Abstract
The cardiometabolic index (CMI), which integrates individual lipid and visceral fat parameters, represents a superior new predictive tool for cardiovascular and metabolic disorders, but its predictive value for gallstones (GS) is unclear. Therefore, the present study used this vacancy to explore the relationship between CMI levels and GS in US adults, assess the mediating role of diabetes mellitus in the pathogenesis of both, and provide new clinical ideas for early prevention and screening of GS in patients with diabetes mellitus. This survey extracted information from the National Health and Nutrition Examination Survey (NHANES) 2017–2020 vintage cycle. Cross-sectional analyses and a variety of statistical techniques were used to analyze the correlation between CMI and GS, including logistic regression, propensity score matching, subject work curves, and restricted cubic spline (RCS). Furthermore, mediation analysis was used to investigate whether and to what extent diabetes mediated the effect of CMI on GS. After analysis of 3,395 participants, a significant positive correlation was observed between elevated CMI levels and increased prevalence of GS In the fully corrected model (Model 4), the prevalence of GS exhibited a 23% increase for every incremental unit rise in logarithmically transformed CM. Mediation analysis showed that diabetes largely mediated the association between CMI and GS, with a mediation ratio of 15.1%. Higher CMI levels are closely linked to a greater occurrence of GS, and diabetes plays a key mediating role in the pathogenesis of both. Addressing dyslipidemia while not neglecting blood glucose levels, and co-management of the two may be a proven way to reduce GS risk.
Similar content being viewed by others
Introduction
Gallstones (GS) represent a prevalent gastrointestinal ailment globally, with a rising prevalence in recent times1. Approximately 10 to 15% of people in developed societies suffer from GS2,3. The United States witnesses over 800,000 cholecystectomies annually, incurring a substantial economic strain of nearly $6.5 billion yearly on households and the healthcare sector4,5,6. The intricate processes leading to GS formation encompass genetic predispositions, gallbladder contractile dynamics, microbiota, estrogen, and dietary factors7,8,9. Recent research indicates that the presence of GS can elevate the likelihood of developing additional health conditions, including cardiovascular diseases, pancreatitis, and malignant tumors, posing a serious threat to the individual’s health and overall quality of life10,11. Consequently, identifying and utilizing simple and controllable clinical indicators are imperative for effectively preventing and managing GS.
Diabetes is a complex chronic metabolic disorder affecting a global population of 425 million individuals, with projections indicating a rise to 629 million by the year 204512,13. Prior research has established that obesity is a significant contributor to the onset of various metabolic disorders, notably diabetes. Obesity can be assessed using a range of metrics, such as surrogate markers of general or central adiposity, like body mass index (BMI) or waist circumference (WC)14,15. It has been suggested that waist-to-height ratio (WHtR) serves as a more precise and responsive indicator of cardiovascular disease and diabetes compared to the conventional measure of obesity, given its capacity to discern between muscle mass and fat distribution. WHtR is a measure of visceral obesity that is calculated by dividing WC by height. It is a more effective indicator of cardiometabolic risk and various non-communicable diseases compared to traditional measures of obesity16,17,18,19. Additionally, several markers of lipid metabolism in humans, such as triglyceride (TG) to high-density lipoprotein cholesterol (HDL-C) ratio, lipid accumulation products (LAP), and triglyceride-glucose (TyG) index20, have been recognized as reliable and practical indicators of insulin resistance and metabolic diseases21.
Cardiovascular metabolic index (CMI) is defined by multiplying TG/HDL-C by WHtR, a novel metabolic index introduced by Ichiro Wakabayashi and colleagues22,23. CMI combines lipid and obesity parameters and is a reliable indicator for early identification of cardiovascular disease and diabetes. Numerous recent studies have confirmed a clear correlation between CMI and a variety of diseases such as coronary heart disease, stroke, atherosclerosis, hypertension, left ventricular dilatation, and kidney disease. In terms of predicting metabolic and cardiovascular diseases, CMI may be a promising clinical indicator23,24,25,26. Nevertheless, the relationship between CMI and GS and the prognostic utility of CMI in predicting GS remains unexplored in the literature. The major aim of this research was to examine the relationship between CMI levels and GS in the US adult population and to assess the possible mediating role of diabetes between the two, to provide new clinical ideas for early prevention and screening of GS in patients with diabetes.
Methods
Data collection and study subjects
The National Health and Nutrition Examination Survey (NHANES) constitutes a comprehensive and methodologically rigorous study of the U.S. population, utilizing advanced sampling techniques to gather extensive data on health indicators and diseases. This is achieved through personal interviews, standardized physical examinations, and laboratory tests. Administered and supervised by the National Center for Health Statistics (NCHS), NHANES provides a collection of nationally representative cross-sectional data. The database encompasses a broad spectrum of biological and medical domains, rendering it an invaluable resource for healthcare professionals and public health initiatives. The collection and distribution of NHANES data followed the Helsinki Principles and were approved by the NCHS Ethics Committee, with all participants signing informed consent forms.
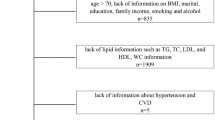
Initially, individuals with missing GS data and those under the age of 20 (n = 6,350) were excluded. Additionally, pregnant women, individuals lacking information on education and marital status, and those with incomplete lipid data were also excluded. Furthermore, participants with missing disease history, dietary data, and incomplete information on covariates, including other physical examinations and laboratory tests, were not considered for inclusion. Following a rigorous assessment and screening process, a total of 3,395 adult participants were recruited for this study. Figure 1 illustrates the participant exclusion and inclusion process in detail and provides comprehensive information.
NHANES 2017–2020 participant selection flowchart.
Diagnosis and definition of GS and diabetes
The assessment of GS was conducted using a questionnaire in which participants responded to the query, “Did your doctor diagnose you with GS?” A response of “yes” was taken as an indication of the presence of GS. This strategy is both uncomplicated and effective, having been extensively applied in past research of a similar nature27,28. A diagnosis of diabetes is established through the presence of a glycosylated hemoglobin level equal to or greater than 6.5%, confirmation by a medical practitioner or healthcare provider, self-reported acknowledgment of the condition, and/or use of medication or insulin to control your blood sugar29.
Calculation of the CMI, LAP and TyG
The determination of CMI, LAP, and TyG necessitates the acquisition of fasting blood samples from participants, along with physical measurements conducted within a laboratory environment30. To ensure the promptness and precision of biochemical marker assessments, blood samples are obtained either via a dedicated mobile screening unit or at a designated location, followed by processing and analysis in adherence to a standardized protocol. Certified medical personnel at the mobile screening centers are tasked with measuring participants’ waist circumference and height. The WHtR is subsequently calculated by dividing waist circumference (in centimeters) by height (in centimeters). The ratio of total cholesterol to HDL cholesterol is utilized as an indicator of the relationship between these two cholesterol levels31. The formulae for the relevant indicators are set out below:
Covariates included in the study
Considering the possible influence of other confounding variables on GS, this study incorporated a comprehensive set of covariates informed by expert clinical knowledge and previous literature. The primary variables in the demographic model include age, sex, race, marital status, and socioeconomic status. Socioeconomic status encompasses educational level and the ratio of household income to poverty (PIR). Lifestyle habits were predominantly characterized by smoking and alcohol consumption. Dietary intake factors comprised carbohydrate, protein, fat, and water intake. BMI and total plasma cholesterol were identified as significant confounding variables among the physical examination and laboratory test indices. The history of existing diseases incorporated covariates (hypertension, diabetes, asthma, coronary heart disease, liver disease, and cancer). Information regarding medication use was ascertained based on whether the subject’s physician had told the use of antihypertensive or lipid-lowering medications (statins and fibrates) as reported in the questionnaire32.
Statistical analysis
Based on the statistical properties of the variables, the baseline characteristics of continuous variables were represented using either parametric tests (mean ± standard deviation) or nonparametric tests (median and interquartile range), while categorical variables were described using absolute values (n) and percentages (%). Stepwise regression analysis was employed to initially screen all covariates, construct a matrix of correlation coefficients, and assess the multicollinearity of the covariates using the condition number (κ) of the independent variable matrix. A condition number κ < 100 indicates a low degree of multicollinearity; κ between 100 and 1000 indicates some degree of multicollinearity; κ > 1000 indicates severe multiple multicollinearity. All covariates included in this study have κ values less than 10033. To thoroughly examine the relationship between CMI, LAP, the TyG index, and GS, four distinct models were developed utilizing one-way and multifactor logistic regression analyses. Model 1 served as the baseline single-factor model, excluding any covariates. Model 2 was adjusted for essential demographic variables, namely sex, age, and race. Building upon the foundation of Model 2, Model 3 incorporated additional variables such as socioeconomic status (education and PIR), marital status, BMI, total cholesterol, lifestyle behaviors (smoking and alcohol consumption), substance use, and medical history. Lastly, Model 4 further extended the analysis by including daily dietary intake as a covariate, resulting in a fully integrated model.
Due to their skewed distributions, CMI, TG/HDL and LAP were log-transformed before statistical analyses were performed and were treated as continuous variables (per 1-unit increment) in the aforementioned models34. The dose-response relationships among CMI, TG/HDL, LAP, TyG, and GS were examined through the application of restricted cubic spline (RCS) curve fitting to ascertain whether these relationships were linear or nonlinear. Hierarchical multiple regression was used in subgroup analyses to investigate possible differences in the relationship between CMI and GS among certain populations. To thoroughly assess heterogeneity among subgroups, particularly concerning gender and race, interaction terms were incorporated into the model and analyzed via the log-likelihood ratio test. Sensitivity analyses were performed to confirm the robustness of the findings. Furthermore, for trend analysis, CMI was converted from a continuous variable into quartiles. Additionally, CMI was categorized into dichotomous groups (high CMI and low CMI) for propensity score matching (PSM). Optimal pairwise matching ratios were employed, with matching variables including gender, age, and race. Finally, the four models were analyzed for causal mediation using the “mediation” software package, and 500 simulations were conducted using the Bootstrap method to assess the confidence intervals of the mediated effects to determine the proportion of the mediated effects attributable to diabetes. The model not only offers statistical evidence for rational analysis but also proves to be effective in uncovering pathways. Specifically, direct effects demonstrate the correlation between CMI and GS, while indirect effects ascertain whether this correlation is influenced by diabetes. Statistical analyses for this study were based on R and MSTATA software, with a predetermined level of significance set at a P value of less than 0.05.
Results
Clinical baseline characteristics of patients with GS
Table 1 compares the clinical characteristics of subjects with and without GS. Of the 3,395 subjects who participated in the study, 363 were diagnosed with GS, with an overall prevalence of 10.69%. The participants had a median age of 52 years, consisting of 49.10% men and 50.90% women. Regarding disease history, 45.18%, 4.27%, and 18.82% of the total were hypertensive, coronary artery disease, and diabetic patients respectively.
Table 2 presents the clinical baseline characteristics of the participants, stratified by gender. The data indicate a notably higher prevalence of GS in females compared to males, with prevalence rates of 15.05% and 6.18%, respectively. Conversely, the prevalence of diabetes mellitus and coronary heart disease is greater in males than in females. Additionally, the TG/HDL ratio, CMI, and TyG index are marginally elevated in males compared to females, with all associated P-values being less than 0.01. In contrast, the LAP values are equivalent between the two groups.
Association between CMI, TG/HDL, LAP, TyG index, and GS
Table 3 shows the results of univariate and multivariate logistic regression analyses between these indicators of CMI, TG/HDL, LAP and TyG and GS. A total of 4 Models were constructed and these results were based on the stepwise inclusion of different covariates. Univariate Model 1 and multivariate Model 2 (adjusted for adjustment for key demographic variables) showed statistically significant positive correlations between the four indices and GS (all P-values were less than 0.01). Model 3 further adjusted for additional factors including marital status, socioeconomic status (education level and PIR), BMI, medical history (hypertension, diabetes mellitus, coronary heart disease, asthma, cancer), total cholesterol, lifestyle habits, and medication use, based on Model 2. The positive correlations between three of the indices and GS remained significant, except for the TyG index (all P-values < 0.05). Upon incorporating all potential confounders in Model 4, only CMI and LAP retained statistically significant associations. Specifically, for each 1-unit increase in log-transformed CMI, the prevalence of GS increased by 23% (OR = 1.23; 95% CI 1.03–1.46), while LAP exhibited an even stronger association (OR = 1.37; 95% CI 1.11–1.69).
Subgroup analysis
To assess the heterogeneity between CMI and GS across subgroups, we performed subgroup analyses and interaction tests, the results of which are shown in Fig. 2. Notably, the positive correlation between CMI and GS was more pronounced in the subgroups of females, non-Hispanic other races, those with higher education or higher education, and those who drank alcohol (all P-values were below 0.05). Interestingly, interaction tests revealed a significant interaction effect for gender (P for interaction = 0.046). In contrast, no statistically significant associations were found in the subgroups regardless of whether alcohol was consumed or not, or whether diabetes was present.
Subgroup analyses and interaction tests of the relationship between CMI and GS. Notes: Adjusted for all covariates except effect modifier.
Sensitivity analyses
To confirm the stability of the findings, sensitivity analyses were performed and the results are shown in Table 4. CMI was further converted from a continuous variable to quartiles and tested for trend. The results showed that in Model 4, which fully adjusted for all covariates, the prevalence of GS increased by 91% (OR = 1.91; 95% CI 1.25–2.98) in participants with the highest quartile of CMI, using the lowest quartile of CMI as the reference group, with a statistically significant effect of the test for trend (P for trend = 0.013). Furthermore, we performed supplementary analyses employing propensity score matching with a 1:1 matching ratio, utilizing gender, age, and ethnicity as matching variables. As demonstrated in Supplementary Table 1, there was an observed 88% increase in the prevalence of GS among individuals with high CMI compared to those with low CMI following matching (OR = 1.88; P < 0.001).
RCS curve fitting analysis
The RCS smoothing curve fitting is quite intuitive and can be visualized through adaptable modeling, as depicted in Fig. 3. Subfigures 3 A through 3D show the dose-response relationship between CMI, TG/HDL, LAP, TyG changes, and GS, respectively. The results showed a statistically significant nonlinear positive correlation between CMI, LAP, and GS (P-overall < 0.05), with the nonlinear component not dominating (P-nonlinear > 0.05). In contrast, the linear positive correlation between TG/HDL, TyG index, and GS was higher (P- overall > 0.05).
RCS curve fitting between CMI and GS. Solid lines represent smooth curve fits between variables. Shaded bands represent 95% confidence intervals from the fit. Subfigure 3 (A): dose-response relationship between CMI and GS; Subfigure 3(B): dose-response relationship between TG/HDL and GS; Subfigure 3(C): dose-response relationship between LAP and GS; Subfigure 3(D): dose-response relationship between TyG and GS.
The mediating role of diabetes
Mediation analyses investigated whether and to what extent diabetes mediated the effect of CMI on GS. Figure 4 visually illustrates the pathways and patterns of mediation analyses. In terms of the overall population, all three effects (including total, direct, and indirect effects) were significant (P < 0.01), suggesting that diabetes mediated the relationship to some extent and that the mediating proportion was as high as 15.1%. Interestingly, the mediating effect was not significant in the male group, whereas it remained significant in the female group. The results of the mediation analyses are summarized in Table 5.
Modal pathways for mediated analyses. CMI is defined as the independent variable; GS is the outcome variable; and diabetes is the mediating variable. Path a represents the regression coefficient for the relationship between CMI and diabetes. Path b represents the regression coefficient of the relationship between diabetes and GS. Path c represents the simple total effect of CMI on GS. Path c’ represents the direct effect of CMI on GS after controlling for diabetes.
Discussion
This study provides a new perspective on the mediating role of diabetes between CMI and GS, and, at the same time, cross-sectionally compares the predictive ability of several similar metrics (LAP, TG/HDL, TyG) for GS. Through cross-sectional analyses of 3,395 adult participants, a significant association was observed between high levels of CMI and the GS population, with diabetes playing a substantial role in mediating this relationship. Furthermore, regression analyses from Model 2 to Model 4 revealed that CMI remained independently associated with GS even after controlling for various potential confounding factors. As part of the sensitivity analyses, subgroup stratification analyses and propensity score matching visually verified the stability of the positive correlation between the two, particularly in the subgroups of females, non-Hispanics of other races, those with high levels of education or attainment, and drinkers. In addition, mediation analyses suggested that diabetes may be a mediator and bridge to the development of GS in people with higher CMI. Overall, increased CMI raised the risk of GS to some extent, and this positive association was mediated in part by diabetes. This finding provides new information on the metabolic risk factors for GS and deepens our understanding of the intricate interactions between diabetes and dyslipidaemic diseases.
In recent years, the interaction of cardiovascular and metabolic diseases with GS has received extensive attention from scholars. In an 8-year prospective cohort study conducted by Wirth et al. in Europe, involving 46,486 participants, it was demonstrated that the risk of cardiovascular disease was 24% higher in individuals with GS compared to those without GS11. Following and meta-analyzing 270,000 participants from the United States, Zheng and colleagues found that subjects with GS have an increased risk of coronary atherosclerotic heart disease by 17%. Interestingly, this correlation was stronger in healthy groups without obesity, diabetes, and hypertension. Furthermore, a meta-analysis incorporating data from seven cohorts demonstrated that the random-effects model pooled risk ratio for individuals with GS was 1.23, irrespective of traditional risk factors, in comparison to individuals without GS35. Ratheesh et al. conducted a comprehensive national population-based cohort study to investigate the association between GS and diabetic individuals both before and following the initiation of insulin therapy. According to the study, insulin-treated diabetic patients experienced a 46% increase in GS incidence compared to those without insulin treatment36. The aforementioned findings underscore the multiple common pathological and physiological underpinnings between GS and cardiovascular diseases, and it has been challenging to separate these common risk factors from potential causally linked pathogenesis. However, a potential association between CMI and GS has not been reported in the literature, and it remains uncertain whether diabetes is involved in and regulates the link between CMI and GS pathogenesis.
CMI represents a novel metabolic indicator that is developed through the amalgamation of obesity parameters and lipid levels, which is simple and cost-effective. CMI has a high potential clinical value in the prediction of cardiovascular and metabolic-related diseases. Previous epidemiological studies with different target populations have reported an association between CMI and various systemic diseases. Tang et al.37 conducted a retrospective analysis of 2,243 patients with type 2 diabetes mellitus and found a significant positive correlation between CMI and atherosclerosis and concluded that CMI is an independent risk factor for atherosclerosis in patients with type 2 diabetes mellitus and is a key marker for predicting atherosclerosis risk. Zhou and colleagues surveyed 3,794 participants and found that elevated CMI levels increased the risk of depression38. Zha et al.39 conducted a retrospective cohort study involving over fifteen thousand Japanese adults to investigate the correlation between CMI and incident diabetes. Their findings indicated a positive association between elevated levels of CMI at baseline and the risk of developing diabetes. A growing number of emerging clinical epidemiological investigations have demonstrated the unique benefits of CMI in the assessment of cardiovascular disease and other metabolic disorders.
In contemporary medical practice, CMI is garnering heightened attention from healthcare professionals as a novel evaluative tool. Clinical practices stand to benefit from the integration of CMI monitoring alongside the co-management of GS risk factors. For instance, conducting comprehensive metabolic assessments—including evaluations of blood glucose, insulin levels, and lipid profiles—on patients exhibiting abnormal CMI values can facilitate the early detection of prediabetes or metabolic syndrome40. Moreover, by considering the patient’s lifestyle and familial medical history, clinicians can devise personalized management plans aimed at mitigating the risk of GS development. Specific guidelines should encompass regular CMI assessments, the identification of outliers, and the implementation of suitable interventions. Prioritizing the management of metabolic dysfunctions, particularly dyslipidemia and insulin resistance, is essential as both a preventive and therapeutic strategy. Dietary modifications are advised for patients with elevated CMI, such as increasing the consumption of foods rich in dietary fiber (e.g., whole grains, vegetables, and fruits) to lower cholesterol levels and diminish the risk of GS formation, while reducing the intake of high-sugar and high-fat foods to alleviate insulin resistance41. Secondly, increased physical activity with moderate-intensity aerobic exercise can improve insulin sensitivity, lower blood lipid levels, and improve gallbladder function. Finally, clinicians should also regularly follow up with patients with abnormal metabolic function. This will not only reduce the incidence of GS but also improve the overall cardiometabolic health of the at-risk population.
The present research is pioneering in examining the connection between CMI and GS with authoritative published data from a substantial sample size, analyzing diabetes as a potential mediator and link. The specific mechanism of how elevated CMI influences the pathogenesis and advancement of GS through diabetes mellitus is unknown. Visceral obesity, insulin resistance, and dysregulated levels of lipid metabolism may be responsible for assessing the vulnerability of diabetic patients to GS. Elevated free fatty acids due to disorders of lipid metabolism may stimulate fatty acid oxidation and peroxidation, resulting in the production of oxidative free radicals, which may exacerbate pancreatic islet cell dysfunction. Secondly, hyperesterolaemia in diabetic patients is usually accompanied by low HDL-C levels, which play an important role in reverse cholesterol transport, and this can lead to a reduced ability of the liver to remove cholesterol, which further exacerbates the accumulation of cholesterol in the bile42,43. Additionally, reduced levels of HDL-C may impair β-cell function, thereby impacting insulin secretion. In individuals with high WHtR, there is a reduction in the number and binding efficiency of insulin receptors on target tissues, resulting in impaired glucose processing44,45. Furthermore, adipocytes in hyperlipidemic states disrupt insulin signaling through various pathways, including activating pro-inflammatory cytokines, elevated levels of free fatty acids, and increased lipotoxicity. These alterations contribute to the onset of insulin resistance, which ultimately affects diabetes development46,47,48.
The association between diabetes mellitus and GS is biologically plausible, given that diabetes mellitus influences several critical processes involved in GS formation, including altered cystic motility, changes in bile composition, and insulin resistance. Autonomic neuropathy is frequently observed in diabetic patients, particularly those with chronic poor glycemic control. This neuropathy impairs the innervation of the gallbladder, leading to a reduced response to stimuli. The contraction of the gallbladder is primarily regulated by the vagus nerve, and diabetes-induced vagal dysfunction results in diminished gallbladder contractility and prolonged bile retention, thereby facilitating the formation of cholesterol crystals. Furthermore, a hyperglycemic state may directly impair the function of gallbladder smooth muscle cells. Chronic hyperglycemia induces increased oxidative stress and an enhanced inflammatory response, which subsequently compromise the structure and function of gallbladder smooth muscle. This damage may further exacerbate gallbladder peristalsis dysfunction. In diabetic patients, dysregulated glucose metabolism, often accompanied by insulin resistance, impairs gallbladder smooth muscle function due to microvascular damage, ultimately leading to impaired gallbladder emptying49,50,51. In addition, changes in bile composition may be one of the important mechanisms for GS formation in diabetic patients. In diabetic patients, the synthesis and secretion of bile acids may be reduced due to altered liver function. Inadequate bile acid secretion decreases cholesterol solubility and increases the risk of cholesterol crystal formation52,53. Insulin resistance is often accompanied by a chronic low-grade inflammatory state, which may promote GS formation through several mechanisms. Chronic inflammation leads to structural and functional changes in the gallbladder wall, such as thickening and reduced contractile function. In addition, inflammatory factors may affect the composition of bile, indirectly promoting GS formation.
The findings of this study offer substantial evidence for a correlation between the cardiovascular marker CMI and GS, with diabetes mellitus serving as a partial mediator in the relationship. As WHtR and lipid parameters can be obtained through routine physical examinations and blood tests, the assessment of CMI in clinical practice is feasible. These results imply that healthcare providers should not neglect blood glucose levels while regulating lipids in patients with high CMI levels, and that co-management of the two may be a proven way to reduce GS risk.
Strengths and limitations
Several noteworthy strengths of this study should be highlighted. Primarily, it stands as the original study to comprehensively assess the correlation between CMI levels and the prevalence of GS within a U.S. demographic, uncovering a potential mediating influence of diabetes and enriching the body of existing literature. Secondly, the large sample size sampling methodology and sensitivity analyses based on U.S. adults add to the persuasiveness and generalisability of the findings. Finally, this study enhances the understanding of CMI as a viable predictor of individuals susceptible to GS, highlighting the importance of cardiovascular markers and lipid metabolism disorders in the pathogenesis of GS. However, there are some limitations to our study. Due to the homogeneous nature of the NHANES database survey method, the diagnosis of GS in this study was obtained based on subjects’ questionnaire interviews and self-reports, which may introduce recall bias, thus affecting the accuracy of the diagnosis. Additionally, due to its observational cross-sectional design, this study is limited in its ability to establish causal relationships, highlighting the necessity for future prospective cohort studies to confirm its findings. Finally, despite adjusting for many covariates based on previous studies and clinical experience, residual or omitted confounders may affect the associations observed in the results.
Conclusion
In summary, this large-scale original investigation suggests that elevated CMI is strongly associated with an increased risk of GS and that diabetes plays a key mediating role in the pathogenesis of both. This indicates that healthcare professionals should not neglect blood glucose levels while addressing dyslipidemia and that co-management of the two may be a proven approach to reducing GS risk.
Data availability
The data utilized in this study were sourced from NHANES, with the original dataset accessible through the corresponding author upon inquiry.
Abbreviations
- GS:
-
Gallstones
- WHtR:
-
Waist-to-height ratio
- BMI:
-
Body mass index
- WC:
-
Waist circumference
- TG:
-
Triglycerides
- HDL-C:
-
High-density lipoprotein cholesterol
- CMI:
-
Cardiovascular metabolic index
- LAP:
-
Lipid accumulation product
- TyG:
-
Triglyceride-glucos
- NHANES:
-
National Health and Nutrition Examination Survey
- NCHS:
-
National Center for Health Statistics
- PIR:
-
Ratio of household income to poverty
- RCS:
-
Restricted cubic spline
- CI:
-
Confidence interval
- OR:
-
Odds ratio
References
Lammert, F. et al. Gallstones. Nat. Rev. Dis. Primers 2, 16024 (2016).
de Sousa, S. et al. Management of suspected common bile duct stones on cholangiogram during same-stay cholecystectomy for acute gallstone-related disease. BMC Surg. 17(1), 39 (2017).
Stinton, L. M. & Shaffer, E. A. Epidemiology of gallbladder disease: Cholelithiasis and cancer. Gut Liver 6(2), 172–187 (2012).
Everhart, J. E. & Ruhl, C. E. Burden of digestive diseases in the united States part III: Liver, biliary tract, and pancreas. Gastroenterology 136(4), 1134–1144 (2009).
Festi, D. et al. Natural history of gallstone disease: Expectant management or active treatment? Results from a population-based cohort study. J. Gastroenterol. Hepatol. 25(4), 719–724 (2010).
Shaffer, E. A. Epidemiology and risk factors for gallstone disease: Has the paradigm changed in the 21st century? Curr. Gastroenterol. Rep. 7(2), 132–140 (2005).
Granel-Villach, L. et al. Factors that influence bile fluid microbiology in cholecystectomized patients. Rev. Gastroenterol. Mex (Engl Ed) 85(3), 257–263 (2020).
Rai, R., Tewari, M., Kumar, M., Singh, T. B. & Shukla, H. S. Expression profile of cholecystokinin type-A receptor in gallbladder cancer and gallstone disease. Hepatobiliary Pancreat. Dis. Int. 10(4), 408–414 (2011).
Shabanzadeh, D. M. et al. Are incident gallstones associated to sex-dependent changes with age? A cohort study. Andrology 5(5), 931–938 (2017).
Jain, K. & Garg, P. K. Could type and size of gallstones influence gallbladder carcinogenesis?? Ann. Surg. 263(3), e57 (2016).
Wirth, J. et al. Presence of gallstones and the risk of cardiovascular diseases: The EPIC-Germany cohort study. Eur. J. Prev. Cardiol. 22(3), 326–334 (2015).
Chen, H. et al. Dissolved oxygen from microalgae-gel patch promotes chronic wound healing in diabetes. Sci. Adv. 6(20), eaba4311 (2020).
Li, X. et al. Targeting long noncoding RNA-AQP4-AS1 for the treatment of retinal neurovascular dysfunction in diabetes mellitus. EBioMedicine 77, 103857 (2022).
Araneta, M. R. et al. Optimum BMI cut points to screen Asian Americans for type 2 diabetes. Diabetes Care. 38(5), 814–820 (2015).
Wiwanitkit, V. BMI, waist circumference and waist-to-hip ratio: Diabetes mellitus type 2. Eur. J. Clin. Nutr. 64(11), 1377 (2010).
Alzeidan, R., Fayed, A., Rabiee, F., Hersi, A. & Elmorshedy, H. Diagnostic performance of waist-to-height ratio in identifying cardiovascular risk factors and metabolic syndrome among adult Saudis. A cross-sectional study. Saudi Med. J. 41(3), 253–260 (2020).
Corrêa, M. M., Thumé, E., De Oliveira, E. R. & Tomasi, E. Performance of the waist-to-height ratio in identifying obesity and predicting non-communicable diseases in the elderly population: A systematic literature review. Arch. Gerontol. Geriatr. 65, 174–182 (2016).
Hou, X. et al. Stronger associations of waist circumference and waist-to-height ratio with diabetes than BMI in Chinese adults. Diabetes Res. Clin. Pract. 147, 9–18 (2019).
Zhang, S. et al. Is waist-to-height ratio the best predictive indicator of cardiovascular disease incidence in hypertensive adults? A cohort study. BMC Cardiovasc. Disord. 22(1), 214 (2022).
Ye, R. et al. Association of cardiometabolic and triglyceride-glucose index with left ventricular diastolic function in asymptomatic individuals. Nutr. Metab. Cardiovasc. Dis. 34(7), 1590–1600 (2024).
Tohidi, M. et al. Association of triglycerides to high-density lipoprotein cholesterol ratio to identify future prediabetes and type 2 diabetes mellitus: Over one-decade follow-up in the Iranian population. Diabetol. Metab. Syndr. 15(1), 13 (2023).
Acosta-García, E. & Concepción-Páez, M. [Cardiometabolic index as a predictor of cardiovascular risk factors in adolescents]. Rev. Salud Publica (Bogota) 20(3), 340–345 (2018).
Shi, W. R. et al. Estimate of prevalent diabetes from cardiometabolic index in general Chinese population: a community-based study. Lipids Health Dis. 17(1), 236 (2018).
Wang, H. et al. Validity of cardiometabolic index, lipid accumulation product, and body adiposity index in predicting the risk of hypertension in Chinese population. Postgrad. Med. 130(3), 325–333 (2018).
Zou, J. et al. Association between the cardiometabolic index and non-alcoholic fatty liver disease: Insights from a general population. BMC Gastroenterol. 22(1), 20 (2022).
Zuo, Y. Q., Gao, Z. H., Yin, Y. L., Yang, X. & Feng, P. Y. Association between the cardiometabolic index and hyperuricemia in an asymptomatic population with normal body mass index. Int. J. Gen. Med. 14, 8603–8610 (2021).
Cheng, J. et al. Association of pro-inflammatory diet with increased risk of gallstone disease: A cross-sectional study of NHANES January 2017-March 2020. Front. Nutr. 11, 1344699 (2024).
Wen, S. H., Tang, X., Tang, T. & Ye, Z. R. Association between weight-adjusted-waist index and gallstones: An analysis of the National health and nutrition examination survey. BMC Gastroenterol. 24(1), 40 (2024).
Global guideline for. Type 2 diabetes. Diabetes Res. Clin. Pract. 104(1), 1–52 (2014).
Nayak, S. S. et al. Diagnostic and prognostic value of triglyceride glucose index: A comprehensive evaluation of meta-analysis. Cardiovasc. Diabetol. 23(1), 310 (2024).
Wakabayashi, I. & Daimon, T. The cardiometabolic index as a new marker determined by adiposity and blood lipids for discrimination of diabetes mellitus. Clin. Chim. Acta. 438, 274–278 (2015).
Gong, R. et al. Associations between TG/HDL ratio and insulin resistance in the US population: A cross-sectional study. Endocr. Connect. 10(11), 1502–1512 (2021).
Liu, M. et al. Predicted fat mass and lean mass in relation to all-cause and cause-specific mortality. J. Cachexia Sarcopenia Muscle 13(2), 1064–1075 (2022).
Liu, X. et al. Cardiometabolic index: A new tool for screening the metabolically obese normal weight phenotype. J. Endocrinol. Invest. 44(6), 1253–1261 (2021).
Zheng, Y. et al. Gallstones and risk of coronary heart disease: Prospective analysis of 270 000 men and women from 3 US cohorts and Meta-Analysis. Arterioscler. Thromb. Vasc Biol. 36(9), 1997–2003 (2016).
Ratheesh, R., Ulrich, M. T., Ghozy, S., Al-Jaboori, M. & Nayak, S. S. The association between diabetes and gallstones: A nationwide population-based cohort study. Prz Gastroenterol. 18(3), 292–299 (2023).
Tang, C. et al. Correlation between the cardiometabolic index and arteriosclerosis in patients with type 2 diabetes mellitus. BMC Cardiovasc. Disord 24(1), 186 (2024).
Zhou, X. et al. Association between cardiometabolic index and depression: National health and nutrition examination survey (NHANES) 2011–2014. J. Affect. Disord 351, 939–947 (2024).
Zha, F. et al. The nonlinear correlation between the cardiometabolic index and the risk of diabetes: A retrospective Japanese cohort study. Front. Endocrinol. (Lausanne) 14, 1120277 (2023).
Chen, X., Zhao, Y., Sun, J., Jiang, Y. & Tang, Y. Identification of metabolic syndrome using lipid accumulation product and cardiometabolic index based on NHANES data from 2005 to 2018. Nutr. Metab. (Lond) 21 (1), 96 (2024).
Carmichael, L. et al. Is vascular insulin resistance an early step in diet-induced whole-body insulin resistance? Nutr. Diabetes 12(1), 31 (2022).
Boden, G. Obesity, insulin resistance, type 2 diabetes and free fatty acids. Expert Rev. Endocrinol. Metab. 1(4), 499–505 (2006).
Kahn, S. E., Hull, R. L. & Utzschneider, K. M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444(7121), 840–846 (2006).
Barazzoni, R., Gortan Cappellari, G., Ragni, M. & Nisoli, E. Insulin resistance in obesity: An overview of fundamental alterations. Eat. Weight Disord 23(2), 149–157 (2018).
Ruze, R. et al. Obesity and type 2 diabetes mellitus: Connections in epidemiology, pathogenesis, and treatments. Front. Endocrinol. (Lausanne) 14, 1161521 (2023).
Poitout, V. & Robertson, R. P. Glucolipotoxicity: Fuel excess and beta-cell dysfunction. Endocr. Rev. 29(3), 351–366 (2008).
Tang, X. et al. Associations of insulin resistance and beta-cell function with abnormal lipid profile in newly diagnosed diabetes. Chin. Med. J. (Engl) 135(21), 2554–2562 (2022).
Taskinen, M. R. & Borén, J. New insights into the pathophysiology of dyslipidemia in type 2 diabetes. Atherosclerosis 239(2), 483–495 (2015).
Arshad, F., Laway, B. A., Rather, T. A., Kuchay, M. S. & Khan, S. H. Impaired gallbladder motility in adults with newly detected type 2 diabetes and lack of reversibility after achieving euglycemia. Can. J. Diabetes. 39(2), 101–104 (2015).
Škrha, J. Jr. Diabetes, lipids, and CV risk. Curr. Atheroscler Rep. 23(3), 8 (2021).
Vergès, B. Pathophysiology of diabetic dyslipidaemia: Where are we? Diabetologia 58(5), 886–899 (2015).
Pendleton, H., Ekman, R., Olsson, R., Ekberg, O. & Ohlsson, B. Motilin concentrations in relation to gastro intestinal dysmotility in diabetes mellitus. Eur. J. Intern. Med. 20(6), 654–659 (2009).
Rehfeld, J. F. et al. Cholecystokinin secretion is suppressed by glucagon-like peptide-1: Clue to the mechanism of the adverse gallbladder events of GLP-1-derived drugs. Scand. J. Gastroenterol. 53(12), 1429–1432 (2018).
Acknowledgements
The authors express their gratitude to the participants from the Neurosurgery Department of Yixing People’s Hospital, as well as those from the Gastrointestinal Surgery Department and Hepatobiliary Surgery Department of Wuxi People’s Hospital, for their valuable contributions to this study.
Funding
This study was supported by grants from Nanjing medical university wuxi medical centre normal project (WXKY202304050), Wuxi Double Hundred Top Talents (BJ20232012), National Natural Science Foundation of China (GA2024ZD02) and Wuxi People’s Hospital Reserve Academic Leader of the ‘Wild Goose Formation’ Talent Program (2024-YZ-HBDTR-WDW-2024).
Author information
Authors and Affiliations
Contributions
Y.Z., X.Q.G., H.L.: conception and design, information provision, software, manuscript writing. H.L., Y.L.W., D.W.W.: interpretation, drawing, review, data analysis. D.W.W., J.W.: review, proofreading, financial support. All authors reviewed and agreed to the final manuscript for publication.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The survey protocols were approved by the Ethics Review Board of the NCHS, and all participants provided written informed consent prior to their involvement in the study.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Zhang, Y., Gu, X., Lv, H. et al. Diabetes is the missing link between cardiometabolic index and gallstones: a large cross-sectional study. Sci Rep 15, 9947 (2025). https://doi.org/10.1038/s41598-025-93908-3
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s41598-025-93908-3