Abstract
Ischemic heart disease, driven largely by myocardial infarction (MI), remains the leading cause of mortality and morbidity. Although early suppression of post-MI inflammation improves outcomes, current therapies have limited efficacy. Here we show that the cellular inhibitor of apoptosis 2 (cIAP2), a regulator of cell death, is upregulated after MI and promotes acute inflammation and cardiac injury. Global deletion of cIAP2, or its loss through bone marrow transfer, reduced inflammatory injury and cardiac dysfunction after MI, indicating that the cardioprotective effect of cIAP2 deficiency is primarily mediated by the hematopoietic compartment. Reduced cardiac inflammation was associated with decreased splenic myeloid cell numbers due to increased cell death and elevated expression of the death-inducing factors TRAIL and TRAIL-R2/DR5. Pharmacologic degradation of cIAP proteins after MI using Smac mimetics similarly reduced cardiac inflammation and protected against injury. Together, these findings identify cIAP2 as a key hematopoietic cell-expressed regulator of survival and inflammation and support its inhibition as a potential immunotherapeutic strategy for MI.
Similar content being viewed by others
Main
Although incidence rates of MI are stabilizing, and survival after acute MI has improved, MI continues to be a major cause of significant morbidity and progression toward heart failure1,2. An effective early inflammatory response is required to promote clearance of dead cells from infarcted tissue and stimulate robust wound healing3, but limiting the intensity and duration of this response is beneficial in mitigating the development of chronic disease4,5. Success of this strategy has been suggested by encouraging results from clinical trials evaluating chronic inhibition of key inflammatory cytokines such as interleukin-1 beta (IL-1β) and interleukin-6 (IL-6)6,7 or broader anti-inflammatory agents such as colchicine that interfere with inflammasome assembly8. However, these molecules have broad, multifaceted biological functions, and long-term inhibition of their activities can lead to adverse effects—notably, elevated risk of infection. Furthermore, high costs, route of administration and risk of patient non-compliance pose obstacles to achieving effective long-term therapeutic benefit. Identification of inflammatory targets that possess focused activity more acutely after MI may lend precision while limiting risks and costs of chronic long-term therapy.
Ischemia triggers cardiomyocyte death and the rapid release of multiple danger signals that drive potent immune inflammatory responses through the activation of cytokines, notably members of the IL-1 or tumor necrosis factor (TNF) families and innate pattern recognition receptors (PRRs) such as Toll-like receptors (TLRs)9,10,11. Inflammatory signaling pathways are activated in a conserved manner by multiple PRRs or cytokines, and many of these factors have been implicated as contributors to post-MI pathology. For example, previous work from our laboratory and others using mouse models employing germline knockout of TNF and TLR signaling, including Tnfa−/−, Tlr4−/−, Myd88−/− (a TLR adaptor protein) and Irak4−/− (IL-1/Toll ligand receptor activated kinase 4) demonstrated reduced cardiac injury after MI12,13,14. We hypothesized that identifying common, nodal regulators of these networks may uncover potential targets for inhibition of inflammation. Therefore, we searched for candidate molecules known to participate in their signal transduction.
Recently, employing an aptamer-based proteomics array, we identified in serum from a clinical cohort of patients with acute heart failure the distinct upregulation of cIAP2/baculoviral IAP repeat-containing 3 (BIRC3) that was consistent with elevation of standard cardiomyopathy biomarkers, including N-terminal prohormone of brain natriuretic peptide (NT-proBNP) and cardiac troponin T (cTnT). This was intriguing as cIAP2 upregulation is principally known as a cancer-associated event, and the protein was not known to be a cell-secreted factor. We subsequently identified Birc3 as an early upregulated mRNA from extracted publicly available GeoSET data of mouse left ventricular infarct tissue15, coinciding with upregulation of a broad array of proinflammatory gene products known to be pathogenic in MI. cIAP2 is a RING (really interesting gene)-type E3 ubiquitin ligase, which is a closely related but distinct enzyme from cIAP1 (Birc2). cIAP proteins participate in signal transduction from PRR and TNF family cytokine receptors, where they participate in the regulation of cellular survival and differentiation as well as the coordination of inflammatory responses16,17,18. After TNFR ligation, cIAP proteins are recruited by and interact with TNF receptor-activated factors (TRAF1 and TRAF2) and ubiquitinate key downstream kinases, including receptor-interacting protein kinase (RIPK)1 and NF-kappa B-inducing kinase (NIK) 19. Ubiquitinated RIPK1 controls assembly of TNFR-stimulated death-inducing signaling complexes (DISCs) to promote cell survival20. Ubiquitination of NIK promotes its degradation, thus preventing cleavage and activation of NF-κB2 (p100/52) to limit non-canonical NF-κB signal transduction21. cIAP proteins also interact with downstream ubiquitin ligases TRAF3 and TRAF6 after exposure to PRR agonists or TNF family cytokines22,23,24,25 and, in turn, modulate NF-κB and MAPK signaling.
The pro-survival and inflammation modulatory effects of cIAP proteins manifest themselves in various disease states, most prominently in several cancers where they are upregulated and act as cooperating oncogenes26. Although they possess high homology, expression of cIAP1 and cIAP2 is often distinct, where tissue distribution varies and cIAP2 has been shown to be uniquely regulated by NF-κB or stress-dependent stimuli27. Control of cIAP protein expression is mediated by the second mitochondrial-derived activator of caspases (Smac)/Diablo protein28. Peptido-mimics of Smac (Smac mimetics) are highly selective inhibitor of apoptosis (IAP) antagonists that promote protein degradation and sensitize cells to TNF family-dependent apoptosis29. Smac mimetics are under evaluation for treatment of various cancers and exhibit a good safety profile to date30,31,32. Here we report the discovery of cIAP2 upregulation after MI, where it differentially contributes to MI pathology relative to cIAP1. We further evaluated the source of this activity and found that hematopoietic expression of cIAP2 drives inflammatory response and, in turn, can be controlled using Smac mimetic compounds.
Results
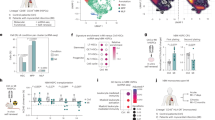
An unbiased proteomics screen of plasma samples from patients with acute ischemic heart failure revealed increased levels of cIAP2 and distantly related BIRC5 (Survivin) but not cIAP1 (Fig. 1a). We next identified that cIAP2 levels were increased in plasma from patients with acute MI compared to those with stable coronary artery disease (CAD) (Fig. 1b). cIAP2 was acutely increased in both human and mouse hearts after acute MI (Fig. 1c,d). Analysis of published microarray data from mouse heart subjected to ischemic infarction (GSE4648)15 showed a distinct expression pattern of cIAP2 relative to other BIRC protein members and consistently aligned with expression of other myeloid inflammatory gene transcripts acutely after MI (Extended Data Fig. 1a–e). Several cIAP2-associated Gene Ontogeny pathways were also upregulated (Extended Data Fig. 1f). In addition, knockout of Nod1 (nucleotide-binding oligomerization domain protein 1), a microbial PRR known to recruit and activate cIAP1/cIAP2 (ref. 14), resulted in improved MI outcomes (Extended Data Fig. 1g), further implicating cIAP proteins as early mediators of injury and inflammation. Knockout of Tnfrsf1a (TNFR1/p55), Tlr4 or Nod1 reduced protein expression of cIAP1 and cIAP2 as well as that of inflammatory factors RIPK1, IRAK4 and TRAF6 after MI (Extended Data Fig. 1h).
cIAP2 is upregulated and participates during cardiac injury after MI. a, SOMAscan measurement of cIAP2, Survivin (BIRC5), cardiac Troponin T (cTNT) and N-Terminal pro-B-type Natriuretic Peptide (NT-proBNP) plasma protein samples from acute heart failure patients (n = 7 measured at admission and D3 and D30 post-admission) and healthy controls (n = 10). b, ELISA of serum cIAP2 from percutaneous coronary intervention patients. Acute MI (n = 9) and CAD (n = 12). c, Immunoblot of human heart tissue lysates probed for IAP proteins cIAP2, cIAP1 and XIAP from healthy or ischemic cardiomyopathy patients. Vinculin was probed as an expression control. d, Immunoblot of cIAP1 and 2 isolated from mouse LV tissue lysates D1, D3 and D7 post-MI. β-tubulin served as an expression control. Immunoblot representative of two experiments. e, Masson’s trichrome stained mouse midventricular heart sections (5 μm thickness) D28 post-MI. Left: wildtype C57Bl/6J (n = 7). Middle: CIap1−/− (n = 6). Right: CIap2−/− (n =7). Scale bars, 1 mm. LV scar area (collagen area − stained blue); morphometry of two representative slices per animal. f, Capillary density estimated by number of isolectin B4-positive (green) structures counted in LV MI border zone. Nuclei (DAPI) stained blue. Scale bars, 50 μm (n = 4 per treatment group). g, LV ejection fraction 28 days post-MI presented as % reduction from baseline (pre-MI) echocardiographic measurement. WT, n = 10; CIap1−/−, n = 6; CIap2−/−, n = 10. h, Heart and lung weights normalized to tibia length measured 4 weeks post-MI. Cont, n = 6 (WT), 4 (CIap1−/−) and 5 (CIap2−/−); MI-treated, n = 15 (WT), 10 (CIap1−/−) and 10 (CIap2−/−). i, Female mice: LV Ejection fraction. Left: WT (n = 8), CIap2−/− (n = 7). Middle: heart weight:tibia length; sham-operated control WT (n = 3), CIap2−/− (n = 3); MI, WT (n = 7), CIap2−/− (n = 7). Right: lung weight:tibia length; sham-operated control WT (n = 3), CIap2−/− (n = 3); MI WT (n = 7), CIap2−/− (n = 7). j, LV MI border zone cell death (TUNEL-positive (red) staining) in 400× magnified 5 μm heart sections. Nuclei (DAPI) stained blue. Triplicate fields counted per study specimen. Scale bar, 50 μm; n = 8 (WT MI) and 7 (CIap2−/− MI), respectively. Mouse data are aggregate from three experiments (male studies) or two experiments (female studies), respectively. Error bars, mean ± standard deviation (s.d.); P values were calculated using two-sided t-test (b,f,j), one-way ANOVA with Dunnett’s post-hoc test (a), one-way ANOVA with Bonferroni’s correction (e) and two-way ANOVA with Bonferroni’s correction (g,h,i). RFU, relative fluorescent units; Cont, control; D, day; HF, heart failure; ICM, ischemic cardiomyopathy; LV, left ventricular.
The role of cIAP2 after MI was evaluated by comparing responses from cIAP2 germline knockout mice (CIap2−/−)27 with those of CIap1−/−33 or C57Bl/6 (wild-type (WT)) mice subjected to coronary ligation. Histology of infarcted heart tissue demonstrated markedly reduced scar area in CIap2−/− mice relative to CIap1−/− and WT animals (Fig. 1e), whereas vascular immunohistochemistry demonstrated increased vascularization of injured tissue (Fig. 1f). Left ventricular end-diastolic and end-systolic dimensions were better preserved (Extended Data Fig. 2a); left ventricular ejection fraction was less severely impaired (Fig. 1g); and heart and lung weight/tibila ratios were reduced (Fig. 1h). No sex-related differences were detected in the post-MI responses (Fig. 1i). Furthermore, CIap2−/− mice showed reduced cardiomyocyte death in the peri-infarct border region acutely after MI (Fig. 1j), accompanied by reduced expression of apoptotic proteins annexin A1, cleaved poly-adenoribosyl polymerase (PARP) and cleaved caspase-8 (Extended Data Fig. 2b). At subsequent intermediate timepoints, we observed reduced transcription of factors associated with adverse remodeling (for example, Nppb (brain natriuretic peptide), Myh7 (myosin heavy chain isoform 7), Tnfa and Tgb1 in CIap2−/− mice relative to WT controls (Extended Data Fig. 2c). Finally, expression of the alarmin high mobility group box 1 (HMGB1) and remodeling cytokine transforming growth factor beta 1 (TGFβ1) were reduced 28 days after infarct in CIap2−/− mice (Extended Data Fig. 2d). Overall, these findings demonstrate that cIAP2 expression contributes to enhanced cardiac injury after MI.
Hematopoietic-expressed cIAP2 drives post-MI inflammation
We assessed the requirement of cIAP2 acutely after MI for cardiac-specific immune cell infiltration and function. Heart histology showed reduced cellular inflammation in left ventricular myocardium of CIap2−/− mice 3 days after MI (Fig. 2a). Cardiac inflammatory responses, demonstrated by left ventricular infiltrating neutrophils, Ly6Chi monocytes and dendritic cells, were equivalent between WT and CIap2−/− mice 1 day after MI (Fig. 2b); however, cellular infiltration became significantly and sustainably reduced in CIap2−/− mice from day 3 after MI and as late as day 28 (Fig. 2b,c). Reduced infiltration, along with decreased transcription of inflammation-promoting chemokines Cxcl1 and Ccl2 lf ventricular tissue of CIap2−/− mice (Fig. 2d), indicated establishment of an attenuated inflammatory microenvironment. We evaluated the immune inflammatory role of cIAP2 by generating bone marrow chimeric mice to restrict cIAP2 deficiency to hematopoietic cells (Fig. 2e). Consistent with our findings in germline CIap2−/− mice, CIap2−/− chimeras showed reduced left ventricular scarring and improved ejection fraction at 28 days after MI (Fig. 2f,g). By contrast, CIap1−/− chimeras demonstrated increased remodeling and worsened ejection fraction when compared to WT chimeric mice (Fig. 2g), supporting the hypothesis that cIAP2 plays a more prominent role in contributing to cardiomyopathy after MI. Although heart weights were relatively unchanged, CIap2−/− chimeric lung weights were decreased relative to WT (Extended Data Fig. 3a), and CIap2−/− chimeras demonstrated improved capillary density (Extended Data Fig. 3b). Cardiac immune infiltration was significantly reduced in CIap2−/− chimeras (Extended Data Fig. 3c). In CIap2−/− chimeric spleens, expression of immune regulator IL-10 was increased, whereas proinflammatory IL-23 was reduced (Extended Data Fig. 3d). We observed concurrent reduction of cardiac myeloid cells and lymphocytes, notably IL-17-expressing T cells that promote myocardial fibrosis and inflammation34 (Extended Data Fig. 3e). Reverse chimeric mice (WT hematopoietic cells, CIap2−/− resident tissues (Fig. 2h)) showed markedly increased scarring and reduced ejection fraction (Fig. 2i,j) as well as worsened cardiac remodeling, reduced vascularization and immune inflammation after MI (Extended Data Fig. 4). Collectively, these findings support the concept that the net cardioprotective effect of cIAP2 deficiency is principally caused by a reduction of hematopoietic-derived immune inflammation.
cIAP2+ hematopoietic cells promote injury and inflammation after MI. a, Inflammatory cell infiltrate 3 days post-MI in WT or CIap2−/− mouse hearts. Full thickness 12.5× magnification; Scale bar, 1mm. Insets: 100× images. Imaging representative of three mice per treatment. b,c, Flow cytometric quantitation of LV inflammatory cellular infiltrate (neutrophils, Ly6Chi monocytes and CD11b+ dendritic cells) at (b) days 1, 3 and 7 and (c) day 28 post-MI. Controls: D1 (n = 3); D3/7/28 (n = 4). WT MI: D1 (n = 3); D3/7/28 (n = 6–9). CIap2−/−: D1 (n = 3); D3/7/28 (n = 5–9). d, Quantitative PCR of Cxcl1 and Ccl2 (neutrophil and monocyte recruitment factors) measured in hearts of WT and CIap2−/− mice 7 days post-MI; n = 3 per group; transcripts normalized to β-actin. e, Generation of WT (CD45.1) recipient bone marrow chimeric mice. f, LV scar area (blue) measured 28 days post-MI (n = 6 from each treatment group). Scale bar, 1 mm. g, LV ejection fraction from WT and CIap2−/− and CIap1−/−-chimeric mice 28 days post-MI. Controls: WT and CIap2−/− (n = 4); CIap1−/− (n = 2). MI: WT and CIap2−/− (n = 17); CIap1−/− (n = 4). h, Generation of reverse bone marrow chimeric mice (WT bone marrow donor to either WT or CIap2−/− recipient mice). i, LV scar area from reverse chimeric mice; n = 5 per treatment. Scale bar, 1mm. j, LV ejection fraction from control or MI-treated WT recipient and CIap2−/− recipient mice. Controls: WT and CIap2−/− (n = 2). MI-treated, n = 7 per group. Error bars denote mean ± s.d.; P values were calculated by one-way ANOVA with Bonferroni’s correction (b,c,g,j), two-sided t-test (d,f,i). BM, bone marrow; BMT, bone marrow transplanted; i.v., intravenous.
In patients who suffered recent MI, cIAP2 protein was upregulated in blood leukocyte isolates (Extended Data Fig. 5a, upper panel); similarly, cIAP2 protein levels were increased in mouse peripheral blood leukocytes 7 days after surgical MI (Extended Data Fig. 5a, lower panel). Leukocyte-isolated cIAP2 levels were higher in acute MI patient samples when compared to those with stable CAD, whereas cIAP1 levels were unchanged (Extended Data Fig. 5b). In the mouse model, we observed during the acute injury phase that CIap2−/− mice exhibited significantly decreased circulating myeloid (CD11b+) cells but not lymphocytes (B or T cells), a deficit that widened by day 3 after injury (Extended Data Fig. 5c–e). We, therefore, investigated the hypothesis that the origin of this suppressed inflammatory response was an inhibition of myeloid cell production from hematopoietic producer sites in the spleen and bone marrow. Analysis of spleen and bone marrow cell numbers indicated that, in the early acute phase (24 hours) after MI, there was a net reduction in CD11b+ populations, which corresponded to increased infiltration to the heart (Figs. 2a and 3a,c). Although cell counts recovered progressively at day 3 after MI in both spleen and bone marrow, there was a protracted recovery in the spleen for CIap2−/− mice relative to WT (Fig. 3a), which contrasted with the apparent full recovery of myeloid counts in the bone marrow (Fig. 3c). Overall, leukocytes were completely restored in both spleen and bone marrow of WT and CIap2−/− mice by day 28 after MI (Fig. 3b,d), suggesting that the impaired production of myeloid cells by CIap2−/− mice was related to the inflammatory state after injury.
Increased TRAIL and DR5 expression in CIap2−/− splenocytes after MI. a, Left: flow cytometric quantitation of myeloid (CD11b+) cells at D3 post-MI spleen samples from WT and CIap2−/− mice. Right: dot plots correspond to representative D1 and D3 post-MI samples, representative of aggregate data from three experiments. b, Quantitation of splenic myeloid (CD11b+) cells D28 post-MI; (controls n = 3; MI, WT n = 10; CIap2−/− n = 7). c, Left: flow cytometry of myeloid (CD11b+) cells at D3. Right: dot plots correspond to representative D1 and D3 post-MI samples. d, Bone marrow myeloid cells measured D28 post-MI from WT and CIap2−/− mice. e, Flow cytometry of spleen neutrophil and Ly6Chi monocytes D7 post-MI of WT (C57Bl/6) or CIap2−/− mice; (controls n = 3; WT n = 7; CIap2−/− n = 5 mice per group. f, Spleen TUNEL+ staining D3 post-MI; (controls n = 4; WT MI n = 7; CIap2−/− MI n = 5. 400× magnification; scale bar, 50 μm. g, Increased TRAIL+ staining of splenocytes from CIap2−/− mice D3 post-MI. Low to undetectable TRAIL staining in matched heart samples. Scale bar, 50 μm; (control n = 4; WT n = 7; CIap2−/− n = 5). h, Left: immunoblotting of spleen DR5 (TRAIL-R2) and TNFR1 expression D3 post-MI. Image representative of two similar immunoblots. Right: immunofluorescence microscopy of splenic DR5 (TRAIL-R2) expression. Images representative of three mice per group. Scale bar, 25 μm. i, High resolution AiryScan images of splenocytes from WT (top) and CIap2−/− (bottom) mice D3 post-MI. Immunostaining for TRAIL (green) and DR5/TRAIL R2 (violet). DAPI (blue), nuclei. Scale bar, 5 μm. Imaging representative of three animals per treatment group. j, TUNEL+ splenocytes reduced D3 post-MI in CIap2−/− mice injected with anti-TRAIL neutralizing antibody. Image representative of three animals per treatment. Scale bar, 50 μm. k, LV ejection fraction and heart weight:tibia length of mice D28 post-MI following TRAIL neutralizing antibody injection. Error bars denote mean ± s.d.; P values were calculated by one-way ANOVA with Bonferroni’s correction (e,f,g), two-way ANOVA with Bonferroni’s correction (a,c,k).
Biochemical analyses of the spleen after MI indicated an important role for cIAP2 in promoting inflammatory responses. Whole spleen lysates collected over time after MI showed that cIAP2 protein was progressively upregulated and correlated with reduction of TRAF3, a known protein degradation target of cIAP2 (Extended Data Fig. 6a). CIap2-deficient but not CIap1-deficient spleens showed preservation of TRAF3 and XIAP, an immune regulatory IAP protein (Extended Data Fig. 6b). Protein expression of nodal inflammatory regulators for both PRR and TNFR signaling (IRAK4, MyD88, RIPK1 and TRAF6) were all decreased in CIap2−/− spleens at 7 days after MI (Extended Data Fig. 6c). cIAP2 co-immunoprecipitation with Traf3 was evident after MI injury (Extended Data Fig. 6d). cIAP2+ splenocytes showed increased proteolytic K48 ubiquitination of TRAF3 and activating K63 ubiquitination of RIPK1 (Extended Data Fig. 6e, upper panel). Although CIap2 deletion did not affect the non-canonical NF-κB pathway in 24-hour post-MI splenocytes (Extended Data Fig. 6e, lower panel), nuclear localized canonical NF-κB factors p65/RelA and NF-κB1 (p50) were reduced in CIap2−/− splenocytes as compared to WT mice 3 days after MI (Extended Data Fig. 6f). Furthermore, levels of Tnfa, Il1b and Il6 were reduced in CIap2−/− spleens (Extended Data Fig. 6g). Altogether, these results suggest that cIAP2 is required for acute post-MI inflammatory responses by specifically modulating effector cells in the spleen (Extended Data Fig. 6h).
The response to MI from splenocytes is critical to maximal inflammatory activity and could be the culprit for worst-case cardiac outcome. It has been established that the spleen provides a critical reservoir of effector monocytes and other inflammatory cells that are rapidly mobilized to the ischemic myocardium35,36. In CIap2−/− mice, we observed reduced numbers of neutrophils and Ly6Chi monocytes from spleens isolated 7 days after MI (Fig. 3e), which correlated with increased local splenocyte cell death (Fig. 3f). By contrast, splenic and cardiac levels of lymphocytes were unchanged throughout the acute post-MI phase (Extended Data Fig. 5e–g). These findings are consistent with the concept that cIAP2 permits splenic myeloid cell survival, which may support sustained heart inflammatory responses (Extended Data Fig. 5h). Aside from controlling NF-κB-associated inflammatory responses, cIAP proteins also suppress activation of TNF superfamily death complexes and, thus, can regulate apoptotic or inflammatory (necrotic) cell death37. CIap1/CIap2 deficiency in myeloid cells, including monocytes, results in cell death after exposure to TNF38. cIAP2 also plays an important role in governing TNF-dependent survival and myeloid differentiation potential of hematopoietic stem cells39. We found that, in contrast to 3 days after MI, Tnfa mRNA and protein levels were increased exclusively in spleen samples from CIap2−/− mice after 7 days (Extended Data Fig. 7a,b). We additionally observed that protein expression of death ligand TRAIL (or TNFSF10/Apo2L) was increased in spleens but not in hearts of CIap2−/− mice 3 days after MI (Fig. 3g). CIap2−/− splenocyte TRAIL and TNF protein levels were increased 7 days after MI as were active forms of Caspase-8 and Caspase-3 and the apoptotic marker Annexin A1 (Extended Data Fig. 7b). TRAIL protein and its permissive transcription factor Interferon Response Factor 1 (IRF-1)40 were increased in CIap2−/− bone marrow cells (Extended Data Fig. 7c). Additionally, CIap2−/− splenocytes upregulated TRAIL-R2/DR5, a TRAIL-binding death promoting receptor, as well as TNFR1, which can promote apoptosis in IAP-deficient cells (Fig. 3h). Immunohistochemistry showed that the numbers of TRAIL-expressing and DR5-expressing cells were both increased in CIap2−/− spleens, with TRAIL+ cells found interacting with DR5+ splenocytes after MI (Fig. 3i and Extended Data Fig. 7d). Injection of TRAIL-neutralizing antibodies prevented splenocyte death in CIap2−/− mice after MI (Fig. 3j). Finally, we confirmed that TRAIL expression is protective after MI, as antibody-mediated neutralization of TRAIL enhanced cardiac injury and inflammation in both WT and CIap2−/− mice (Fig. 3k and Extended Data Fig. 7e,f) and nullified protection associated with cIAP2 deficiency. These data collectively demonstrate that cIAP2 ablation increases splenocyte sensitivity to TNF-associated death signals, in part by upregulation of effector proteins such as TRAIL and DR5, contributing to the reduction of splenic inflammatory cell burden (Extended Data Fig. 7g).
cIAP inhibitory Smac mimetics are cardioprotective after MI
Our findings suggested that cIAP2 could be a pharmacologic target during the acute early phase after MI. We evaluated the monovalent Smac mimetic LCL161 as a model agent to assess the effect of cIAP inhibition after MI (Fig. 4a). In vivo, single 10 mg kg−1 doses of LCL161 effectively degraded splenocyte cIAP1/cIAP2 (Fig. 4b) and reduced levels of circulating blood neutrophils and inflammatory Ly6Chi monocytes (Extended Data Fig. 8a), effects achieved at five-fold lower doses than required for mouse tumor regression29. By contrast, LCL161 had no effect on cardiac cIAP protein levels at doses lower than tolerance limits from phase 1 clinical trial evaluation41. Pretreatment of ex vivo bone marrow-derived dendritic cells with LCL161 reduced broad K63 ubiquitination as well as expression of IRAK4, MyD88 and RIPK1 in response to stimulation by freeze−thaw heart lysate (Extended Data Fig. 8b). Mice subjected to MI were administered LCL161 at 1 day and 4 days after MI (Fig. 4c). Compared to mice receiving vehicle control, LCL161-treated mice exhibited significantly reduced mortality after MI (Fig. 4d). LCL-treated mice also exhibited reduced left ventricular scarring, heart weight and left ventricular remodeling accompanied by improvement in left ventricular ejection fraction (Fig. 4e–h). LCL161 treatment also reduced cardiac immune infiltration after MI (Fig. 4i and Extended Data Fig. 8c,d). cIAP2 serves as the principal inflammatory target after MI, evidenced by the differential outcomes achieved by LCL161 in CIap1−/− MI-operated mice compared to CIap2−/− mice (Extended Data Fig. 8e). Cardiac expression of remodeling and inflammatory genes was lower in LCL161-treated mice relative to vehicle controls 28 days after MI (Fig. 4j and Extended Data Fig. 8g).
Smac mimetic LCL161 is cardioprotective after MI. a, LCL161 stimulates degradation of cIAP1/2. b, In vivo cIAP1/2 degradation in spleen and heart tissues following dose titration of LCL161. Immunoblot representative of two similar experiments. c, Strategy for LCL161 administration following MI. d, Kaplan–Meier survival analysis of mice subjected to MI receiving vehicle or LCL161 gavage. Vehicle-treated controls, n = 18 mice (start of experiment); LCL161-treated controls, n = 15 mice (start of experiment). e, LV scar area of MI-operated mice receiving vehicle control (top) or LCL161 (10 mg kg−1, bottom); n = 7 per group. Scale bars, 1mm. f, Heart weight:tibia length D28 post-MI. g, LV end systolic diameter and measurements taken D28 post-MI. h, LV ejection fraction measured D28 post-MI. Control operated mice, n = 7 (3 LCL-treated, 4 vehicle treated); MI-operated mice, n = 10 vehicle treated, n = 11 LCL161-treated. i, Flow cytometry of heart infiltrating neutrophils, Ly6Chi monocytes and dendritic cells D28 post-MI. Control-operated mice, n = 3 (two vehicle-treated, one LCL161-treated); MI-operated mice, n = 6 per group. Data aggregate from three experiments (d–i). j, Quantitative PCR of remodeling gene products D28 post-MI; n = 4 per MI treatment group. Error bars denote mean ± s.d.; P values were calculated using Gehan-Breslow-Wilcoxon test (d), two-sided t-test (e,j), one-way ANOVA with Bonferroni’s correction (f,g,h,i). LCL, LCL161; LVESD, left ventricular end-systolic diameter; Veh, vehicle.
Single administration of LCL161 24 hours after MI was also cardioprotective when assessed at 7 days, as demonstrated by reduced early scar formation, improved left ventricular ejection fraction and reduction of heart weight as well as reduction of cardiac inflammatory cells (Fig. 5a–c). Consistent with the effect of cIAP2 deficiency, myeloid inflammatory cell numbers were reduced in the spleens of LCL161-treated mice 7 days after MI (Fig. 5d). Overall, cardiac transcription of inflammatory and remodeling genes was unchanged in LCL161-treated mice relative to vehicle controls, with the exception of mRNA for key inflammatory chemokine Ccl2 and its receptor Ccr2 (Fig. 5e). Further evidence of LCL161 effectiveness to reduce cIAP inflammatory activity was revealed by increased TUNEL staining selectively in splenocytes, concomitant with greater expression of TRAIL in LCL161-treated MI-operated mice (Fig. 5f,g). Critically, and in agreement with a dependence on persistent inflammatory state to influence cell fate, numbers of splenic and bone marrow leukocytes had rebounded by day 28 after MI (Extended Data Fig. 8h). Altogether, our results demonstrate a critical role for cIAP2 in maintaining a maximal inflammatory response in mice after MI. This response is mediated by cIAP2 control over cell death signaling to promote increased survival of myeloid cells, the importance of which being demonstrated by the protective inflammatory cell-reducing effects after MI of cIAP-inhibitory Smac mimetic compounds.
a, Single dose regimen for mice with LCL161. b, Masson’s trichrome staining of hearts showing reduced LV scar formation at D7 post-MI following single dose LCL administration of mice, with accompanying improvement in ejection fraction and reduction in heart weight normalized to tibia length. Scale bars, 1 mm; n = 3 per group. c, Reduction of immune infiltration at D7 post-MI following single dose LCL gavage. Controls, n = 3; MI-operated, n = 6 per group. Data are aggregate of two representative experiments. d, Flow cytometric measurement of spleen neutrophils and Ly6Chi monocytes D7 post-MI. n = 4 mice per group. e, Quantitative PCR measurement of representative inflammatory transcripts normalized to HPRT from spleens D7 post-MI. n = 3 mice per group. f, increased TUNEL+ splenocytes in LCL-treated mice D7 post-MI. Scale bars, = 50 μm. Vehicle treated, n = 6; LCL-treated, n = 5. g, Immunofluorescence microscopy showing TRAIL+ staining of splenocytes from mice 7 days post-MI. Scale bars, 25 μm; n = 5 mice per group. Error bars denote mean ± s.d.; P values were calculated using two-sided t-tests.
Discussion
Mortality rates from acute MI are in steady decline42,43; however, ischemic heart disease remains the primary cause of non-infectious death1,44. Survivors of ischemic injury are prone to chronic inflammatory disease and increased risk of subsequent cardiovascular events or heart failure2,45,46. Additionally, patient quality of life is markedly diminished with severity of patient-reported impact related to prognosis47.
Increasing attention and research focus have been directed to the inflammatory response after MI. It has been established that increased systemic inflammation is present after acute ischemic cardiac injury, with a strong myeloid inflammatory component48,49,50. Prognosis is also negatively influenced by sustained elevation of circulatory leukocytes45,51. Clinical investigation has highlighted the exciting potential of anti-inflammatory therapies to improve acute outcomes after MI6,7,8. However, results are mixed when the treatments are administered in the chronic phase after MI; although the studies established proof of concept, the agents were associated with increased risk of infection. Therefore, we searched for candidate inflammatory modulators that can be targeted earlier after MI to more effectively recalibrate the downstream chronic inflammatory response. Multiple key innate pattern recognition receptors and pro-inflammatory cytokine receptors are activated after MI9, and signal transduction pathways initiated by these molecules show redundancy and conservation, notably in the activation of transcription factors such as NF-κB. TLR, NLR as well as TNF and IL-1 receptors commonly recruit cIAP1 and cIAP2—RING-type E3 ligases that regulate cell survival and inflammation52,53. Impairment of cIAP protein pro-survival function has been the dominant rationale in developing inhibitory small-molecule strategies for the treatment of malignancies that may feature their overexpression26,54.
This study suggests that cIAP2 plays a critical role as a regulator of innate inflammatory cells, where its loss of expression after MI results in reduced cell accumulation in the heart while also reducing cellular burden in the spleen. The spleen is an important source of inflammatory cell mobilization after MI55,56, and this systemic reservoir can incite further chronic inflammatory consequence if left unregulated57. Although hematopoiesis plays an important role in the maintenance of chronic inflammatory states, we postulate that, in the setting of cIAP2 deficiency, the key therapeutic effect is upon cell death, particularly of myeloid cells. This is because of the well-described importance of cIAP proteins, particularly cIAP2, to promote myeloid cell survival in response to MI-relevant inflammatory ligands such as TNF or agonists of TLRs38,53,58. Our findings show that cIAP2 deficiency or cIAP ablation induced a marked reduction of myeloid cells, concomitant with lowered cardiac inflammation and overall attenuated inflammatory cellular signaling in the acute phase after MI. The viability of cIAP2 as an early target for such an acute, aggressive injury is evident by the observation that the overall immune system response returned to a balanced state in latent timepoints. With relevance to the death-inducing effect of cIAP2 deficiency, we found elevated expression of pro-apoptotic TNF family factor TRAIL59 after MI in CIap2−/− mice. Upregulation of the mouse ortholog of TRAIL-selective death receptor DR5 (TRAIL-R2) was also observed, which is known to be an essential permissive step for TRAIL bioactivity during many apoptotic cell responses60. In cardiovascular disease, TRAIL expression infers protective effects, as high serum levels of TRAIL are associated with favorable outcomes61,62, whereas circulatory (soluble) DR5 predicts increased mortality63. However, its role in the heart after injury remains poorly understood. In vivo studies have suggested disparate effects of TRAIL activity. Acute blockade of DR5 reduced myocardial ischemia−reperfusion injury64; conversely, agonists of DR5 reduced cardiac injury and remodeling in a chronic injury model65.
Expression of mouse TRAIL and its receptor is widely distributed in the body. Consistent with other TNF ligands, it possesses pleiotropic functions, including regulation of NF-κB-dependent inflammation. Notably, TRAIL appears to demonstrate an ability to selectively induce apoptosis, typically sparing healthy or non-malignant cells in various studies66. Thus, it is possible that cellular fates in response to TRAIL activation are dependent upon the tissue environment or cell type being targeted. Acutely after MI, TRAIL expression is upregulated in the spleen but not in cardiac left ventricular tissue of CIap2−/− mice, suggesting that a critical site for inflammatory suppression is the spleen, consequently reducing supply of inflammatory cells to the heart. Antibody neutralization of TRAIL notably increased levels of inflammation and impacted cardiac function in both WT and cIAP2-deficient mice after MI, although we could not evaluate the effect of TRAIL neutralization in the acute post-MI heart. A limitation of using such strategies is the potential for generalized immunomodulation associated with administration of bulk IgG in technical control animals, thus potentially reducing the biological effects of the injury67. Use of activators or antagonists of DR5 could, in this context, be very informative to the role of TRAIL during the overall post-MI response.
Consistent with the above observation is the potent degree of antiinflammatory activity mediated by the IAP antagonist LCL161 (a Smac mimetic) after MI. Smac is an evolutionarily encoded IAP inhibitor68 and is the template for many compounds now in clinical development for cancer treatments69. Smac mimetics show variable levels of inhibition of cIAP1, cIAP2 and XIAP; however, there are differences in their inhibitory potency and/or their degradative capacity against the enzymes70,71. Molecular studies suggest that efficient degradation of cIAP proteins is strongly dependent upon expression of cIAP1 (ref. 72), which tends to be expressed more broadly across cell and tissue types and at more stable levels. By contrast, cIAP2, although highly homologous in sequence and structure to cIAP1, is a stress-responsive member of the IAP gene family26,53. This property may explain, in part, why our treatment, when applied early after MI, was effective to produce a lasting effect on cardioprotection. Transient ablation of cIAP2 (along with cIAP1) can potently reduce acute inflammation; therefore, avoidance of continuous administration may subsequently reduce the potential toxicity of the Smac mimetic. IAPs are known to play protective roles in resident tissue cell types after injury73,74,75. We established through dose titration of LCL161 that IAP inhibition could be achieved in splenocytes without impacting the protein levels in cardiac tissue, with protein degradation observed at quantities lower than predicted to be safely effective clinically. As evidence of broader immunomodulatory potential, leukocyte studies have demonstrated beneficial activity for Smac mimetics in the treatment of inflammatory bowel disease76 and a mouse model of pneumonia77. Ultimately, irrespective of the promise of these agents, further study is needed to confirm sparing of non-target organs and cellular effectors with cIAP inhibitory strategies.
In summary, we highlight cIAP2 as a potent proinflammatory factor that plays a critical role in maintaining cardiac inflammatory cell supply after MI. Cardiac injury and function are improved when hematopoietic expression of cIAP2 is ablated, and this protective effect is reproduced using a clinically relevant cIAP antagonist, LCL161. We propose that cIAP2 inhibition is an important new immunomodulatory strategy for improvement of cardiac outcomes after MI.
Methods
Human samples
Patient samples (blood whole plasma, peripheral blood mononuclear cells (PBMCs) and whole unfractionated leukocyte lysates) were obtained with consent and used under approval from the Ottawa Health Science Network Research Ethics Board (OHSN-REB protocols no. 20140869, no. 20150428 and no. 20160516-01H). Patient characteristics are summarized in Supplementary Table 1. For whole plasma cell-free protein analysis, samples were isolated from N = 7 randomized patients presenting with acute heart failure at the time of admission and subsequently at days 3 and 30 after admission and compared with N = 10 healthy controls. Analytes were screened using a SomaScan aptamer-tagged proteomics array (SomaLogic) as described previously78. Plasma was frozen until analysis at −80 °C. Acute MI versus CAD PBMC analyses were conducted using isolates from patients admitted to the University of Ottawa Heart Institute percutaneous coronary intervention clinic, where approximately 30 ml of whole blood samples was obtained within 2 hours of admission (for planned CAD intervention, N = 12) or diagnosis of ST-segment elevated MI (STEMI, referred to as ‘MI’, N = 9). PBMC isolates were performed by gradient centrifugation of PBS-diluted whole blood over a 2:1 (v/v) Percoll gradient at 675g for 30 minutes at room temperature, followed by concentration of buffy coat cell content in RPMI 1640 + 5% (v/v) FBS (Thermo Fisher Scientific) to a final concentration of 5 × 106 cells per milliliter. Standardized quantities of cells (1 × 106 per milliliter per sample) were reserved for flow cytometric analysis. For collection of cell lysate from unfractionated leukocytes, patient samples were collected at the time of clinical assessment, followed by removal of red blood cells using hypotonic ammonium chloride/potassium bicarbonate buffer and subsequent lysis of nucleated cell pellets. Lysates were stored for use in immunoassays. Patient sera were collected from patients with MI versus patients with CAD by centrifugation of 2 ml of whole blood at 1,000g at room temperature, followed by extraction of aqueous (non-packed hematocrit) fraction. Sera were stored for analysis at −80 °C.
Mouse post-MI gene set enrichment analysis
Left ventricular expression of gene products over a 48-hour timecourse was catalogued in the National Center for Biotechnology Information (NCBI)-curated, open-source dataset GSE4648 (GDS2329) (for reference, see https://www.ncbi.nlm.nih.gov/sites/GDSbrowser?acc=GDS2329). The dataset was uploaded onto the Salk Institute open-access R Shiny bioinformatics platform BART (Bioinformatics Array Research Tool)79, and data were organized and catalogued using applications encoded in the software package (N = 2 per MI timepoint in infarcted left ventricular region or N = 2 for controls at representative timepoints). Data for differential gene expression and Gene Ontology annotation were collected from the 24-hour post-MI timepoint, and the summary of differential genes is located in Supplementary Tables 2 and 3. Statistical analyses were conducted within the software application.
Animals
C57Bl/6 (WT), Ciap2−/−, Ciap1−/−, Nod1−/−, Tlr4−/− and Tnfrsf1a−/− mice were bred and co-housed for multiple generations under identical specific pathogen-free conditions (including consistent chow, water and air supply) in the University of Ottawa Heart Institute vivarium prior to experimental procedures. Prior to breeding and use in this study, the principal experimental mouse strains Ciap2−/− and Ciap1−/− were similarly normalized to C57Bl/6 background by backcrossing as described in ref. 27 and ref. 33, respectively. Bl/6 CD45.1 mice (B6.SJL-Ptprca Pepcb/BoyJ) were purchased from The Jackson Laboratory. Mice were 12−16 weeks of age prior to surgical experiments. Mice were kept on a consistent 6:00−20:00 light/dark cycle, at constant 22° C temperature and 40% humidity and were provided chow and drinking water ad libitum. Both male and female mice were used for experimental MI procedures for germline knockout CIap2−/− studies; otherwise, males were used in reported experimental work. In all experiments, groups of animals were operated on in random order, and assignment to either experimental MI or control surgery was conducted in a blinded fashion. All animal experimentation was conducted under the guidance and with approval of the University of Ottawa Animal Care Committee (ACC), with experimental work adhering to ACC animal usage protocols HIb-2218, HIb-3536, HIe-2246 and HIe-3727. All surgical procedures were carried out with strict adherence to university-mandated protocols, including perioperative and postoperative analgesia and wellness assessment. Experiments were conceived and conducted to ensure compliance with directives from the Canadian Council for Animal Care and the ARRIVE criteria for responsible animal research.
MI
Male or female mice, aged 12−16 weeks, were randomly assigned to MI or sham (control) surgery. One hour prior to surgical procedure, mice were injected subcutaneously with a 1.2 mg kg−1 solution of buprenorphine slow-release formulation and a 1.0 mg kg−1 solution of meloxicam. Mice were anesthetized with 2−4% isoflurane (mixed with O2); hair on the left chest was removed with a depilatory cream; and mice were intubated with a 20-gauge soft catheter attached to a ventilator operating at 130−150 strokes per minute. Once in adequate anesthetic surgical plane, the chest was opened on the left rib cage between either the 4th or 5th intercostal space; the pericardium was disrupted; and the left anterior descending artery (LAD) was located and ligated with a 7-0 silk suture approximately 2 mm below the atrial appendage. Arterial occlusion was verified by blanching of the distal myocardial tissue, at which time the chest was closed with a 6-0 Surgipro polyvinyl suture. Sham-operated controls underwent the same procedure foregoing the terminal LAD ligation. The animals were allowed to recover from anesthetic in a 30 °C hyperbaric incubator and received 1.0 mg kg−1 meloxicam daily for two subsequent days. Food and water were not withdrawn at any point prior to or after surgery. Mice were subsequently monitored for the duration of the experimental protocol, determined at random to last 1, 3, 7 or 28 days unless endpoint was determined by wellness assessment.
Isoproterenol treatment
Twelve- to 16-week-old male C57Bl/6 mice were injected subcutaneously (subscapular) with 10 mg kg−1 isoproterenol-HCl in saline. Twenty-eight days after injection, cells and protein lysates were isolated and used in flow cytometry or immunoblotting assays.
Blood, spleen, bone marrow and heart immune cell isolation
PBMCs
Whole blood (50−100 μl) was collected by temporal vein lancet puncture and lysed with 20 volumes (1−1.5 ml) of cold red blood cell lysis buffer (150 mM ammonium chloride (NH4Cl), 20 mM potassium carbonate (K2HCO3) and 0.1 mM EDTA). Suspensions were centrifuged at 330g, and non-lysed cell pellets were resuspended in either PBS supplemented with 1% BSA for flow cytometry analyses or cell lysis buffer (PBS supplemented with 1% (v/v) Triton X-100, 0.25% (v/v) sodium deoxycholate, 5 mM EDTA, 10 mM sodium orthovanadate, 2 mM sodium fluoride and 1× concentrated cOmplete protease inhibitor cocktail).
Spleens
Whole spleens from sham or MI-treated mice were excised, washed in cold sterile PBS and disrupted by crushing through 40-μm nylon mesh strainers. Cells were then centrifuged at 330g and resuspended in red blood cell lysis buffer. Non-lysed cells were recentrifuged at 330g, and cell pellets were resuspended in PBS plus 1% BSA (for flow cytometry), RPMI 1640 + 5% FBS (for adoptive transfer) or cell lysis buffer (for immunoblot protein analyses).
Bone marrow
Long bones (either tibia or femurs) were excised; muscle and connective tissues were removed; and bones were washed in cold 70% ethanol and PBS, consecutively. Bones were then crushed using a mortar and pestle. Marrow was resuspended in cold PBS and passed through a 40-μm nylon strainer, and cell suspensions were centrifuged at 330g, followed by resuspension in PBS/1% BSA, RPMI 1640 + 5% FBS or cell lysis buffer as above.
Hearts
Hearts were excised and flushed with cold PBS, and left ventricular tissue was dissected. In preliminary experiments where both MI or isoproterenol treatments were evaluated, the apical (approximate lower third) region was removed, and cells were isolated; in focused MI experiments, the infarct-affected (border) region, which we defined as the approximate 2-mm boundary on either side of the infarcted region (identified by fibrosis or blanching of the myocardium), was removed and used for cell isolation and flow cytometry or for whole-tissue molecular analyses (RT−qPCR or immunoblotting; Supplementary Fig. 1). Left ventricular tissue extracts were weighed and digested in tissue digestion buffer (113 mM NaCl, 0.6 mM KH2PO4, 0.6 mM Na2HPO4, 20 mM HEPES, 20 mM NaHC03, 4.7 mM KCl, 2 mM MgCl2, 1 mM EDTA, 60 U ml−1 DNAse I (MilliporeSigma), 100 U ml−1 hyaluronidase (MilliporeSigma), 450 U ml−1 Collagenase I (Worthington Biochemical), 250 U ml−1 Collagenase II (Worthington Biochemical) and 125 U ml−1 Collagenase D (Worthington Biochemical)) by shaking at 37 °C for 1 hour. Liberated cells were gravity filtered through a 40-μm nylon strainer by washing with cold PBS. Cell suspensions were centrifuged at 330g and resuspended in either PBS/1% BSA or RPMI 1640/5% FBS. For quantitation purposes, aliquots of all cell suspensions (prior to red blood cell lysis or centrifugation) were collected; 1 in 10 dilutions were made in PBS; and cells were counted using a hemocytometer.
Flow cytometry and fluorescence-activated cell sorting
Cell isolates collected as described above were stained with specified antibody cocktails at a concentration of approximately 2.5 × 106 cells per milliliter in PBS supplemented with 1% BSA for 90 minutes on ice protected from light. Antibody concentrations were prepared as master cocktail mixes with specific antibodies employed at the concentrations indicated in Supplementary Table 4. For cytometric analysis, stained cells were washed with PBS, spun down at 330g and resuspended in PBS supplemented with 1% v/v paraformaldehyde. Stained cells were either immediately analyzed or stored overnight at 4 °C protected from light. Gating strategies for cytometry experiments are provided in the Supplementary Data file. For fluorescence-activated cell sorting (FACS) purification and cell recovery, stained cell isolates were washed, resuspended in RPMI 1640/5% FBS and immediately resolved. Cells were collected into chilled 5-ml round-bottom polypropylene tubes containing 1 ml of RPMI 1640/5% FBS. Where possible, cell purifications were verified by performing reanalysis on aliquots of recovered sample or by microscopic assessment on a hemocytometer. All cytometric processing was carried out on a FACSAriaIII four-laser cell sorter/cytometer (BD Biosciences). Experiments were acquired using BD FACSDiva version 8.2 software, and analysis was performed using either FACSDiva or FlowJo version 10.8 software (Tree Star).
Bone marrow transplantation
Bone marrow chimeric mice were established by lethally irradiatiating 6−8-week-old B6 CD45.1 mice with exposure to two cycles (spaced by 20-minute intervals) of 450-cGy gamma irradiation using a Gammacell 3000 Cesium137 source irradiator (Best Theratronics). Within 3 hours of irradiation, mice were injected intravenously with 2.5 × 106 sterile-isolated, filtered bone marrow cells from C57Bl6/, CIap2−/− or CIap1−/− donors. Mice were allowed to recover over a minimum 6-week period in a sterilized housing unit, after which a blood sample was collected to assess hematopoietic reconstitution by flow cytometric analysis of CD45.1 (recipient strain) and CD45.2 (donor strain) isotypes as well as verification of PBMC populations. Chimerism of 90% or greater was consistently achieved, with no apparent deficiency in specific cell populations. Animals were subsequently randomized into MI experiments, with sample groups aggregated from three separate similar experimental protocols. In all experiments, small groups of mice were randomly assigned to sham surgical procedure. For ‘reciprocal’ bone marrow transplantation experiments, CD45.2 (C57Bl/6 or CIap2−/−) mice were irradiated and served as recipient strains for CD45.1 bone marrow donation in order to assess the role of resident cardiac cIAP2 expression after injury. Experiments were presented as aggregate data from two similar experimental protocols.
Antibody injections for in vivo neutralization studies
TRAIL (Tnfsf10) neutralization using N2B2 monoclonal antibody
C57Bl/6 or CIap2−/− mice (as above) were injected with either 250 μg of rat anti-TRAIL IgG2a(κ) monoclonal antibody (clone N2B2; BioLegend, 109308) or equivalent quantity of isotype control rat IgG2a (clone RTK2758; BioLegend, 400502) via intraperitoneal injection 3 days before surgery, perioperatively or 3 days after surgery. Apoptosis in spleens of MI-operated mice was evaluated in a subgroup of animals at 3 days after MI, and the remaining animals were euthanized at day 28 for assay of cell infiltration or cardiac injury, as indicated in Fig. 3j,k and Extended Data Fig. 7e,f.
Smac mimetic treatments
The monovalent Smac mimetic LCL161 was purchased from Selleck Chemicals. For in vitro experiments, LCL161 was dissolved in dimethylsulphoxide (DMSO) and used at 5-μM concentrations applied to cultured bone marrow-derived dendritic cells (described below) for 48 hours in the presence or absence of inflammatory stimulation. Control cultures were treated with DMSO only. For in vivo experiments, mice were first weighed and then administered 10 mg kg−1 LCL161 dissolved in sodium acetate buffer (0.07 M NaOAc, 0.03 M HCl (pH 4.6)) orally by intragastric gavage at day 1 or days 1 and 4 after MI. Control mice were gavaged with acetate buffer only. Animals were monitored for up to 7 days or 28 days after MI, as indicated.
Echocardiography
Mice were sedated with 1.5% isoflurane (per liter O2), and hair was removed from the left chest. Parasternal long axial (high resolution, electrocardiogram-gated kilohertz visualization (EKV) and low resolution, B mode) and short axial (M mode) images were collected using a Vevo 770 or a Vevo 3100 ultrasound imaging station (FUJIFILM/VisualSonics). Systolic function (ejection fraction and stroke volume) was estimated by the observed change in two-dimensional area measurements of endocardial wall from end-diastole to end-systole using parasternal long axis (planar projection of whole ventricle from aortic valve to apex) high-resolution (300 images per beat) EKV image analysis; dimensional change and myocardial wall thickness were estimated by linear measurement of midventricular chamber from anterior to posterior wall in short axial (vertical projection) images. Imaging was performed by a blinded operator, and image analysis was carried out in random order by a blinded investigator. Measurement and calculation of imaging results was performed with Vevo LAB software (version 3.0-770 or version 3.1.1).
Histology
Hearts were excised, flushed with cold PBS and immersed in 0.5 M KCl for 5 minutes to enhance ventricular dilatation. Subsequently, hearts were fixed in 20 volumes (approximately 3 ml for mouse hearts) of 4% paraformaldehyde for 24−48 hours, reimmersed in PBS for another 48 hours and then equilibrated in autoclaved water and stored at 4 °C until being embedded in paraffin. For tissue sectioning, full thickness (concentric) 5-μm sections were cut using a microtome and collected onto slides. Tissue sections were deparaffinized using toluene and rehydrated by immersion in stepwise decreasing ethanol gradients and then subjected to hematoxylin and eosin staining, Masson’s trichrome or immunofluorescence microscopy. Midventricular sections corresponding to regions spanning the infarction were selected (based upon presence of silk suture fibers from LAD ligation; approximately similar tissue regions were selected from sham-operated mice) and used for analysis.
Tissue morphometry
Serial midventricular sections from MI-treated or sham-operated mouse hearts were stained with Masson’s trichrome (to expose collagenous scar tissue, stained blue), and collagen area was estimated as a fraction of whole left ventricle by blinded measurement using ImageJ (version 3) open-source software (National Institutes of Health). Mean area calculations from each subject were used to estimate scar area, where a minimum of five animals per MI treatment group were used for measurement.
Cell culture experiments
Reagents were purchased from Thermo Fisher Scientific unless otherwise indicated. Isoproterenol-HCl was purchased from MilliporeSigma. iE-DAP and poly I:C were purchased from InvivoGen. Flt3 ligand (Flt3L) was purchased from R&D Systems/Bio-Techne. Bone marrow-derived plasmacytoid dendritic cells: Whole bone marrow was isolated from long bones as described, and 2 × 106 cells per milliliter were seeded into 12-well culture dishes (500 ml per well) with RPMI/5% FBS supplemented with 50 ng ml−1 Flt3L and re-fed every third day for 7 days. The final 3 days, cells were additionally administered 5 µM poly I:C to further mature the cell populations80. Cells were subsequently stimulated with 10 μM ie-DAP and 20 μg necrotic heart tissue. Following 48 hours, cultured cell lysates were collected and immunoblotting was performed for proteins as presented in as presented in Extended Data Fig. 8b.
Immunofluorescence microscopy
Formalin fixed, paraffin-embedded heart midventricular sections were deparaffinized, and antigen retrieval was performed by either a 15-minute incubation in 1× proteinase K at 37 °C (for TUNEL staining or intracellular antigen staining) or a 10-minute steaming at 65 °C with 10 mM sodium citrate buffer (for surface antigen staining or isolectin B4 staining). TUNEL assay was conducted using a Click-iT EdU kit (AlexaFluor 594) (Thermo Fisher Scientific) according to the manufacturer’s specifications with nuclear counterstaining using DAPI. For immunostaining, sections were blocked for 30 minutes with 2% normal goat or donkey serum (see below) in PBS. Primary antibodies (Supplementary Table 5) were incubated with sections using indicated dilutions overnight at 4 °C, followed by washing with PBS plus 0.05% (v/v) Tween 20 detergent. Secondary antibodies (goat anti-mouse or donkey anti-rabbit) conjugated to various AlexaFluor fluorochromes were used at reported dilutions in the appropriate blocking buffer (determined by host species) and incubated on sections at room temperature for 90 minutes, followed by washing with PBS 0.05%/Tween 20 and PBS, sequentially. Staining of myocardial vasculature was performed using Griffonia simplicifolia isolectin B4 conjugated with AlexaFluor 488 (Thermo Fisher Scientific) at a concentration of 1μg ml−1 (diluted in 10 mM MgCl2/MnCl2/CaCl2) overnight at 4 °C and counterstained with DAPI. Slides were mounted with ProLong Antifade Gold (Thermo Fisher Scientific). The photomicrographs were captured in a blinded fashion using an Olympus IX81 laser confocal microscope, and images were prepared with Fluoview version 4.3 software (Olympus Corporation). Quantitation of analytes or antigens was conducted by counting events per high-powered field, minimum four fields per ventricle or splenic section, using at least triplicate animals per treatment. For high-resolution micrographs, slides were imaged on a Zeiss ELYRA LSM 880 microscope using a ×20 planar apochromat objective with AiryScan function. Images were acquired and processed using Zeiss ZEN 2.3 software (Carl Zeiss) and Imaris Viewer (version 9.8.0, Oxford Instruments) (for z-stack deconvolution).
Protein extracts from fresh-frozen tissue
Border regions of post-MI left ventricular tissue and spleens were excised at predetermined timepoints after the experimental procedure and washed in cold PBS. After being patted dry on a sterile filter paper and then weighed, tissues were placed in 2.0-ml screwcap cryovials and immersed in liquid nitrogen for storage at −80 °C. For protein isolation, tissues were thawed on ice in cold lysis buffer, finely minced with scissors and then disrupted using a glass homogenizer. Suspensions were incubated on ice for 1 hour with occasional vortexing and then sonicated twice for 10-second intervals. Supernatants were clarified by centrifugation at 12,000g at 4 °C for 10 minutes and transferred to new sample tubes for use in immunoblotting. Prior to electrophoresis, protein quantities were determined by Bradford assay.
Nuclear extracts
Halved spleens (approximately 35−40 mg), either freshly isolated or frozen, were incubated in 1.5-ml Eppendorf tubes on ice in cold PBS followed by PBS diluted 1:5 in distilled deionized water. Tissue was minced into small pieces with scissors and spun at 500g, and PBS solution was replaced with hypotonic lysis buffer (10 mM HEPES (pH 7.5), 10 mM KCl, 0.1 mM EDTA, 1 mM DTT, 1× concentrate cOmplete protease inhibitor cocktail and 50 mM sodium orthovanadate). Tissue was further minced, vortexed gently and incubated for 10 minutes on ice, followed by addition of 0.5% Triton X-100. Cell suspensions were spun at 1,500g; supernatants (‘cytoplasmic’ fraction) were transferred to collection tubes; and pellets were resuspended in nuclear extraction buffer (20 mM HEPES (pH 7.5), 20 mM KCl, 80 mM NaCl, 25% (v/v) glycerol, 0.5 mM EDTA, 1 mM DTT, 1× concentrate cOmplete protease inhibitor cocktail and 50 mM sodium orthovanadate) for 30 minutes on ice. Suspensions were intermittently vortexed and finally centrifuged at 12,000g (4 °C) for 15 minutes. Supernatants were collected, and both cytoplasmic and nuclear extracts were stored at −80 °C for use in immunoblotting. Prior to electrophoresis, sample protein concentration was measured by Bradford assay.
Immunoprecipitation and immunoblotting
Immunoprecipitation
300 μg of cell extracts was incubated in lysis buffer and 2 μg ml−1 mouse monoclonal anti-TRAF3 (Santa Cruz Biotechnology), rabbit ployclonal anti-TRAF6 (Cell Signaling Technology) or mouse monoclonal anti-RIPK1 (BD Biosciences) antibodies, respectively. Samples were rotated continuously at 4 °C overnight and subsequently incubated with 0.10 volumes of Protein G-conjugated Dynabeads (Thermo Fisher Scientific) at 4 °C for an additional 90 minutes. Antigen-antibody complexes were immobilized using a DynaSpin column (Thermo Fisher Scientific), and the unbound fraction was saved for immunoblot analysis. Beads were subsequently washed in lysis buffer and PBS, sequentially, and antigen-antibody complexes were eluted using 4× Laemmli sample loading buffer and heating at 95 °C for 5 minutes. Samples were loaded onto 8% SDS-PAGE gels, and isolated proteins were subsequently probed by immunoblotting for proteins as indicated. Ubiquitin pulldowns were performed using a Pierce Ubiquitin Enrichment Kit (Thermo Fisher Scientific) according to the manufacturer’s protocol, and eluted proteins were stored at −80 °C prior to immunoblotting.
Immunoblotting
Protein lysates collected as described were loaded as 10-μg aliquots onto 6% or 12% SDS-PAGE gels. Separated proteins were transferred onto polyvinyl difluoride (PVDF) membranes and immunoblotted with primary antibodies as indicated (listed in Supplementary Table 6). HRP-conjugated secondary antibodies (1:25,000 dilution; Bio-Rad) were incubated with blots, and labeled antibody-antigen complexes were developed using West Pico PLUS ECL reagent (Thermo Fisher Scientific) according to the manufacturer’s protocol. Blots were exposed and images collected on a Bio-Rad ChemiDoc XRS+ imaging system, and analysis was conducted using Image Lab version 6 software.
RNA isolation and real-time qPCR
Frozen peri-infarct region left ventricular tissue or spleen (approximately 40−60 mg) was digested in 800 µl of TRIzol solution (Thermo Fisher Scientific), followed by standard chloroform separation and 75% ethanol RNA precipitation. RNA concentrations were determined with a NanoDrop ND-1000 spectrophotometer (Thermo Fisher Scientific), and 1 μg of RNA was reverse transcribed to cDNA using All-In-One 5× RT PCR MasterMix (Applied Biological Materials) according to the manufacturer’s protocol. Sample cDNA was then diluted 1:10 and used for qPCR gene expression analysis using BrightGreen 2× qPCR MasterMix (Applied Biological Materials), with 10 mM target-selective primers as listed in Supplementary Table 7. PCR reactions were performed using the following cycling conditions: 95 °C for 15 seconds, 60 °C for 15 seconds and 72 °C for 15 seconds over a 40-cycle reaction sequence on a Roche LightCycler 96 instrument. Quantitation of transcript was performed using the ΔΔCt method, using HPRT or β-actin (indicated in the figure legends) as reference (control) PCR products. For most experiments, ΔΔCt values (the effect of MI on the target gene of interest) were compared between WT and CIap2−/− mice, and a relative expression ratio was calculated with WT MI expression change being assigned a reference value of 1.
ELISA
For clinical serum or lysate samples, proteins were isolated as described above and were used for measurement by human cIAP1 or cIAP2 ELISA (MilliporeSigma, RAB1369 and RAB0027). Assays were conducted as recommended according to the manufacturer’s protocols. Intracellular-isolated cIAP measurements (from PBMC lysates) were normalized by standard Bradford protein assay. For mouse analyses, spleen cells were cultured at densities of 1 × 106 cells per milliliter in RPMI/5% FBS for 48 hours, and conditioned media were collected and stored at −80 °C for use in ELISA. Conditioned media from splenocyte cultures were collected 48 hours after cell plating of 1 × 106 cells per well (12-mm wells) and subsequently used for detection of IL-10 or IL-23 using DuoSet ELISA kits (IL-10: DY417; IL-23: DY1887 (R&D Systems/Bio-Techne)) according to the manufacturer’s protocols.
Statistical analysis
Sample size calculations were performed for mouse MI experiments based upon estimation of effect sizes from preliminary experiments. In general, observation of relative performance (that is, magnitude of biological effect size) was compared among WT, CIap2−/− or CIap1−/− mice. Results were applied to subsequent experiments, with sample sizes determined according to methods summarized in ref. 81 using calculation formulas suitable for continuous variable measurements. A single exception was the use of linear regression statistical testing for survival analysis in Fig. 5d, where subject animal differences were evaluated ad hoc during experiments for cardiac injury measurements. Statistical calculations for experimental outputs were made from individual raw data using GraphPad Prism version 8 or version 9 software (GraphPad Software); sample sizes and selection of statistical tests are indicated in the figure legends.
Reporting summary
Further information on research design is available in the Nature Portfolio Reporting Summary linked to this article.
Data availability
All data supporting the findings in this study are included in the main article and associated files. Source data are provided in the Supplementary Information attachment accompanying the paper. Source data from gene set enrichment analysis and gene expression analyses were tabulated and are available in Supplementary Tables 2 and 3.
References
Martin, S. S. et al. 2024 Heart Disease and Stroke Statistics: a report of US and global data from the American Heart Association. Circulation 149, e347–e913 (2024).
Wellings, J., Kostis, J. B., Sargsyan, D., Cabrera, J. & Kostis, W. J. Risk factors and trends in incidence of heart failure following acute myocardial infarction. Am. J. Cardiol. 122, 1–5 (2018).
Frangogiannis, N. G. Regulation of the inflammatory response in cardiac repair. Circ. Res. 110, 159–173 (2012).
Andreadou, I. et al. Immune cells as targets for cardioprotection: new players and novel therapeutic opportunities. Cardiovasc. Res. 115, 1117–1130 (2019).
Swirski, F. K. & Nahrendorf, M. Cardioimmunology: the immune system in cardiac homeostasis and disease. Nat. Rev. Immunol. 18, 733–744 (2018).
Ridker, P. M. et al. Antiinflammatory therapy with canakinumab for atherosclerotic disease. N. Engl. J. Med. 377, 1119–1131 (2017).
Broch, K. et al. Randomized trial of interleukin-6 receptor inhibition in patients with acute ST-segment elevation myocardial infarction. J. Am. Coll. Cardiol. 77, 1845–1855 (2021).
Tardif, J.-C. et al. Efficacy and safety of low-dose colchicine after myocardial infarction. N. Engl. J. Med. 381, 2497–2505 (2019).
Epelman, S., Liu, P. P. & Mann, D. L. Role of innate and adaptive immune mechanisms in cardiac injury and repair. Nat. Rev. Immunol. 15, 117–129 (2015).
Newton, K. & Dixit, V. M. Signaling in innate immunity and inflammation. Cold Spring Harb. Perspect. Biol. 4, a006049 (2012).
Jaén, R. I. et al. Innate immune receptors, key actors in cardiovascular diseases. JACC Basic Transl. Sci. 5, 735–749 (2020).
Sun, M. et al. Excessive tumor necrosis factor activation after infarction contributes to susceptibility of myocardial rupture and left ventricular dysfunction. Circulation 110, 3221–3228 (2004).
Maekawa, Y. et al. Survival and cardiac remodeling after myocardial infarction are critically dependent on the host innate immune interleukin-1 receptor-associated kinase-4 signaling. Circulation 120, 1401–1414 (2009).
Fallach, R. et al. Cardiomyocyte Toll-like receptor 4 is involved in heart dysfunction following septic shock or myocardial ischemia. J. Mol. Cell. Cardiol. 48, 1236–1244 (2010).
Harpster, M. H. et al. Earliest changes in the left ventricular transcriptome post-myocardial infarction. Mamm. Genome 17, 701–715 (2006).
Lalaoui, N. & Vaux, D. L. Recent advances in understanding inhibitor of apoptosis proteins. F1000Res. 7, F1000 Faculty Rev-1889 (2018).
Silke, J. & Meier, P. Inhibitor of apoptosis (IAP) proteins—modulators of cell death and inflammation. Cold Spring Harb. Perspect. Biol. 5, a008730 (2013).
Jensen, S., Seidelin, J. B., LaCasse, E. C. & Nielsen, O. H. SMAC mimetics and RIPK inhibitors as therapeutics for chronic inflammatory diseases. Sci. Signal. 13, eaax8295 (2020).
Rothe, M., Pan, M.-G., Henzel, W. J., Ayres, T. M. & Goeddel, D. V. The TNFR2−TRAF signaling complex contains two novel proteins related to baculoviral inhibitor of apoptosis proteins. Cell 83, 1243–1252 (1995).
Peltzer, N., Darding, M. & Walczak, H. Holding RIPK1 on the ubiquitin leash in TNFR1 signaling. Trends Cell Biol. 26, 445–461 (2016).
Varfolomeev, E. et al. Cellular inhibitors of apoptosis are global regulators of NF-κB and MAPK activation by members of the TNF family of receptors. Sci. Signal. 5, ra22 (2012).
Zarnegar, B. J. et al. Noncanonical NF-κB activation requires coordinated assembly of a regulatory complex of the adaptors cIAP1, cIAP2, TRAF2 and TRAF3 and the kinase NIK. Nat. Immunol. 9, 1371–1378 (2008).
Vallabhapurapu, S. et al. Nonredundant and complementary functions of TRAF2 and TRAF3 in a ubiquitination cascade that activates NIK-dependent alternative NF-κB signaling. Nat. Immunol. 9, 1364–1370 (2008).
Tseng, P.-H. et al. Different modes of ubiquitination of the adaptor TRAF3 selectively activate the expression of type I interferons and proinflammatory cytokines. Nat. Immunol. 11, 70–75 (2010).
Zhong, B. et al. Ubiquitin-specific protease 25 regulates TLR4-dependent innate immune responses through deubiquitination of the adaptor protein TRAF3. Science Signaling 6, ra35 (2013).
Hunter, A. M., LaCasse, E. C. & Korneluk, R. G. The inhibitors of apoptosis (IAPs) as cancer targets. Apoptosis 12, 1543–1568 (2007).
Conte, D. et al. Inhibitor of apoptosis protein cIAP2 is essential for lipopolysaccharide-induced macrophage survival. Mol. Cell. Biol. 26, 699–708 (2006).
Du, C., Fang, M., Li, Y., Li, L. & Wang, X. Smac, a mitochondrial protein that promotes cytochrome c–dependent caspase activation by eliminating IAP inhibition. Cell 102, 33–42 (2000).
Beug, S. T. et al. Smac mimetics and innate immune stimuli synergize to promote tumor death. Nat. Biotechnol. 32, 182–190 (2014).
Chesi, M. et al. IAP antagonists induce anti-tumor immunity in multiple myeloma. Nat. Med. 22, 1411–1420 (2016).
Yang, C. et al. LCL161 increases paclitaxel-induced apoptosis by degrading cIAP1 and cIAP2 in NSCLC. J. Exp. Clin. Cancer Res. 35, 158 (2016).
Pemmaraju, N. et al. Final results of a phase 2 clinical trial of LCL161, an oral SMAC mimetic for patients with myelofibrosis. Blood Adv. 5, 3163–3173 (2021).
Conze, D. B. et al. Posttranscriptional downregulation of c-IAP2 by the ubiquitin protein ligase c-IAP1 in vivo. Mol. Cell. Biol. 25, 3348–3356 (2005).
Yan, X. et al. Deleterious effect of the IL-23/IL-17A axis and γδT cells on left ventricular remodeling after myocardial infarction. J. Am. Heart Assoc. 1, e004408 (2012).
Ismahil, M. A. et al. Remodeling of the mononuclear phagocyte network underlies chronic inflammation and disease progression in heart failure. Circ. Res. 114, 266–282 (2014).
Leuschner, F. et al. Rapid monocyte kinetics in acute myocardial infarction are sustained by extramedullary monocytopoiesis. J. Exp. Med. 209, 123–137 (2012).
de Almagro, M. C., Goncharov, T., Newton, K. & Vucic, D. Cellular IAP proteins and LUBAC differentially regulate necrosome-associated RIP1 ubiquitination. Cell Death Dis. 6, e1800 (2015).
McComb, S. et al. cIAP1 and cIAP2 limit macrophage necroptosis by inhibiting Rip1 and Rip3 activation. Cell Death Differ. 19, 1791–1801 (2012).
Yamashita, M. & Passegué, E. TNF-α coordinates hematopoietic stem cell survival and myeloid regeneration. Cell Stem Cell 25, 357–372 (2019).
Clarke, N., Jimenez-Lara, A. M., Voltz, E. & Gronemeyer, H. Tumor suppressor IRF-1 mediates retinoid and interferon anticancer signaling to death ligand TRAIL. EMBO J. 23, 3051–3060 (2004).
Infante, J. R. et al. Phase I dose-escalation study of LCL161, an oral inhibitor of apoptosis proteins inhibitor, in patients with advanced solid tumors. J. Clin. Oncol. 32, 3103–3110 (2014).
Nauta, S. T., Deckers, J. W., van Domburg, R. T. & Akkerhuis, K. M. Sex-related trends in mortality in hospitalized men and women after myocardial infarction between 1985 and 2008. Circulation 126, 2184–2189 (2012).
Ketchum, E. S. et al. The Seattle Post Myocardial Infarction Model (SPIM): prediction of mortality after acute myocardial infarction with left ventricular dysfunction. Eur. Heart J. Acute Cardiovasc. Care 3, 46–55 (2014).
Peters, S. A. E. et al. Trends in recurrent coronary heart disease after myocardial infarction among US women and men between 2008 and 2017. Circulation 143, 650–660 (2021).
Ye, Q., Zhang, J. & Ma, L. Predictors of all-cause 1-year mortality in myocardial infarction patients. Medicine 99, e21288 (2020).
Sulo, G. et al. Trends in the risk of early and late-onset heart failure as an adverse outcome of acute myocardial infarction: a Cardiovascular Disease in Norway project. Eur. J. Prev. Cardiol. 24, 971–980 (2017).
Pocock, S. et al. Health-related quality of life 1–3 years post-myocardial infarction: its impact on prognosis. Open Heart 8, e001499 (2021).
Massa, M. et al. Increased circulating hematopoietic and endothelial progenitor cells in the early phase of acute myocardial infarction. Blood 105, 199–206 (2005).
van der Laan, A. M. et al. A proinflammatory monocyte response is associated with myocardial injury and impaired functional outcome in patients with ST-segment elevation myocardial infarction: monocytes and myocardial infarction. Am. Heart J. 163, 57–65 (2012).
Zeng, S. et al. Trajectories of circulating monocyte subsets after ST-elevation myocardial infarction during hospitalization: latent class growth modeling for high-risk patient identification. J. Cardiovasc. Trans. Res. 11, 22–32 (2018).
Chia, S. et al. Association of leukocyte and neutrophil counts with infarct size, left ventricular function and outcomes after percutaneous coronary intervention for ST-elevation myocardial infarction. Am. J. Cardiol. 103, 333–337 (2009).
Bertrand, M. J. M. et al. Cellular inhibitors of apoptosis cIAP1 and cIAP2 are required for innate immunity signaling by the pattern recognition receptors NOD1 and NOD2. Immunity 30, 789–801 (2009).
Mahoney, D. J. et al. Both cIAP1 and cIAP2 regulate TNFα-mediated NF-κB activation. Proc. Natl Acad. Sci. USA 105, 11778–11783 (2008).
Fulda, S. Promises and challenges of Smac mimetics as cancer therapeutics. Clin. Cancer Res. 21, 5030–5036 (2015).
Dutta, P. et al. Myocardial infarction activates CCR2+ hematopoietic stem and progenitor cells. Cell Stem Cell 16, 477–487 (2015).
Swirski, F. K. et al. Identification of splenic reservoir monocytes and their deployment to inflammatory sites. Science 325, 612–616 (2009).
Dutta, P. et al. Myocardial infarction accelerates atherosclerosis. Nature 487, 325–329 (2012).
Wong, W. W.-L. et al. cIAPs and XIAP regulate myelopoiesis through cytokine production in an RIPK1- and RIPK3-dependent manner. Blood 123, 2562–2572 (2014).
Wiley, S. R. et al. Identification and characterization of a new member of the TNF family that induces apoptosis. Immunity 3, 673–682 (1995).
Wu, G. S., Burns, T. F., Zhan, Y., Alnemri, E. S. & El-Deiry, W. S. Molecular cloning and functional analysis of the mouse homologue of the KILLER/DR5 tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) death receptor. Cancer Res. 59, 2770–2775 (1999).
Niessner, A. et al. Prognostic value of apoptosis markers in advanced heart failure patients. Eur. Heart J. 30, 789–796 (2009).
Secchiero, P. et al. Potential prognostic significance of decreased serum levels of TRAIL after acute myocardial infarction. PLoS ONE 4, e4442 (2009).
Skau, E. et al. GDF-15 and TRAIL-R2 are powerful predictors of long-term mortality in patients with acute myocardial infarction. Eur. J. Prev. Cardiol. 24, 1576–1583 (2017).
Wang, X. et al. Blocking the TRAIL-DR5 pathway reduces cardiac ischemia–reperfusion injury by decreasing neutrophil infiltration and neutrophil extracellular traps formation. Cardiovasc. Drugs Ther. 39, 983−999 (2024).
Tanner, M. A., Dougherty, K. & Grisanti, L. A. Death receptor 5 agonists mitigate cardiac pathology in a chronic isoproterenol-induced cardiac remodeling and dysfunction. J. Pharmacol. Exp. Ther. 392, 103600 (2025).
Benedict, C. A. & Ware, C. F. TRAIL: not just for tumors anymore? J. Exp. Med. 209, 1903–1906 (2012).
Hematianlarki, M. & Nimmerjahn, F. Immunomodulatory and anti-inflammatory properties of immunoglobulin G antibodies. Immunol. Rev. 328, 372–386 (2024).
Chai, J. et al. Structural and biochemical basis of apoptotic activation by Smac/DIABLO. Nature 406, 855–862 (2000).
Bai, L., Smith, D. C. & Wang, S. Small-molecule SMAC mimetics as new cancer therapeutics. Pharmacol. Ther. 144, 82–95 (2014).
Yang, Q.-H. & Du, C. Smac/DIABLO selectively reduces the levels of c-IAP1 and c-IAP2 but not that of XIAP and Livin in HeLa cells. J. Biol. Chem. 279, 16963–16970 (2004).
Creagh, E. M., Murphy, B. M., Duriez, P. J., Duckett, C. S. & Martin, S. J. Smac/Diablo antagonizes ubiquitin ligase activity of inhibitor of apoptosis proteins. J. Biol. Chem. 279, 26906–26914 (2004).
Darding, M. et al. Molecular determinants of Smac mimetic induced degradation of cIAP1 and cIAP2. Cell Death Differ. 18, 1376–1386 (2011).
Rodrigue-Gervais, I. G. et al. Cellular inhibitor of apoptosis protein cIAP2 protects against pulmonary tissue necrosis during influenza virus infection to promote host survival. Cell Host Microbe 15, 23–35 (2014).
Annibaldi, A. et al. Ubiquitin-mediated regulation of RIPK1 kinase activity independent of IKK and MK2. Mol. Cell 69, 566–580 (2018).
Biswas, D. D. et al. Cellular inhibitor of apoptosis 2 (cIAP2) restricts neuroinflammation during experimental autoimmune encephalomyelitis. J. Neuroinflammation 19, 158 (2022).
Seidelin, J. B. et al. IAPs and RIPK1 mediate LPS-induced cytokine production in healthy subjects and Crohn’s disease. Clin. Exp. Immunol. 215, 291–301 (2024).
Li, S., Yang, P., Xu, L. & Li, M. Blocking of Birc3/TLR4/Myd88 signaling protects carbapenem-resistant klebsiella pneumoniae in a mouse model of infection. Transplant. Immunol. 69, 101464 (2021).
Zhang, L. et al. Insulin-like growth factor-binding protein-7 (IGFBP7) links senescence to heart failure. Nat. Cardiovasc. Res. 1, 1195–1214 (2022).
Amaral, M. L., Erikson, G. A. & Shokhirev, M. N. BART: bioinformatics array research tool. BMC Bioinformatics 19, 296 (2018).
Chen, Y.-L. et al. A type I IFN–Flt3 ligand axis augments plasmacytoid dendritic cell development from common lymphoid progenitors. J. Exp. Med. 210, 2515–2522 (2013).
Dell, R. B., Holleran, S. & Ramakrishnan, R. Sample size determination. ILAR J. 43, 207–213 (2002).
Acknowledgements
This work was funded by the Canada First Research Excellence Fund for the Brain-Heart Interconnectome (CFREF-2022-00007); the Government of Canada through Genome Canada and Ontario Genomics (OGI-2050); and the Brain Canada Foundation and the Heart and Stroke Foundation of Canada for Heart-Brain Connection IMPACT Award to P.P.L. R.G.K. is a recipient of funding support from the Canadian Institutes of Health Research. D.S. was a recipient of a Heart and Stroke Foundation of Canada research fellowship.
Author information
Authors and Affiliations
Contributions
D.S. developed the study concept, planned and performed experiments, analyzed data and wrote and edited the paper. L.Z. and M.A.-K. performed experiments, analyzed data and edited the paper. S.R., M.G., F.D., B.M., Q.D., C.B. and X.Z. performed experiments. R.S. planned experiments, performed surgeries, analyzed data and edited the paper. R.J. and S.P. recruited and collected clinical patient samples and analyzed data. M.O. planned experiments and edited the paper. S.T.B. and E.C.L. planned experiments, analyzed data and edited the paper. K.J.R. planned experiments, analyzed data and edited the paper. T.W.M. provided experimental resources and edited the paper. B.H. coordinated clinical recruitment, analyzed data and edited the paper. R.G.K. provided experimental resources, analyzed data and edited the paper. P.P.L. is the principal investigator, developed the study concept, coordinated clinical recruitment, analyzed data and wrote and edited the paper.
Corresponding author
Ethics declarations
Competing interests
E.C.L. is a co-founder and the Chief Scientific Officer of Protaxis Therapeutics, Inc. All other authors report no competing interests.
Peer review
Peer review information
Nature Cardiovascular Research thanks the anonymous reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Upregulation of cIAP2 post-MI.
a) and b) Gene expression changes within infarcted left ventricle (‘infarct’) are marked at 24- and 48-hours post-MI, as assessed by hierarchical clustering and principal component analyses of the open-source GEO dataset GSE4648. Analyses carried out using Geo2R and the open-source BART bioinformatics tool. c) Differential gene expression patterns identified in 24-hour post-MI ventricle compared with sham control. cIAP2 (Birc3) is significantly upregulated. d) LV infarct region gene expression timecourse of cIAP2 (maroon) and related transcripts cIAP1 (blue) and Xiap (green), contrasted with measured cardiac marker tnnt2 (Cardiac Troponin T) and inflammatory marker cxcl1. e) Compiled expression timecourse of representative myeloid-related transcripts (maroon) and lymphoid transcripts (blue) from LV infarct region post-MI. ptprc (black) = hematopoietic marker gene transcript. f) Gene Ontogeny (GO) pathway enrichment following MI identifies multiple networks requiring cIAP2. g) NOD1−/− mice have reduced systolic dysfunction and cardiac inflammation following MI relative to WT control mice. Control-operated mice: N = 3; MI-operated mice: N = 5. Ejection fraction: N = 4 MI-operated per group. h) cIAP2 is expressed at lower levels, along with inflammatory genes RIPK1, IRAK4 and TRAF6, in multiple danger signaling receptor-deficient mouse models following MI. Image is representative of two similar experiments. Error bars denote Mean +/- SD; p values were calculated using one-way ANOVA with Bonferroni’s correction (g, upper panel) or two-sided t-test (g, lower panel).
Extended Data Fig. 2 cIAP2 deficiency reduces post-MI injury.
a) Left ventricular end-diastolic dimension (LVEDD) and left ventricular end-systolic dimension (LVESD) measurements presented as % change from baseline (pre-MI) echocardiography in 28-day MI-operated mice. Right panel shows absolute LVESD measurements including Control operated mice, noting differences in systolic dimension of control animals. b) Immunoblot of apoptotic proteins Annexin A1, cleaved Poly-adenoribosyl polymerase (PARP) and cleaved Caspase 8 from Day 3 post-MI left ventricle tissue lysates. β-actin was used as an expression control c) Quantitative PCR measuring expression of representative injury-associated genes Brain-Natriuretic Peptide (Nppb), Myosin Heavy chain Isoform 7 (Myh7), Tumour Necrosis Factor-alpha (Tnfa) and Transforming Growth Factor-beta (Tgfb1) 28 days post-MI. N = 4 per treatment, target genes were normalized to expression of β-actin. d) Immunoblot of cardiac HMGB1 and TGFβ1 from Day 28 post-MI left ventricle tissue lysates. Vinculin was used as an expression control. Imaging representative of three similar experiments. e) Flow cytometry gating strategy for cell quantitation in Figs. 2b and 3b). Error bars denote Mean +/-SD; p values were calculated using two-way ANOVA with Bonferroni’s correction (a right panel), one-way ANOVA using Bonferroni’s correction (a left and centre panels), two-sided t-tests (c).
Extended Data Fig. 3 Immune-derived cIAP2 drives inflammation post-MI.
a) Bone marrow chimeric recipient heart and lung weights 28 days post-MI. Controls: N = 5 (WT)/3 (ciap2−/−); MI-operated: N = 17 per group. Results represent aggregate data from three experiments. b) Capillary density (green) of MI infarct border region. Scale bar = 50 μm. N = 4 per group, mean of 3 representative fields per animal. c) Flow cytometric quantitation of heart-infiltrating neutrophils and Ly6Chi monocytes from bone marrow transplanted mice Day 28 post-MI. Control: N = 3; WT chimeric MI: N = 4; ciap2−/− chimeric MI: N = 4. d) Inflammatory cytokine production from splenocytes isolated from bone marrow chimeric mice 28 days post-MI. N = 6 splenocyte cultures/treatment group. e) Left panel: flow cytometric quantitation of lymphocyte populations (CD4 + T cells, CD8 + T cells, B cells) from left ventricle tissue Day 28 post-MI. Right panels: Immunofluorescence microscopy assessing Th17 cell presence (white arrowheads) in left ventricular MI border zone. Green – IL-17A; red – CD3ζ chain; blue – DAPI. 400x magnification; scale bar denotes 50 μm. Cell quantitation was performed in WT: N = 3, ciap2−/−: N = 4, 4 fields counted per animal. Error bars denote Mean +/- SD; p values were calculated using two-way ANOVA with Bonferroni’s correction (a), one-way ANOVA with Bonferroni’s correction (b, c) or two sided t-test (d, e).
Extended Data Fig. 4 cIAP2−/− reverse chimeric mice exhibit greater post-MI injury.
a) Heart and lung weights normalized to tibia length from reverse bone marrow transplanted mice. b) LVESD measurements of reverse bone marrow chimeric mice. a)-b): Controls: N = 4; WT recipient MI: N = 8; cIAP2−/− recipient MI: N = 7. c) Estimation of capillary density by Isolectin B4 staining (green); nuclei (DAPI) – blue; scale bar = 50μM. N = 4 per treatment. Data presented are aggregates from two experiments. d) and e) Flow cytometry and cell quantitation from reverse bone marrow chimeric mice 28 days post-MI. Control operated mice: N = 3; MI-operated mice: N = 5 per group. Data presented are aggregates from two experiments. Error bars denote Mean +/- SD; p values were calculated using one-way ANOVA with Bonferroni’s correction.
Extended Data Fig. 5 Altered myeloid immune cell dynamics in cIAP2−/− mice.
a) (upper panel) cIAP2 upregulation was observed in patient blood leukocyte lysates from MI-sufferers, and not from control or non-MI, heart failure-diagnosed patients. (lower panel) Murine circulating leukocytes demonstrated increased cIAP2 protein expression Day 7 post-MI. b) Quantitation of leukocyte intracellular cIAP1 and cIAP2 protein expression from stable coronary artery disease (CAD) or MI patients using respective enzyme-specific immunoassay (ELISA). Outputs were normalized to whole protein (μg/ml) of cell lysate. N = 6 representative samples per group. c) Representative gating of peripheral blood myeloid (CD11b+) cells Day 3 post-MI. Representative gating d) and quantitation e) of peripheral blood B and T lymphocytes (B cells, CD4+ T cells, CD8+ T cells) from Day 3 post-MI WT and cIAP2−/− mice. f) Measurement of splenic lymphocytes collected 3- and 7- days post-MI. g) Measurement of LV infiltrating lymphocytes collected 7 days post-MI. h) Schematic diagram summarizing cardiac injury inducing leukocyte production and mobilization from the bone marrow and spleen, with subsequent infiltration into the heart being suppressed in the setting of cIAP2 deficiency. Error bars denote Mean +/- SD; p values were calculated using two-sided t-test (a) or one-way ANOVA followed by Bonferroni’s correction (c, d).
Extended Data Fig. 6 cIAP2 promotes MI-stimulated inflammatory cell signaling.
a) cIAP2 expression coincides with TRAF3 loss in splenocytes following MI. Data are representative of two separate experiments. b) Representative immunoblotting from mouse splenocyte homogenates 48 h post-MI showing expression changes of IAP proteins and TRAF3 in WT, cIAP2−/− or cIAP1−/− mice. c) Spleen expression of pro-inflammatory signal transduction intermediates from WT or cIAP2−/− mice 7 days post-MI. Immunoblot representative of three similar experiments. d) cIAP2 co-immunoprecipitation with TRAF3, an identified substrate for cIAP2 E3 ligase activity, from splenocytes Day 3 post-MI. Immunoblots are representative of three experiments, quantitation by densitometry relative to TRAF3. e) cIAP2 promotes TRAF3 (upper panel) K48 polyubiquitination (26S proteasomal targeting signal) and RIPK1 (lower panel) K63 polyubiquitination (activation/anti-apoptotic signal) in 24-hour MI-conditioned splenocytes. Blots of input protein lysate show TRAF3, RIPK1 cIAP1/2 expression as well as changes to non-cannonical NF-kB pathway factors (NF-κB-inducible kinase, IκB kinase-alpha, NF-kB2 (p100/52) and RelB). Representative of four experiments. f) Reduced nuclear translocation of NF-kB components RelA (p65) and NF-kB1 (p50) in cIAP2-deficient splenocytes isolated following MI. Lamin B is a nuclear antigen control. N = 3 per treatment group; image is representative of two similar experiments. g) Expression of inflammatory genes Tnfa, Il1b and Il6 from WT and cIAP2−/− splenocytes at Day 3 post-MI (N = 3/group). Values presented as expression normalized to respective strain sham controls. h) Schematic diagram depicting proposed role for cIAP2 following MI. Danger signal engagement drives cIAP2-mediated regulation of pro-inflammatory signaling, promoting NF-kB activity and promoting degradation of regulator TRAF3. Error bars denote Mean +/- SD; p values were calculated using two-sided t test (g).
Extended Data Fig. 7 TRAIL is upregulated by cIAP2−/− mice post-MI.
a) mRNA expression of Tnfa, Il1b, and IL6 normalized to HPRT from Day 7 post-MI WT or cIAP2−/− spleens. N = 3 per treatment group. b) Immunoblotting of splenocyte lysates from 7-day post-MI from WT or cIAP2−/− mice were immunoblotted for TRAIL (top) and TNFα, cleaved (active) Caspase 8 (p41 and p18), cleaved Caspase 3 (p17/19), Annexin A1 or vinculin. Imaging representative of two similar experiments, respectively. c) (Top panel) TRAIL and IRF1 protein expression from bone marrow cells collected from WT and cIAP2−/− mice 24-hours post-MI. (Bottom panel) Expression of TRAIL compared between dendritic cells (CD11c+) isolated from WT, cIAP2−/− and cIAP1−/− bone marrow 24-hours post-MI. d) High-resolution immunofluorescence microscopy of spleen sections detecting TRAIL and DR5 in cIAP2−/− mice 3 days post-MI; arrowheads indicate increased colocalized staining of TRAIL on cell surface of BST2+ splenocytes. Scale bar = 25 μm; images representative of three animals per treatment. e) Increased LVESD 28 days post-MI in both WT and cIAP2−/− MI recipient mice after anti-TRAIL (N2B2) antibody treatments. Control IgG2 recipients: N = 3 sham operated controls (WT); N = 4 WT and cIAP2−/− mice per MI treatment group. Anti-TRAIL recipients: N = 3 sham operated controls (WT); N = 5 WT and cIAP2−/− mice per MI treatment group. f) Flow cytometry of inflammatory cells from hearts (left panels) and spleens (right panels) of MI recipient mice following TRAIL neutralizing antibody treatment. cIAP2−/− mice exhibit more pronounced increases in cardiac tissues. Control IgG2 recipients: N = 3 sham operated controls (WT); N = 4 WT and cIAP2−/− mice per MI treatment group. Anti-TRAIL recipients: N = 3 sham operated controls (WT); N = 5 WT and cIAP2−/− mice per MI treatment group. g) Schematic summary of proposed activity of splenic cIAP2−/− pDCs following TNFα or death-promoter TNFSF ligand (TRAIL) signaling (via TNFR1 or DR5, respectively). Error bars denote Mean +/- SD; p values were calculated using two-sided t test (a), two-way ANOVA with Tukey’s test (e) or two-way ANOVA with Bonferroni’s correction (f).
Extended Data Fig. 8 LCL161 treatment reduces MI injury.
a) in vivo dose titration of LCL161. Peripheral blood cells were isolated 48 h following stimulation of mice with isoproterenol (10 mg/kg) and gavage of vehicle control or 2, 10 or 25 mg/kg of LCL161. Data shown is of representative 2 vehicle control-gavaged mice (top) or 10 mg/kg-gavaged LCL161 (bottom). b) Expression of inflammatory signal transduction intermediates by ex vivo-differentiated bone marrow-derived dendritic cells. Cell lysates were collected following 48-hour stimulation as indicated in presence or absence of LCL161. Immunoblot is representative of 2 similar experiments. c) flow cytometry gating for cell quantitation of monocytes or neutrophils from hearts of MI-treated C57Bl/6 mice receiving either vehicle (top) or LCL161 (10 mg/kg; bottom). Dotplot is representative of 5 mice per treatment. d) intracellular flow cytometry measuring cardiac Th17 cells from mice treated as in c). Controls: N = 2; MI-operated: N = 3 per group. e) LCL161-treated cIAP1−/−, but not cIAP2−/−, mice demonstrate improvement in ejection fraction, remodeling and heart weight improvement following MI. WT: MI + Vehicle: N = 5, MI + LCL: N = 5; cIAP1−/−: MI + Vehicle: N = 6, MI + LCL: N = 6; cIAP2−/−: MI + Vehicle: N = 8, MI + LCL: N = 7. Data are presented as aggregates from two representative experiments. f) reduced inflammatory cell infiltrate in hearts of cIAP1−/− mice treated with LCL161. Controls: N = 2; MI-operated: N = 4 vehicle treated; N = 5 LCL treated. g) Quantitative PCR of heart tissue isolates at 28 days post-MI for measurement of injury/inflammation gene transcription. N = 4/group for each target gene. h) Quantitation of spleen and bone marrow myeloid (CD11b+) cells Day 28 post-MI. Controls: N = 2/mouse strain; MI: N = 4/mouse strain. Error bars denote Mean +/- SD; p values were calculated using one-way ANOVA with Bonferroni’s correction (d, f) two-way ANOVA with Bonferroni’s correction (e) or two-sided t test (g).
Supplementary information
Supplementary Information (download PDF )
Supplementary Fig. 1.
Supplementary Data (download PDF )
Flow cytometry gating schematic.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 1 (download PDF )
Unprocessed immunoblots and unprocessed micrographs.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download PDF )
Unprocessed immunoblots and unprocessed micrographs.
Source Data Fig. 4 (download XLSX )
Statistical dource data.
Source Data Fig. 4 (download TIF )
Unprocessed immunoblots.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download PDF )
Unprocessed micrographs.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download TIF )
Unprocessed immunoblots.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download TIF )
Unprocessed immunoblots.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download TIF )
Unprocessed immunoblots.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 6 (download PDF )
Unprocessed immunoblots.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download PDF )
Unprocessed immunoblots.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 8 (download TIF )
Unprocessed immunoblots.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Smyth, D., Zhang, L., Al-Khalaf, M. et al. Hematopoietic expression of cIAP2 drives inflammation and heart failure after myocardial infarction. Nat Cardiovasc Res 5, 246–261 (2026). https://doi.org/10.1038/s44161-026-00782-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44161-026-00782-x