viral, and fungal infections in real world settings. Our data support the idea that weight loss thresholds used as humane endpoint should be adapted to the experimental model used. Whereas preclinical mouse models remain the best way to decipher the complex biological processes underlying sepsis and to test new treatment options, they must be conducted with strict ethical considerations in mind. From this point of view, our calculations allowed us to adjust the weight loss thresholds to minimize mice suffering without increasing unreasonably the size of the experimental groups. Thus, we recommend an infection-model-dependent evaluation of weight loss thresholds used in preclinical models of sepsis.
Data availability
All data generated or analyzed during this study are included in this article and its supplementary information files or have been deposited at https://github.com/mbrochut/weight_loss.
Change history
24 October 2024
A Correction to this paper has been published: https://doi.org/10.1038/s41598-024-76906-9
Abbreviations
- CFU:
-
Colony forming unit
- IV:
-
Influenza virus
- LD50 :
-
Lethal dose 50
- LPS:
-
Lipopolysaccharide
References
Hickman, D. L., Johnson, J., Vemulapalli, T. H., Crisler, J. R. & Shepherd, R. Commonly used animal models. Principles Animal Res. Graduate Undergraduate Students. https://doi.org/10.1016/B978-0-12-802151-4.00007-4 (2017).
Wang, N., Lu, Y., Zheng, J. & Liu, X. Of mice and men: Laboratory murine models for recapitulating the immunosuppression of human sepsis. Front. Immunol. 13, 956448. https://doi.org/10.3389/fimmu.2022.956448 (2022).
Demers, G. et al. Animal research. Harmonization of animal care and use guidance. Science 312, 700–701. https://doi.org/10.1126/science.1124036 (2006).
Morton, D. B. A systematic approach for establishing humane endpoints. ILAR J 41, 80–86. https://doi.org/10.1093/ilar.41.2.80 (2000).
Fox, J. G. Laboratory Animal Medicine (Elsevier, 2015).
Workman, P. et al. Guidelines for the welfare and use of animals in cancer research. Br. J. Cancer 102, 1555–1577. https://doi.org/10.1038/sj.bjc.6605642 (2010).
Franco, N. H., Correia-Neves, M. & Olsson, I. A. How “humane” is your endpoint? Refining the science-driven approach for termination of animal studies of chronic infection. PLoS Pathog. 8, e1002399. https://doi.org/10.1371/journal.ppat.1002399 (2012).
Hankenson, F. C. et al. Weight loss and reduced body temperature determine humane endpoints in a mouse model of ocular herpesvirus infection. J. Am. Assoc. Lab. Anim. Sci. 52, 277–285 (2013).
Shrum, B. et al. A robust scoring system to evaluate sepsis severity in an animal model. BMC Res. Notes 7, 233. https://doi.org/10.1186/1756-0500-7-233 (2014).
Mei, J. et al. Refining humane endpoints in mouse models of disease by systematic review and machine learning-based endpoint definition. ALTEX 36, 555–571. https://doi.org/10.14573/altex.1812231 (2019).
Dudis, R. S. et al. Longitudinal temperature measurement can determine humane endpoints in BALB/c mouse models of ESKAPEE infection. Virulence 14, 2186331. https://doi.org/10.1080/21505594.2023.2186331 (2023).
Talbot, S. R. et al. Robustness of a multivariate composite score when evaluating distress of animal models for gastrointestinal diseases. Sci. Rep. 13, 2605. https://doi.org/10.1038/s41598-023-29623-8 (2023).
Talbot, S. R. et al. Defining body-weight reduction as a humane endpoint: A critical appraisal. Lab. Animals 54, 99–110 (2020).
Cajander, S. et al. Profiling the dysregulated immune response in sepsis: Overcoming challenges to achieve the goal of precision medicine. Lancet Respir. Med. https://doi.org/10.1016/S2213-2600(23)00330-2 (2023).
Singer, M. et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 315, 801–810. https://doi.org/10.1001/jama.2016.0287 (2016).
Slim, M. A. et al. Towards personalized medicine: A scoping review of immunotherapy in sepsis. Crit. Care 28, 183. https://doi.org/10.1186/s13054-024-04964-6 (2024).
Schlapbach, L. J. et al. Swiss Sepsis National Action Plan: A coordinated national action plan to stop sepsis-related preventable deaths and to improve the support of people affected by sepsis in Switzerland. Front. Med. (Lausanne) 10, 1114546. https://doi.org/10.3389/fmed.2023.1114546 (2023).
Ciarlo, E., Savva, A. & Roger, T. Epigenetics in sepsis: Targeting histone deacetylases. Int. J. Antimicrobial Agents 42(Suppl), S8-12. https://doi.org/10.1016/j.ijantimicag.2013.04.004 (2013).
Schrijver, I. T. et al. High levels of monocytic myeloid-derived suppressor cells are associated with favorable outcome in patients with pneumonia and sepsis with multi-organ failure. Intensive Care Med. Exp. 10, 5. https://doi.org/10.1186/s40635-022-00431-0 (2022).
Schrijver, I. T. et al. Myeloid-derived suppressor-like cells as a prognostic marker in critically ill patients: Insights from experimental endotoxemia and intensive care patients. Cells. https://doi.org/10.3390/cells13040314 (2024).
Ouertatani-Sakouhi, H. et al. A new class of isothiocyanate-based irreversible inhibitors of macrophage migration inhibitory factor. Biochemistry 48, 9858–9870. https://doi.org/10.1021/bi900957e (2009).
Ouertatani-Sakouhi, H. et al. Identification and characterization of novel classes of macrophage migration inhibitory factor (MIF) inhibitors with distinct mechanisms of action. J. Biol. Chem. 285, 26581–26598. https://doi.org/10.1074/jbc.M110.113951 (2010).
Kerschbaumer, R. J. et al. Neutralization of macrophage migration inhibitory factor (MIF) by fully human antibodies correlates with their specificity for the beta-sheet structure of MIF. J. Biol. Chem. 287, 7446–7455. https://doi.org/10.1074/jbc.M111.329664 (2012).
Meller, S. et al. T(H)17 cells promote microbial killing and innate immune sensing of DNA via interleukin 26. Nat. Immunol. 16, 970–979. https://doi.org/10.1038/ni.3211 (2015).
Ciarlo, E. et al. Impact of the microbial derived short chain fatty acid propionate on host susceptibility to bacterial and fungal infections in vivo. Sci. Rep. 6, 37944. https://doi.org/10.1038/srep37944 (2016).
Ciarlo, E. & Roger, T. Screening the impact of sirtuin inhibitors on inflammatory and innate immune responses of macrophages and in a mouse model of endotoxic shock. Methods Mol. Biol. 1436, 313–334. https://doi.org/10.1007/978-1-4939-3667-0_21 (2016).
Roger, T. et al. High expression levels of macrophage migration inhibitory factor sustain the innate immune responses of neonates. Proc. Natl. Acad. Sci. USA 113, E997-1005. https://doi.org/10.1073/pnas.1514018113 (2016).
Ciarlo, E. et al. Sirtuin 3 deficiency does not alter host defenses against bacterial and fungal infections. Sci. Rep. 7, 3853. https://doi.org/10.1038/s41598-017-04263-x (2017).
Ciarlo, E. et al. Sirtuin 2 deficiency increases bacterial phagocytosis by macrophages and protects from chronic staphylococcal infection. Front. Immunol. 8, 1037. https://doi.org/10.3389/fimmu.2017.01037 (2017).
Heulot, M. et al. The anticancer peptide TAT-RasGAP(317–326) exerts broad antimicrobial activity. Front. Microbiol. 8, 994. https://doi.org/10.3389/fmicb.2017.00994 (2017).
Heinonen, T. et al. Sirtuin 5 deficiency does not compromise innate immune responses to bacterial infections. Front. Immunol. 9, 2675. https://doi.org/10.3389/fimmu.2018.02675 (2018).
Heinonen, T., Ciarlo, E., Le Roy, D. & Roger, T. Impact of the dual deletion of the mitochondrial sirtuins SIRT3 and SIRT5 on anti-microbial host defenses. Front. Immunol. 10, 2341. https://doi.org/10.3389/fimmu.2019.02341 (2019).
Heinonen, T. et al. Dual deletion of the sirtuins SIRT2 and SIRT3 impacts on metabolism and inflammatory responses of macrophages and protects from endotoxemia. Front. Immunol. 10, 2713. https://doi.org/10.3389/fimmu.2019.02713 (2019).
Ciarlo, E. et al. Trained immunity confers broad-spectrum protection against bacterial infections. J. Infect. Dis. 222, 1869–1881. https://doi.org/10.1093/infdis/jiz692 (2020).
Katzmarski, N. et al. Transmission of trained immunity and heterologous resistance to infections across generations. Nat. Immunol. 22, 1382–1390. https://doi.org/10.1038/s41590-021-01052-7 (2021).
Theroude, C. et al. Trained immunity confers prolonged protection from listeriosis. Front. Immunol. 12, 723393. https://doi.org/10.3389/fimmu.2021.723393 (2021).
Kleinbaum, D. G. & Klein, M. Survival Analysis a Self-Learning Text (Springer, 1996).
Greenland, S. et al. Statistical tests, P values, confidence intervals, and power: A guide to misinterpretations. Eur. J. Epidemiol. 31, 337–350. https://doi.org/10.1007/s10654-016-0149-3 (2016).
Lehmann, E. L., Romano, J. P. & Casella, G. Testing Statistical Hypotheses Vol. 3 (Springer, 1986).
White, J. W., Rassweiler, A., Samhouri, J. F., Stier, A. C. & White, C. Ecologists should not use statistical significance tests to interpret simulation model results. Oikos 123, 385–388. https://doi.org/10.1111/j.1600-0706.2013.01073.x (2013).
Trammell, R. A. & Toth, L. A. Markers for predicting death as an outcome for mice used in infectious disease research. Comparative Med. 61, 492–498 (2011).
Mai, S. H. C. et al. Body temperature and mouse scoring systems as surrogate markers of death in cecal ligation and puncture sepsis. Intensive Care Med. Exp. 6, 20. https://doi.org/10.1186/s40635-018-0184-3 (2018).
Sulzbacher, M. M. et al. Adapted murine sepsis score: Improving the research in experimental sepsis mouse model. Biomed. Res. Int. 2022, 5700853. https://doi.org/10.1155/2022/5700853 (2022).
Drechsler, S. et al. Why do they die? Comparison of selected aspects of organ injury and dysfunction in mice surviving and dying in acute abdominal sepsis. Intensive Care Med. Exp. 3, 48. https://doi.org/10.1186/s40635-015-0048-z (2015).
Warn, P. A. et al. Infrared body temperature measurement of mice as an early predictor of death in experimental fungal infections. Lab. Anim. 37, 126–131. https://doi.org/10.1258/00236770360563769 (2003).
Adamson, T. W., Diaz-Arevalo, D., Gonzalez, T. M., Liu, X. & Kalkum, M. Hypothermic endpoint for an intranasal invasive pulmonary aspergillosis mouse model. Comp. Med. 63, 477–481 (2013).
Nemzek, J. A., Xiao, H. Y., Minard, A. E., Bolgos, G. L. & Remick, D. G. Humane endpoints in shock research. Shock 21, 17–25. https://doi.org/10.1097/01.shk.0000101667.49265.fd (2004).
Goncalves, M. C., Horewicz, V. V., Luckemeyer, D. D., Prudente, A. S. & Assreuy, J. Experimental sepsis severity score associated to mortality and bacterial spreading is related to bacterial load and inflammatory profile of different tissues. Inflammation 40, 1553–1565. https://doi.org/10.1007/s10753-017-0596-3 (2017).
Acknowledgements
We thank all the people that participated to in vivo experiments from which the results were analyzed in this study, and Frédéric Schütz (Center for Integrative Genomics, University of Lausanne and Swiss Institute of Bioinformatics, Lausanne, Switzerland) for discussions and advise on data analyses.
Funding
TR was supported by Grants from the Swiss National Science Foundation (SNSF, Grant number 310030_207418), Promex Stiftung für die Forschung (Geneva, Switzerland), the Horizon 2020 Grant ImmunoSep (number 847422) and HDM-FUN (number 847507). TH received a scholarship from the Société Académique Vaudoise (Lausanne, Switzerland).
Author information
Authors and Affiliations
Contributions
Conceptualization, MB, TH and TR; Formal analysis, MB and TR; Funding acquisition, TR; Investigation, MB, TH, TS, CG, DLR, and TR; Methodology, MB and TR; Project administration, TR; Resources, TR; Software, MB; Supervision, TR; Visualization, MB and TR; Writing – original draft, MB and TR; Writing – review & editing, MB, TH, TS, CG, DLR, and TR. All the authors revised the manuscript. All authors agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error in the Funding section. Full information regarding the correction made can be found in the correction for this Article.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brochut, M., Heinonen, T., Snäkä, T. et al. Using weight loss to predict outcome and define a humane endpoint in preclinical sepsis studies. Sci Rep 14, 21150 (2024). https://doi.org/10.1038/s41598-024-72039-1
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-024-72039-1