Abstract
Esophageal cancer, with its aggressive nature and high mortality, poses diverse epidemiological challenges worldwide. Over the past three decades, esophageal cancer has exhibited a substantial global burden, marked by a significant increase in absolute numbers, contrasting with a decline in age-standardized metrics. Prevalence nearly doubled, reaching 0.961 million in 2019, while the age-standardized rate (ASR) decreased to 11.6 per 100,000 cases. New incidence cases surged by 67.07%, yet the age-standardized incidence rate reduced to 6.5 per 100,000 cases. Deaths increased to 0.498 million, with a decline in age-standardized mortality to 6.1 per 100,000 cases. Disability-Adjusted Life Years (DALYs) rose to 11.67 million, but the ASR decreased to 139.8 per 100,000 cases. Gender-specific analysis revealed consistently higher rates in males, with increasing gaps over time. Correlations with SDI indicated a negative association, and frontier analysis underscored the impact of socio-economic progress on disease control. Projections suggest a continued rise in prevalence, incidence, deaths, and DALYs, with gender-specific variations. The research underscores the importance of continued efforts in public health and medical research to adapt to and manage the changing landscape of esophageal cancer globally.
Similar content being viewed by others
Introduction
Esophageal cancer, as a major global health concern, presents a diverse and complex epidemiological landscape1. Its incidence and mortality rates, exhibiting significant geographical variation, underscore the need for a comprehensive understanding of its global burden2. This malignancy, characterized by aggressive progression and poor prognosis, ranks among the leading causes of cancer-related deaths worldwide. The variability in its occurrence, influenced by a myriad of factors including dietary habits, lifestyle choices, and genetic predispositions, necessitates an in-depth exploration of its global, regional, and national impact2,3.
The burden of esophageal cancer extends beyond mere incidence and mortality rates. Its impact on public health is profound, with substantial implications for healthcare systems worldwide4. The disease not only leads to a significant loss of life but also imparts considerable morbidity among survivors5. This dual burden, encompassing both fatal and non-fatal outcomes, amplifies the need for a detailed analysis that goes beyond traditional epidemiological measures. Such an analysis must encompass a comprehensive assessment of the disease’s prevalence, its incidence trends, and the resultant mortality, alongside an evaluation of the broader impact encapsulated by Disability-Adjusted Life Years (DALYs).
Despite growing awareness and research efforts, there remains a gap in a systematic, multi-dimensional analysis of esophageal cancer at a global scale. Current literature often focuses on isolated aspects of the disease, lacking a holistic view that integrates various epidemiological metrics across different regions6,7,8. This gap hinders the formulation of effective global health strategies and policies, essential for addressing the challenges posed by esophageal cancer. Moreover, with changing global health dynamics and evolving risk factors, there is an urgent need for updated and comprehensive data that reflect current and future trends.
In light of these considerations, our study seeks to bridge this gap by leveraging the Global Burden of Disease (GBD) database to conduct a systematic and expansive analysis of esophageal cancer. By utilizing this robust and extensive dataset, we aim to provide a detailed exploration of the disease’s prevalence, incidence, deaths, and DALYs, alongside forecasting trends up to 2030. This approach not only enriches our understanding of esophageal cancer’s epidemiological patterns but also furnishes critical insights for public health decision-making and healthcare planning. Ultimately, our study endeavors to inform and shape the global response to this formidable health challenge, contributing significantly to the improvement of health outcomes for affected populations worldwide.
Methods
Study design and data source
Our research, grounded in a comprehensive analysis of esophageal cancer, utilized the 2019 GBD Study data. This study employed advanced statistical tools, including DisMod-MR 2.1, a meta-regression tool adept at integrating diverse epidemiological data, particularly for chronic diseases9,10. By applying a sophisticated modeling framework, we ensured a rigorous and thorough synthesis of the data, providing a robust foundation for our analysis.
Scope and variables
Focusing on esophageal cancer, our analysis centered on four primary variables: prevalence, incidence, deaths, and DALYs. We obtained data from the Global Health Data Exchange (GHDx), which offers comprehensive annual data from 1990 to 2019, segmented by age, sex, and location. Our study encompassed a broad range, covering 204 countries and territories. These were grouped into 21 regions based on geographical proximity and categorized into five clusters using the Socio-demographic Index (SDI) as a benchmark.
SDI
The SDI, a composite indicator reflecting a region’s socio-economic status, combines factors like income per capita, educational attainment, and fertility rates11. It ranges from 0 to 1, with higher values indicating better socio-economic conditions. We divided SDI into quintiles for detailed analysis.
Data analysis
Our initial data analysis involved calculating both raw and age-standardized rates (ASRs) for the primary outcome measures. We analyzed the variations in these metrics from 1990 to 2019 at global, regional, and national levels. The analysis also involved calculating the percentage of relative changes in prevalence, incidence, mortality, and DALYs over this period.
For temporal trend analysis, we employed Joinpoint regression analysis to detect statistically significant changes in esophageal cancer trends over time. This approach identifies “joinpoints,” points at which the slope of the trend significantly shifts, thus segmenting the entire study period into multiple sub-intervals. For each sub-interval, we calculated the Annual Percentage Change (APC) to quantify the magnitude and direction of changes. We then computed the Average Annual Percentage Change (AAPC) over predefined intervals, weighting segment-specific APCs by their respective time spans, in line with established methodologies12,13. Trends were deemed increasing if both the APC/AAPC estimate and its lower CI boundary were greater than zero, decreasing if both the APC/AAPC estimate and its upper CI boundary were less than zero, and stable otherwise. Additionally, we used Pearson correlation to examine the association between the SDI and various esophageal cancer indicators, providing insight into the intensity and direction of their linear relationships across diverse geographic settings.
To explore optimal benchmarks for esophageal cancer burden in relation to socio-economic development, we utilized a frontier analysis encompassing multiple disease indicators—including prevalence, incidence, deaths, and DALYs—to identify the lowest potentially achievable levels at any given SDI14,15. This approach employs a data envelope analysis using the free disposal hull method to derive non-linear frontiers, reflecting the minimal expected burden across these metrics for each SDI value. To account for uncertainty, we performed 1,000 bootstrapped samples with replacement, calculated mean values for each SDI, and applied LOESS regression (local polynomial degree 1, span 0.3) to generate a stable, smoothed frontier curve. Super-efficient outliers were excluded to prevent distortion. Countries or territories performing closer to this frontier are considered leaders in managing esophageal cancer relative to their development status, while those with greater distances from the frontier have greater scope for improvement.
To forecast esophageal cancer trends up to 2030, we employed the Bayesian Age-Period-Cohort (BAPC) model, which incorporates age, period, and cohort effects to capture temporal and generational influences on disease trends16. The model utilizes a second-order random walk (RW2) framework with prior distributions to ensure smoothness and account for similarities between adjacent effects17. By leveraging the integrated nested Laplace approximation (INLA) method, the BAPC model achieves efficient approximation of marginal posterior distributions, avoiding the mixing and convergence challenges often associated with traditional Markov Chain Monte Carlo techniques17. Statistical analyses were executed using R version 4.2.3.
Results
Global trends
Over the last three decades, the global esophageal cancer burden has seen a substantial increase in absolute numbers, yet a contrasting trend is observed in the age-standardized metrics. Prevalence nearly doubled from 0.489 million cases in 1990 to 0.961 million in 2019. Despite this rise, the age-standardized prevalence rate (ASPR) decreased from 12 to 11.6 per 100,000 individuals. New incidence cases surged by 67.07%, from 0.320 million to 0.535 million, while the age-standardized incidence rate (ASIR) saw a reduction from 8.1 to 6.5 per 100,000 individuals. The number of deaths also increased from 0.319 million to 0.498 million, yet the age-standardized mortality rate fell from 8.2 to 6.1 per 100,000 individuals. DALYs went up from 8.21 million to 11.67 million, but the ASR decreased from 199.3 to 139.8 per 100,000 individuals (Tables 1, 2, 3 and 4).
Temporal trends from joinpoint regression analyses reveal marked variations over the studied period. The prevalence of esophageal cancer showed distinct phases, with a sharp decrease in APC from 2004 to 2010 at -1.90%. Incidence rates witnessed a notable reduction, especially between 2004 and 2014 with an APC of -2.88%. Mortality trends also showed a significant decline in APC during the same period at -3.19%, and DALYs followed suit with the most substantial decrease in APC from 2004 to 2014 at -3.49%. The overall trend, as indicated by the AAPC, was negative for all measures: -0.14% for prevalence, -0.75% for incidence, -1.02% for mortality, and − 1.23% for DALYs (Fig. 1).
Global trends by sex
The ASR for both prevalence and incidence in males consistently exceeded that of females throughout the three-decade period. A declining trend in ASR over time is observed for both sexes, with the rates for males remaining notably higher. In terms of absolute numbers, a steady increase in cases for both prevalence and incidence among males, with a more pronounced rise observed in deaths and DALYs. Although females also exhibit an increase in cases, the gap between the sexes appears to widen over time, particularly in the number of deaths and DALYs (Figs. 2 and 3).
The ASR trends for esophageal cancer reveal gender-specific patterns in prevalence and incidence across the global spectrum. For prevalence, the ASR in males peaked around 2004 with a notable APC of 2.31% before declining, while females showed a similar trend with a peak APC of 2.10%. Subsequent years marked a decline in prevalence ASRs for both sexes, with females experiencing a sharper fall, particularly from 2010 to 2013 (APC = -3.75%). The incidence ASRs after 2004 decreased steadily for both genders; however, the fall was more pronounced in females with an APC of -4.26% from 2004 to 2013, compared to males with an APC of -2.18% from 2004 to 2010. The AAPC for both prevalence and incidence was − 0.14% and − 0.75%, indicating a gradual decrease over the study period (Fig. 4).
In examining the death rates and DALYs, both genders showed a downward trend post-2004. The death rates for females declined at an APC of -4.78% from 2004 to 2012, which was more substantial than the decline for males, with an APC of -2.65% from 2004 to 2014. The DALYs also displayed a consistent decrease, with females experiencing a sharper decline (APC = -5.38% from 2004 to 2009). The AAPC for DALYs stood at -1.95% for females and − 0.96% for males (Fig. 5).
Global trends by age group
The age-related distribution of esophageal cancer rates displays significant differences by sex across various age groups. The prevalence and incidence rates for males are considerably higher than for females, particularly in the older age brackets, with a stark increase observed from the age of 40–44 years and peaking in 70–74 years for prevalence and 85–89 years for incidence. The death rates from esophageal cancer follow a similar pattern, where rates escalate with age and are distinctly higher in males, especially pronounced in the 85–89 years age group. In terms of DALYs, the rate is substantially higher in males across all age groups. The DALYs rate dramatically rises from the 45–49 years age group onwards, accentuating the substantial burden of esophageal cancer in the older population (Fig. 6).
Regional trends
The regional trends in esophageal cancer from 1990 to 2019 reveal stark differences in disease dynamics across the 21 GBD regions. Central Asia reports the most pronounced decline in both prevalence and incidence rates, with AAPCs of -2.51% [95% CI, -2.76 to -2.26] and − 2.45% [95% CI, -2.65 to -2.25], respectively. While High-income Asia Pacific indicates a moderate rise in prevalence rates, Western Sub-Saharan Africa’s incidence has notably increased with an AAPC of 0.89% [95% CI, 0.78 to 0.99]. In contrast, Southern Latin America has made significant strides in reducing prevalence with an AAPC of -1.32% [95% CI, -1.57 to -1.07], and Central Latin America demonstrates a substantial decrease in incidence rates with an AAPC of -1.29% [95% CI, -1.63 to -0.95]. Despite an uptick in the number of cases, High-income North America shows a slight increase in the ASIR with an AAPC of 0.26% [95% CI, 0.17 to 0.35] (Tables 1 and 2).
Transitioning from prevalence and incidence to deaths and DALYs, the differences are equally pronounced. In Central Asia, the age-standardized death rate (ASDR) and DALYs have significantly decreased, with AAPCs of -2.43% and − 2.57%, respectively. Southern Latin America mirrors this positive trend with reductions in both ASDR (AAPC of -1.67%) and DALYs (AAPC of -1.83%). Conversely, Western Sub-Saharan Africa presents a contrasting scenario, with increases in both ASDR and DALYs (AAPCs of 0.9% and 0.79%, respectively), pointing to a growing burden of esophageal cancer. High-income North America displays a slight increase in the ASDR (AAPC of 0.19%), yet the rate of DALYs remains relatively stable (AAPC of -0.02%). Meanwhile, East Asia shows significant improvements, with notable decreases in both ASDR (AAPC of -1.79%) and DALYs (AAPC of -2.02%) (Tables 3 and 4)
National trends
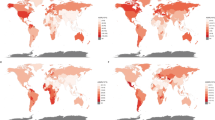
In the global landscape of esophageal cancer from 1990 to 2019, the trends in prevalence and incidence exhibit significant regional disparities. Among 204 countries, most witnessed a decrease in prevalence numbers, with Kazakhstan recording the steepest decline of -49.24%, while the United Arab Emirates (UAE) exhibited an unprecedented increase of 1183.22%. In ASRs, 107 countries, notably Turkmenistan with a -69.77% reduction, experienced a decline, contrasting with 97 countries where rates increased, the most significant being in a specific Asian region at 174.97%. The AAPC patterns align with these trends: 108 countries saw negative AAPCs, with Turkmenistan having the largest decline at -4.18 [-4.82 to -3.55], and 96 countries reported positive AAPCs. Incidence rates mirrored this variability, with 18 countries including the UAE showing notable increases, up to 1088.98%, and 186 countries, led by Kazakhstan with a -49.93% decrease, observing a decline. Turkmenistan again led the decrease in ASIRs with − 70.11%, while 78 countries, such as Northern Mariana Islands, saw an increase of 88.58%. The AAPC in incidence rates varied accordingly, with 127 countries displaying negative values, Turkmenistan recording the largest at -4.13 [-4.72 to -3.53], and 77 countries including the Netherlands showing positive values, the highest being 2.23 [1.84 to 2.62] (Tables S1–S2, Figs. S1–S6).
The deaths and DALYs due to esophageal cancer during the same period further highlights the varied impact across different regions. In terms of deaths, 19 countries including the UAE saw increases, with the UAE having the highest rise at 1044.14%, while a majority of 185 countries, such as Kazakhstan, experienced a decrease in numbers. The ASDRs reflect a similar pattern, with 136 countries, including Turkmenistan with the most significant decrease of -70.22%, witnessing a decline, and 68 countries, led by Northern Mariana Islands at an increase of 83.62%, showing upward trends. The AAPC in death rates mirrors these observations, with 137 countries showing negative AAPCs, Turkmenistan having the largest decrease at -4.14 [-4.72 to -3.56], and 67 countries recording positive values, Northern Mariana Islands being the highest at 2.08 [1.83 to 2.34]. DALYs followed a comparable trend, with the UAE showing the largest increase in numbers at 1128.5%, and Turkmenistan experiencing the most substantial decrease in ASRs at -70.32%. The AAPCs in DALYs rates varied widely, with 137 countries showing negative AAPCs, led by Turkmenistan at -4.14 [-4.75 to -3.52], and 67 countries displaying positive AAPCs (Tables S3–S4, Figs. S7–S12).
Correlations of ASR with SDI
The prevalence of esophageal cancer and incidence across 21 regions showed no statistically significant correlation with SDI, with p-values of 0.408 and 0.655, respectively. However, a significant negative correlation was found for deaths (R = -0.23, P = 0.316) and DALYs (R = -0.269, P = 0.239), although the p-values suggest these associations are not statistically significant. In the broader scope of 204 countries and territories, the ASR of prevalence exhibited a weak negative correlation with SDI (R = -0.112, P = 0.005), statistically significant. Incidence rates (R = -0.294, P < 0.001) and deaths (R = -0.354, P < 0.001) both showed moderate and significant negative correlations with SDI, indicating that higher SDI is associated with lower ASRs for both incidence and deaths. DALYs (R = -0.373, P < 0.001) also demonstrated a significant negative correlation with SDI (Figs. 7 and 8).
Frontier analysis
The frontier analysis of esophageal cancer from 1990 to 2019 highlights that as countries progress socioeconomically, the age-standardized prevalence and incidence rates of the disease tend to decrease. Specifically, for the year 2019, the analysis identified countries like Japan and the Netherlands as having the most significant effective differences in prevalence rates, far from the aspirational benchmark. Conversely, nations such as Niger, Mali, and Guinea exemplified leading performance, operating closer to the frontier. When examining the incidence rates in 2019, countries with higher SDIs such as Ireland and the United Arab Emirates showed a relatively high effective difference. In contrast, nations with lower SDIs, notably in regions of Africa, stood closer to the frontier, reflecting better-than-expected control of esophageal cancer incidence relative to their SDI (Fig. 9).
The pattern where higher SDI is often accompanied by lower ASRs for both deaths and DALYs of the disease was also discerned from 1990 to 2019. For the death rate and DALYs in 2019, countries with the largest gaps from the frontier, indicative of the greatest potential for improvement, included Greenland, Botswana, Mongolia and other 12 countries. In contrast, despite higher SDIs, Ireland, the United Kingdom and other countries had not reached their potential, as evidenced by their significant effective differences. Countries like Niger, Mali, and Gambia, despite lower SDIs, were closer to the frontier for deaths and DALYs (Fig. 10).
Trends of esophageal cancer from 1990 to 2030
The projections for esophageal cancer suggest a continued increase in prevalence and incidence numbers for both genders leading up to 2030, with a notably sharper rise among males. While the prevalence numbers climb steadily, the ASPRs are expected to see a gradual increase, showing a relative stabilization particularly in females. Incidence trends follow a similar pattern, with the male population driving a significant part of the anticipated increase post-2020, a trend that is likely to continue into the next decade. In contrast, the incidence growth for females is projected to be more tempered. The ASR for incidence also suggests a slight upward trend post-2020, with male rates seeing a modest rise and female rates tending towards stability, resulting in an overall incremental but controlled rise in ASR for both genders combined (Figs. 11 and 12).
The BAPC model forecasts a rise in the number of esophageal cancer-related deaths, with a pronounced escalation in males compared to a more gradual increase in females. The ASDRs for both genders indicate a trend towards stabilization after 2020. Similarly, for DALYs, the model predicts a continuous augmentation in numbers for both sexes, with males showing a more significant upward trend. The ASRs of DALYs are expected to see a slight elevation over the coming decade, primarily influenced by male trends, whereas female rates are projected to achieve relative stability (Figs. 13 and 14).
Discussion
Over the past three decades, the global landscape of esophageal cancer has evolved significantly, marked by an increase in absolute numbers but a decline in ASRs, indicative of improved healthcare interventions. Males consistently show higher rates than females in prevalence, incidence, deaths, and DALYs, with the disparity widening over time, particularly in mortality and DALYs. Age-wise, the burden intensifies with advancing age, especially in males. Regional analyses reveal notable variations: Central Asia has seen significant reductions in both prevalence and incidence, contrasting with rising trends in Western Sub-Saharan Africa and High-income North America. Nationally, while Turkmenistan exhibits significant decreases, the United Arab Emirates has experienced drastic increases. Correlation analyses suggest that higher SDI is associated with lower disease rates, a pattern reinforced by frontier analysis highlighting disparities in healthcare efficacy. Projections to 2030 indicate a continued rise in esophageal cancer cases, more pronounced in males, underscoring the need for enhanced public health strategies and healthcare resource allocation to address this escalating burden.
In the global trends of esophageal cancer, the dichotomy between the rise in absolute numbers of esophageal cancer cases and the decline in ASRs over the past three decades calls for a multifaceted interpretation. On the one hand, the doubling of prevalence and the significant surge in incidence cases reflect an undeniable growth in the disease’s burden. This increase could be attributed to various factors, including aging populations and heightened exposure to risk factors such as tobacco use, dietary patterns, and perhaps even environmental changes that are yet to be fully understood5,6,18,19. Additionally, socioeconomic changes, such as urbanization and shifts in dietary habits, could further explain these trends, particularly in regions where traditional diets are being replaced by processed foods, potentially increasing the risk of esophageal cancer. On the other hand, the decline in ASRs suggests that global health initiatives may be yielding positive results. Improvements in healthcare infrastructure, widespread availability of early detection methods, and more effective treatment options could be playing pivotal roles in reducing the per capita disease burden. Public health campaigns focused on reducing tobacco and alcohol consumption, along with better education about cancer risks, may also have contributed to these positive trends. These findings underscore the importance of continuing to focus on both prevention and treatment strategies, particularly in regions where the absolute burden of esophageal cancer continues to rise despite the decline in ASRs.
The gender disparity in incidence and mortality rates, with men consistently at higher risk, highlights potential biological susceptibilities and differences in exposure to risk factors such as tobacco and alcohol use18. The sharper decrease in female death rates and DALYs post-2004 may reflect targeted healthcare interventions and the efficacy of public health policies, underlining the necessity for gender-specific strategies. This disparity could also be influenced by differences in healthcare-seeking behaviors between men and women, with women possibly engaging more frequently in preventive health measures, which may contribute to earlier detection and better outcomes. As the global burden of esophageal cancer shifts, with rising cases but decreasing ASRs, future research and policy must focus on understanding and mitigating gender-based risk, improving diagnostic and treatment pathways, and ensuring that advancements in esophageal cancer care are equitable across all demographics. It will be critical to develop targeted interventions that address both biological and behavioral factors, ensuring that prevention efforts are tailored to the specific needs of each gender.
The age-specific analysis of esophageal cancer reveals a pronounced disparity between males and females across various age groups, underscoring the significant impact of gender and age on the disease’s epidemiology. Notably, males consistently exhibit higher rates of prevalence, incidence, deaths, and DALYs, with these rates escalating sharply after the age of 40, peaking in the 70–74 and 85–89 age groups for prevalence and incidence, respectively. This trend is particularly alarming in the elderly male population, where the rates of death and DALYs are exceptionally high, suggesting that older males constitute a high-risk group requiring targeted interventions and more rigorous screening protocols. The reasons behind this male predominance may be multifaceted, involving a combination of biological, lifestyle, and environmental factors, such as higher exposure to risk factors like tobacco and alcohol use in men. The dramatic rise in DALYs from the 45–49 years age group onward highlights the considerable burden of esophageal cancer in aging populations, emphasizing the need for age-specific healthcare strategies and resource allocation. It is also crucial to ensure that screening and prevention programs are accessible and tailored to older populations, as early detection in these high-risk age groups could significantly reduce the overall disease burden.
The regional and national trends in esophageal cancer from 1990 to 2019 depict a multifaceted global landscape, marked by varying disease dynamics across different geographies. Central Asia’s notable decrease in prevalence and incidence rates reflects the success of region-specific health interventions, contrasting sharply with the rising prevalence in Western Sub-Saharan Africa, which may indicate emerging health challenges or variations in risk factor exposure20,21. Southern Latin America’s significant reduction in prevalence and incidence highlights the effectiveness of localized health strategies, while the slight increase in ASIR in High-income North America despite a rise in cases suggests a complex interplay of healthcare advances and disease burden22. On a national scale, the dramatic rise in prevalence in the UAE and the steep decrease in Kazakhstan underscore the diverse impact of esophageal cancer across countries, further complicated by varied changes in ASRs as seen in Turkmenistan and a specific Asian region. These regional and national disparities, coupled with the varied trends in deaths and DALYs, emphasize the need for tailored esophageal cancer control strategies that consider the unique socioeconomic, healthcare, and lifestyle factors of each region23,24,25. Future efforts should focus on addressing the gaps in healthcare access and risk factor control, especially in regions experiencing rising trends, to better manage the global burden of esophageal cancer.
While the prevalence, incidence, deaths and DALYs of esophageal cancer across 21 regions did not show a significant correlation with SDI, the moderate and significant negative correlations observed in broader analyses across 204 countries indicate that higher SDIs are generally associated with lower ASRs for both incidence and deaths. This trend suggests that socioeconomic advancement may contribute to more effective health interventions and risk factor management, thereby reducing the ASR of esophageal cancer. Frontier analysis further accentuates this complexity; countries like Japan and the Netherlands, despite high SDIs, show significant effective differences in prevalence rates, indicating a gap in achieving the lowest possible burden of the disease. In contrast, countries with lower SDIs, such as Niger, Mali, and Guinea, demonstrate better-than-expected control relative to their development status. This disparity highlights that while economic and social progress is a crucial component, it is not the sole determinant in the fight against esophageal cancer. A comprehensive approach, considering both socioeconomic and health system factors, is imperative to effectively tackle the global burden of this disease.
The projected trends of esophageal cancer up to 2030 depict an escalating burden, particularly emphasized by a more pronounced increase in prevalence and incidence among males. This gender disparity signals underlying differences in exposure to risk factors, potentially necessitating gender-specific public health interventions26,27. While prevalence numbers steadily rise, the ASRs exhibit a relative stabilization, especially in females, suggesting effectiveness in early detection and management strategies. However, the anticipated rise in incidence, predominantly driven by male trends post-2020, highlights the urgent need for enhanced prevention and screening programs28,29. The forecasted increase in esophageal cancer-related deaths, with males experiencing a steeper escalation, underscores a pressing need for improved therapeutic and palliative care approaches1. Additionally, the predicted growth in DALYs for both sexes, more so in males, reflects not only the increasing incidence and mortality but also the prolonged impact of the disease on quality of life30,31,32. These trends collectively point towards the necessity of a multifaceted approach in combating esophageal cancer, encompassing prevention, early detection, effective treatment, and supportive care, tailored to address the distinct patterns observed in both genders.
Our study has several limitations. First, there are inherent uncertainties in the predictive models due to evolving risk factors, advancements in diagnostic and screening methodologies, and the possibility of unforeseen global health events. Second, relying predominantly on historical data may not fully reflect emerging trends or the influence of recent healthcare innovations. Third, geographical variability in disease prevalence and healthcare infrastructure could limit the generalizability of our findings across different regions. Moreover, variations in data collection methods and the absence of detailed histological stratification in some sources may introduce bias, potentially affecting the interpretation and comparability of results. Lastly, as global lifestyles shift—most notably with rising obesity rates—changing risk profiles may alter the patterns of disease incidence and outcomes over time. Future research should aim to address these limitations by improving data quality and granularity, accounting for regional differences, and refining predictive models to remain robust in the face of evolving epidemiological landscapes.
Conclusions
Our study highlights a rising global burden of esophageal cancer up to 2030, with a notable increase in cases, especially among males. Despite advancements in healthcare leading to stabilized ASRs, the escalating prevalence and incidence underscore the need for enhanced prevention and targeted treatment strategies. The findings stress the importance of addressing gender disparities and regional variations in disease dynamics, guiding future healthcare policies and research efforts towards effective management and reduction of esophageal cancer’s impact worldwide.
The APC and AAPC of ASR for the prevalence (A), incidence (B), deaths (C) and DALYs (D) associated with esophageal cancer at the global level based on the joinpoint regression analysis model.
The trends in ASR of prevalence (A), incidence (B), deaths (C), and DALYs (D) for esophageal cancer among different sexes, female and male, from 1990 to 2019.
The trends in numbers of prevalence (A), incidence (B), deaths (C), and DALYs (D) for esophageal cancer among different sexes, female and male, from 1990 to 2019.
The APC and AAPC of ASR for prevalence (A) and incidence (B) by gender (both, female and male) in esophageal cancer at the global level based on the joinpoint regression analysis model.
The APC and AAPC of ASR for deaths (A) and DALYs (B) by gender (both, female and male) in esophageal cancer at the global level based on the joinpoint regression analysis model.
Comparison of prevalence (A), incidence (B), deaths (C), and DALYs (D) for esophageal cancer across different genders (female and male) and age groups ranging from under 5 years to 95 + years.
Pearson correlation analysis between the SDI and ASR of prevalence (A), incidence (B), deaths (C), and DALYs (D) for esophageal cancer across 21 regional levels in 2019 (The cases of prevalence, incidence, deaths, and DALYs from 21 regions in 2019 are represented by circles. The size of the circles increased with the cases of prevalence, incidence, deaths, and DALYs).
Pearson correlation analysis between the SDI and ASR of prevalence (A), incidence (B), deaths (C), and DALYs (D) for esophageal cancer at the country and territorial levels in 2019 (The cases of prevalence, incidence, deaths, and DALYs from 204 countries and territories in 2019 are represented by circles. The size of the circles increased with the cases of prevalence, incidence, deaths, and DALYs).
Frontier analysis, represented by the solid black lines, depicted through solid black lines, delving into the interplay between the SDI and ASR for prevalence (A, B) and incidence (C, D) concerning esophageal cancer. The color spectrum in graphs (A) and (C) serves as a visual timeline, transitioning from lighter shades signifying 1990 to the deepest hues marking 2019. In graphs (B) and (D), each data point symbolizes a distinct country or territory for the year 2019, with the top 15 nations manifesting the most pronounced deviation from the frontier highlighted in black. Countries characterized by a low SDI (< 0.455) and minimal departure from the frontier are accentuated in blue, whereas those with a high SDI (> 0.805) and conspicuous divergence reflective of their developmental standing are underscored in red. The color of the dots delineates the directional shift from 1990 to 2019 in ASR: decrease dots denote a decrease, while increase dots signify an increase.
Frontier analysis, represented by the solid black lines, depicted through solid black lines, delving into the interplay between the SDI and ASR for deaths (A, B), and DALYs (C, D) concerning esophageal cancer. The color spectrum in graphs (A) and (C) serves as a visual timeline, transitioning from lighter shades signifying 1990 to the deepest hues marking 2019. In graphs (B) and (D), each data point symbolizes a distinct country or territory for the year 2019, with the top 15 nations manifesting the most pronounced deviation from the frontier highlighted in black. Countries characterized by a low SDI (< 0.455) and minimal departure from the frontier are accentuated in blue, whereas those with a high SDI (> 0.805) and conspicuous divergence reflective of their developmental standing are underscored in red. The color of the dots delineates the directional shift from 1990 to 2019 in ASR: decrease dots denote a decrease, while increase dots signify an increase.
Projected numbers and ASR of prevalence for esophageal cancer by gender (both, male, and female) from 2020 to 2030 based on the BAPC model.
Projected numbers and ASR of incidence for esophageal cancer by gender (both, male, and female) from 2020 to 2030 based on the BAPC model.
Projected ASR (A) and numbers (B) of deaths for esophageal cancer by gender (both, male, and female) from 2020 to 2030 based on the BAPC model.
Projected ASR (A) and numbers (B) of DALYs for esophageal cancer by gender (both, male, and female) from 2020 to 2030 based on the BAPC model.
Data availability
The data comes from a public database, can through this link: https://vizhub.healthdata.org/gbd-results/ for the relevant data.
References
Smyth, E. C. et al. Oesophageal cancer. Nat. Rev. Dis. Primers. 3, 17048 (2017).
Morgan, E. et al. The global landscape of esophageal squamous cell carcinoma and esophageal adenocarcinoma incidence and mortality in 2020 and projections to 2040: new estimates from GLOBOCAN 2020. Gastroenterology 163 (3), 649–658 (2022).
Domper, A. M., Ferrández, A. Á. & Lanas, A. Á. Esophageal cancer: Risk factors, screening and endoscopic treatment in western and eastern countries. World J. Gastroenterol. 21(26), 7933–7943 (2015).
Thrift, A. P. Global burden and epidemiology of Barrett oesophagus and oesophageal cancer. Nat. Rev. Gastroenterol. Hepatol. 18(6), 432–443 (2021).
Goh, K. J. et al. Characteristics and outcomes of airway involvement in esophageal cancer. Ann. Thorac. Surg. 112(3), 912–920 (2021).
Menya, D. et al. Traditional and commercial alcohols and esophageal cancer risk in Kenya. Int. J. Cancer 144(3), 459–469 (2019).
Roerecke, M. et al. Estimates of alcohol-related oesophageal cancer burden in Japan: Systematic review and meta-analyses. Bull. World Health Organ. 93(5), 329–338 (2015).
Li, S. et al. Changing trends in the disease burden of esophageal cancer in China from 1990 to 2017 and its predicted level in 25 years. Cancer Med. 10(5), 1889–1899 (2021).
Global National burden of spinal cord injury, 1990–2019: A systematic analysis for the global burden of disease study 2019. Lancet Neurol. 22 (11), 1026–1047 (2023).
Li, C. et al. The global incidence and disability of eye injury: An analysis from the global burden of disease study 2019. eClinicalMedicine 62, 102134 (2023).
Global burden. Of 369 diseases and injuries in 204 countries and territories, 1990–2019: a systematic analysis for the global burden of Disease Study 2019. Lancet 396 (10258), 1204–1222 (2020).
Yang, F. et al. Thirty-year trends of depressive disorders in 204 countries and territories from 1990 to 2019: An age-period-cohort analysis. Psychiatry Res. 328, 115433 (2023).
Cao, F. et al. Trends and cross-country inequalities in the global burden of osteoarthritis, 1990–2019: A population-based study. Ageing Res. Rev. 99, 102382 (2024).
Ruan, R. et al. Global, regional, and national advances toward the management of rheumatic heart disease based on the global burden of Disease Study 2019. J. Am. Heart Assoc. 12(13), e28921 (2023).
Xie, Y. et al. Analysis of the global burden of disease study highlights the global, regional, and national trends of chronic kidney disease epidemiology from 1990 to 2016. Kidney Int. 94(3), 567–581 (2018).
Du, Z., Chen, W., Xia, Q., Shi, O. & Chen, Q. Trends and projections of kidney cancer incidence at the global and national levels, 1990–2030: A bayesian age-period-cohort modeling study. Biomark. Res. 8, 16 (2020).
Hu, W., Fang, L., Zhang, H., Ni, R. & Pan, G. Global disease burden of COPD from 1990 to 2019 and prediction of future disease burden trend in China. Public Health 208, 89–97 (2022).
Pelucchi, C., Gallus, S., Garavello, W., Bosetti, C. & La, V. C. Alcohol and tobacco use, and cancer risk for upper aerodigestive tract and liver. Eur. J. Cancer Prev. 17(4), 340–344 (2008).
Andrici, J. & Eslick, G. D. Hot food and beverage consumption and the risk of esophageal cancer: A meta-analysis. Am. J. Prev. Med. 49(6), 952–960 (2015).
Allemani, C. et al. Global surveillance of trends in cancer survival 2000-14 (CONCORD-3): Analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 391(10125), 1023–1075 (2018).
Middleton, D. R. et al. Hot beverages and oesophageal cancer risk in western Kenya: Findings from the ESCCAPE case-control study. Int. J. Cancer 144(11), 2669–2676 (2019).
Eyck, B. M. et al. Ten-year outcome of neoadjuvant chemoradiotherapy plus surgery for esophageal cancer: The randomized controlled CROSS trial. J. Clin. Oncol. 39(18), 1995–2004 (2021).
Lu, L., Mullins, C. S., Schafmayer, C., Zeißig, S. & Linnebacher, M. A global assessment of recent trends in gastrointestinal cancer and lifestyle-associated risk factors. Cancer Commun. (Lond.) 41(11), 1137–1151 (2021).
Maret-Ouda, J., Markar, S. R. & Lagergren, J. Gastroesophageal reflux disease: A review. JAMA 324(24), 2536–2547 (2020).
Wang, N. et al. The effect of socioeconomic status on health-care delay and treatment of esophageal cancer. J. Transl. Med. 13, 241 (2015).
Zeng, H. et al. Disparities in stage at diagnosis for five common cancers in China: A multicentre, hospital-based, observational study. Lancet Public Health 6(12), 877 (2021).
Lee, J. H. et al. Increased variation in esophageal cancer treatment and geographic healthcare disparity in Michigan. J. Am. Coll. Surg. 237(5), 779–785 (2023).
Patel, A. V. et al. American college of sports medicine roundtable report on physical activity, sedentary behavior, and cancer prevention and control. Med. Sci. Sports Exerc. 51(11), 2391–2402 (2019).
Zakko, L., Lutzke, L. & Wang, K. K. Screening and preventive strategies in esophagogastric cancer. Surg. Oncol. Clin. N. Am. 26(2), 163–178 (2017).
Toh, Y. et al. Health-related quality of life after esophagectomy in patients with esophageal cancer. Esophagus 19 (1), 47–56 (2022).
Jezerskyte, E. et al. Long-term quality of life following transthoracic and transhiatal esophagectomy for esophageal cancer. J. Gastrointest. Surg. 25(7), 1657–1666 (2021).
Katz, A. et al. Long-term quality of life after esophagectomy for esophageal cancer. Ann. Thorac. Surg. 115(1), 200–208 (2023).
Acknowledgements
Our team extends heartfelt thanks to all the contributors and collaborators of the GBD Study, whose exhaustive datasets were instrumental in our research. We express our sincere appreciation to the data analysts and statisticians for their detailed work in analyzing the data, skillfully employing R software and robust statistical methods like joinpoint regression analysis and the BAPC model. We are also grateful to our fellow researchers and healthcare experts whose invaluable input and perspectives significantly deepened our analysis and understanding of the trends in esophageal cancer.
Funding
This work was supported by the National Natural Science Foundation (Nos. 81972864, 82172720), Shandong Provincial Natural Science Foundation (ZR2020LZL018), CSCO-Nav HER2-related Solid Tumors Research Foundation (Y-2022HER2AZMS-0291).
Author information
Authors and Affiliations
Contributions
Liangchao sun: Data curation, Formal analysis, Investigation, Methodology, Validation, Visualization, Roles/Writing - original draft, Writing - review & editing. Kaikai Zhao: Investigation, Validation. Xiaoli Liu: Conceptualization, Data curation, Formal analysis, Methodology, Resources, Software, Visualization, Roles/Writing - original draft, Writing - review & editing. Xue Meng: Funding acquisition, Project administration, Supervision, Writing - review & editing.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Our research involved a secondary evaluation of the publicly accessible GBD Study dataset, without primary data collection. Hence, no ethical approval was necessary.
Informed consent
No informed consent was required since our research is a secondary analysis of public data.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Sun, L., Zhao, K., Liu, X. et al. Global, regional, and national burden of esophageal cancer using the 2019 global burden of disease study. Sci Rep 15, 3284 (2025). https://doi.org/10.1038/s41598-025-86244-z
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-86244-z
Keywords
This article is cited by
-
Optimization and characterization of acute radiation-induced esophagitis in mice
Radiation Oncology (2025)
-
MRI outperforms CT for tracheal and vascular invasion staging in esophageal cancer
European Radiology (2025)