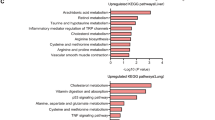
Abstract
Increased reactive oxygen species (ROS) levels are a hallmark of inflammatory bowel disease (IBD) and constitute a major mechanism of epithelial cell death. Approaches to broadly inhibit ROS have had limited efficacy in treating IBD. Here we show that lipid peroxidation contributes to the pathophysiology of IBD by promoting ferroptosis, an iron-dependent form of programmed cell death. Mechanistically, we provide evidence of heterocellular crosstalk between intestinal fibroblasts and epithelial cells. In IBD tissues and mouse models of chronic colitis, acyl-CoA synthetase long-chain family 4 (ACSL4) is overexpressed in fibroblasts. ACSL4 in fibroblasts reprograms lipid metabolism and mediates intestinal epithelial cell sensitivity to ferroptosis. In mouse models, overexpressing ACSL4 in fibroblasts results in increased intestinal epithelial ferroptosis and worsened colitis, while pharmacological inhibition or deletion of fibroblast ACSL4 ameliorates colitis. Our work provides a targeted approach to therapeutic antioxidant treatments for IBD.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The publicly available bulk RNA-seq data of IBD and non-IBD tissues (GSE57945 (ref. 18), GSE109142 (ref. 20), GSE117993 (ref. 47) and GSE199906 (ref. 21)) and the publicly available scRNA-seq data of IBD and non-IBD tissues (GSE114374 (ref. 25), GSE214695 (ref. 26), E-MTAB-8901 (ref. 27) and SCP1884 (ref. 28)) were reanalyzed. We provided the lipidomics dataset as Supplementary Information and most of the raw data as Source data. Any additional data and materials not publicly available can be provided upon reasonable request by contacting the corresponding author. Source data are provided with this paper.
Code availability
We used the Seurat (v.3.2.2) R package for the published dataset collection and reanalysis. The custom computer code and algorithms used in this study are available on Zenodo (https://doi.org/10.5281/zenodo.15191281)52.
References
Tavakoli, P. et al. A review of inflammatory bowel disease: a model of microbial, immune and neuropsychological integration. Public Health Rev. 42, 1603990 (2021).
Huang, W., Aabed, N. & Shah, Y. M. Reactive oxygen species and ferroptosis at the nexus of inflammation and colon cancer. Antioxid. Redox Signal. 39, 551–568 (2023).
Chen, L. et al. The antioxidant procyanidin reduces reactive oxygen species signaling in macrophages and ameliorates experimental colitis in mice. Front. Immunol. 8, 1910 (2017).
Ananthakrishnan, A. N. et al. Statin use is associated with reduced risk of colorectal cancer in patients with inflammatory bowel diseases. Clin. Gastroenterol. Hepatol. 14, 973–979 (2016).
Moura, F. A. et al. Antioxidant therapy for treatment of inflammatory bowel disease: does it work? Redox Biol. 6, 617–639 (2015).
Juan, C. A. et al. The chemistry of reactive oxygen species (ROS) revisited: outlining their role in biological macromolecules (DNA, lipids and proteins) and induced pathologies. Int. J. Mol. Sci. 22, 4642 (2021).
Jiang, X., Stockwell, B. R. & Conrad, M. Ferroptosis: mechanisms, biology and role in disease. Nat. Rev. Mol. Cell Biol. 22, 266–282 (2021).
Kobayashi, Y. et al. Association between dietary iron and zinc intake and development of ulcerative colitis: a case-control study in Japan. J. Gastroenterol. Hepatol. 34, 1703–1710 (2019).
Millar, A. D., Rampton, D. S. & Blake, D. R. Effects of iron and iron chelation in vitro on mucosal oxidant activity in ulcerative colitis. Aliment Pharm. Ther. 14, 1163–1168 (2000).
Mayr, L. et al. Dietary lipids fuel GPX4-restricted enteritis resembling Crohn’s disease. Nat. Commun. 11, 1775 (2020).
Werner, T. et al. Depletion of luminal iron alters the gut microbiota and prevents Crohn’s disease-like ileitis. Gut 60, 325–333 (2011).
Xu, M. et al. Ferroptosis involves in intestinal epithelial cell death in ulcerative colitis. Cell Death Dis. 11, 86 (2020).
Wang, S. et al. Curculigoside inhibits ferroptosis in ulcerative colitis through the induction of GPX4. Life Sci. 259, 118356 (2020).
Doll, S. et al. ACSL4 dictates ferroptosis sensitivity by shaping cellular lipid composition. Nat. Chem. Biol. 13, 91–98 (2017).
Magtanong, L. et al. Context-dependent regulation of ferroptosis sensitivity. Cell Chem. Biol. 29, 1409–1418.e6 (2022).
Wei, Z. et al. Human umbilical cord mesenchymal stem cells derived exosome shuttling mir-129-5p attenuates inflammatory bowel disease by inhibiting ferroptosis. J. Nanobiotechnology 21, 188 (2023).
Lam, I. H. et al. ACSL4 mediates inflammatory bowel disease and contributes to LPS-induced intestinal epithelial cell dysfunction by activating ferroptosis and inflammation. Open Med. 19, 20240993 (2024).
Haberman, Y. et al. Pediatric Crohn disease patients exhibit specific ileal transcriptome and microbiome signature. J. Clin. Invest. 124, 3617–3633 (2014).
Keller, J. N. et al. 4-Hydroxynonenal, an aldehydic product of membrane lipid peroxidation, impairs glutamate transport and mitochondrial function in synaptosomes. Neuroscience 80, 685–696 (1997).
Haberman, Y. et al. Ulcerative colitis mucosal transcriptomes reveal mitochondriopathy and personalized mechanisms underlying disease severity and treatment response. Nat. Commun. 10, 38 (2019).
Sosnovski, K. E. et al. GATA6-AS1 regulates intestinal epithelial mitochondrial functions, and its reduced expression is linked to intestinal inflammation and less favourable disease course in ulcerative colitis. J. Crohns Colitis 17, 960–971 (2023).
Kagan, V. E. et al. Oxidized arachidonic and adrenic PEs navigate cells to ferroptosis. Nat. Chem. Biol. 13, 81–90 (2017).
Castillo, A. F. et al. New inhibitor targeting Acyl-CoA synthetase 4 reduces breast and prostate tumor growth, therapeutic resistance and steroidogenesis. Cell. Mol. Life Sci. 78, 2893–2910 (2021).
Trzpis, M. et al. Epithelial cell adhesion molecule: more than a carcinoma marker and adhesion molecule. Am. J. Pathol. 171, 386–395 (2007).
Kinchen, J. et al. Structural remodeling of the human colonic mesenchyme in inflammatory bowel disease. Cell 175, 372–386.e17 (2018).
Garrido-Trigo, A. et al. Macrophage and neutrophil heterogeneity at single-cell spatial resolution in human inflammatory bowel disease. Nat. Commun. 14, 4506 (2023).
Elmentaite, R. et al. Cells of the human intestinal tract mapped across space and time. Nature 597, 250–255 (2021).
Kong, L. et al. The landscape of immune dysregulation in Crohn’s disease revealed through single-cell transcriptomic profiling in the ileum and colon. Immunity 56, 444–458.e5 (2023).
Farahani, R. M. & Xaymardan, M. Platelet-derived growth factor receptor alpha as a marker of mesenchymal stem cells in development and stem cell biology. Stem Cells Int. 2015, 362753 (2015).
Cockcroft, S. Mammalian lipids: structure, synthesis and function. Essays Biochem. 65, 813–845 (2021).
Killion, E. A. et al. A role for long-chain acyl-CoA synthetase-4 (ACSL4) in diet-induced phospholipid remodeling and obesity-associated adipocyte dysfunction. Mol. Metab. 9, 43–56 (2018).
Monternier, P. A. et al. Therapeutic potential of deuterium-stabilized (R)-pioglitazone-PXL065-for X-linked adrenoleukodystrophy. J. Inherit. Metab. Dis. 45, 832–847 (2022).
Bersuker, K. et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature 575, 688–692 (2019).
Chen, D. et al. iPLA2beta-mediated lipid detoxification controls p53-driven ferroptosis independent of GPX4. Nat. Commun. 12, 3644 (2021).
Jiao, L. et al. Deficiency of group VIA phospholipase A2 (iPLA2beta) renders susceptibility for chemical-induced colitis. Dig. Dis. Sci. 60, 3590–3602 (2015).
Dorninger, F. et al. Plasmalogens, platelet-activating factor and beyond—ether lipids in signaling and neurodegeneration. Neurobiol. Dis. 145, 105061 (2020).
Orlando, U. et al. Characterization of the mouse promoter region of the acyl-CoA synthetase 4 gene: role of Sp1 and CREB. Mol. Cell. Endocrinol. 369, 15–26 (2013).
Chiba, M., Nakane, K. & Komatsu, M. Westernized diet is the most ubiquitous environmental factor in inflammatory bowel disease. Perm. J. 23, 18–107 (2019).
Minaiyan, M., Mostaghel, E. & Mahzouni, P. Preventive therapy of experimental colitis with selected iron chelators and anti-oxidants. Int. J. Prev. Med. 3, S162–S169 (2012).
Scaioli, E., Liverani, E. & Belluzzi, A. The imbalance between n-6/n-3 polyunsaturated fatty acids and inflammatory bowel disease: a comprehensive review and future therapeutic perspectives. Int. J. Mol. Sci. 18, 2619 (2017).
de Silva, P. S. et al. Dietary arachidonic and oleic acid intake in ulcerative colitis etiology: a prospective cohort study using 7-day food diaries. Eur. J. Gastroenterol. Hepatol. 26, 11–18 (2014).
Ajabnoor, S. M. et al. Long-term effects of increasing omega-3, omega-6 and total polyunsaturated fats on inflammatory bowel disease and markers of inflammation: a systematic review and meta-analysis of randomized controlled trials. Eur. J. Nutr. 60, 2293–2316 (2021).
Singhal, R. et al. HIF-2α activation potentiates oxidative cell death in colorectal cancers by increasing cellular iron. J. Clin. Invest. 131, e143691 (2021).
Hall, B. et al. Genome editing in mice using CRISPR/Cas9 technology. Curr. Protoc. Cell Biol. 81, e57 (2018).
Ostanin, D. V. et al. T cell transfer model of chronic colitis: concepts, considerations, and tricks of the trade. Am. J. Physiol. Gastrointest. Liver Physiol. 296, G135–G146 (2009).
Schwarzler, J. et al. PUFA-induced metabolic enteritis as a fuel for Crohn’s disease. Gastroenterology 162, 1690–1704 (2022).
Pelia, R. et al. Profiling non-coding RNA levels with clinical classifiers in pediatric Crohn’s disease. BMC Med. Genomics 14, 194 (2021).
Solanki, S. et al. Dysregulated amino acid sensing drives colorectal cancer growth and metabolic reprogramming leading to chemoresistance. Gastroenterology 164, 376–391.e13 (2023).
Bell, H. N., Das, N. K. & Shah, Y. M. Protocol for isolation and analysis of small volatile microbiome metabolites from human or mouse samples. STAR Protoc. 3, 101311 (2022).
Patt, A. et al. MDM2-dependent rewiring of metabolomic and lipidomic profiles in dedifferentiated liposarcoma models. Cancers 12, 2157 (2020).
Huang, W. et al. Dietary iron is necessary to support proliferative regeneration after intestinal injury. J. Nutr. 154, 1153–1164 (2024).
Oh, T. G. ACSL4 gene expression in fibroblast among IBD single cell sequencing datasets (Fig. 4a–b, Extended Data Fig. 5). Zenodo https://doi.org/10.5281/zenodo.15191281 (2025).
Acknowledgements
Schematic diagrams (Figs. 1d, 5a and 7a,m and Extended Data Fig. 1) were created with BioRender.com. This work was funded by NIH grants R01CA148828, R01CA245546 and R01DK095201 (Y.M.S.) and UMCCC Core Grant P30CA046592 (Y.M.S.). W.H. was supported by the NIH grant F30DK131851. Y.Z. was supported by the Rackham International Students Fellowship/Chia-Lun Lo Fellowship. J.S. was supported in part by NIH grant R37CA262209. J.A.C. was supported by NIH grants R01ES028802, P30 ES017885 and NCI R21 CA273646. J.R.S. was supported by NIDDK R01 DK137806 and NIDDK RC2 DK140862. S.P.H. was supported by the Crohn’s Colitis Foundation Senior Research Award. A.D.P. was supported by NIH grant S10OD021750, NIH/National Institute of Environmental Sciences grant R35 ES035027 and the US Department of Agriculture National Institute of Food and Federal Appropriations under project PEN047702 and accession number 7006412. A.S.G. was supported by USDA grants USDA 58-8050-9-004 and USDA NIFA 2022-67018-37186 and by NIH grant P30DK046200. A.N. was supported by grant DK59888.
Author information
Authors and Affiliations
Contributions
W.H., Y.Z., N.K.D. and Y.M.S. conceived of and designed the study. W.H., Y.Z., N.K.D., S.S., J.S., S.P.H., M.K.D., J.R.S., A.D.P. and J.A.C. developed the methodologies. W.H., Y.Z., N.K.D., S.S., C.J., M.O.E.-D., H.N.B., N.A., R.S., C.C., K.B., Y.Y., J.D., I.K. and A.S.G. acquired the data. W.H., Y.Z., N.K.D., M.O.E.-D., J.D., A.S., P.D.R.H., S.P.H., J.A.C., T.G.O., A.D.P., A.S.G., J.K.G. and A.N. analyzed and interpreted the data. W.H., Y.Z. and Y.M.S. supervised the study and wrote the paper; all authors edited and provided inputs to the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks Dirk Haller, Eun-Woo Lee and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Yanina-Yasmin Pesch, in collaboration with the Nature Metabolism team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Schematics for different colitis mouse models.
For acute DSS-induced colitis, mice were placed on 3% DSS in the drinking water for 7 days. For chronic DSS-induced colitis, the mice were placed in 1% DSS water for 21 days, and the DSS concentration was increased by 0.5% DSS every 7 days until the mice lost 30% of their starting body weight (1% DSS for 21 days, 1.5% DSS for 7 days, 2% DSS for 7 days, etc.). For the T-cell colitis model, CD4+CD45RBhigh (experimental) and CD4+CD45RBlow (control) T cells from male and female C57BL/6 mice were isolated from the spleen using a MojoSort mouse CD4 T-cell isolation kit and sorted via flow cytometry using a 1:100 dilution of an anti-mouse CD4 PE-Cy7 antibody and a 1:100 dilution of an anti-mouse CD45RB APC antibody. IP was injected into male and female immunodeficient Rag1 KO mice. Notably, male or female donor mice can be generated from male Rag1 KO mice, while only female donor mice can be generated from female Rag1 KO mice. Mice were monitored weekly for weight loss and signs of diarrhea. Mice were sacrificed at the end of 8 weeks. Created in BioRender. Shah, Y. (2025) https://BioRender.com/l21i803.
Extended Data Fig. 2 Intestinal Gpx4 or Slc7a11 deletion exacerbates chronic, but not acute, colitis.
a, Representative H&E images and 4HNE images, b, inflammatory histology score, c, percentage positive (%) of 4HNE staining, and d, colon lengths from Rag1 KO; Gpx4f/f mice (n = 8) and Rag1 KO; VilERT2; Gpx4f/f mice (n = 5) under T cell transfer induced chronic colitis model. e, Relative gene expression of Slc7a11 in small intestine tissue and colon tissue from Slc7a11f/f mice (n = 3) and VilERT2; Slc7a11f/f mice (n = 3). f, representative H&E images of the small intestine and colon tissue of VilERT2; Slc7a11f/f mice and Slc7a11f/f mice. g, Representative H&E images, h, inflammatory histology scores, and i, colon lengths of VilERT2; Slc7a11f/f mice (n = 7) and Slc7a11f/f mice (n = 6) with acute DSS-induced colitis. j, Representative H&E images, k, inflammatory histology scores, and l, colon lengths of VilERT2; Slc7a11f/f mice (n = 11) and Slc7a11f/f mice (n = 7) with chronic DSS-induced colitis. Data are shown as mean ± SEM, statistical analysis was performed using two-tailed unpaired T-test (for two column comparison) or 2-way ANOVA (for groups comparison). All experiments were repeated at least twice independently, with similar results.
Extended Data Fig. 3 Intestinal FPN deletion has no effects on chronic colitis.
a, Relative gene expression and b, Protein expression of Ferroportin (Fpn) in colon tissue from Fpnf/f mice (n = 5) and VilERT2; Fpnf/f mice (n = 5). c, Representative H&E images, d, inflammatory histology scores, and e, colon lengths of VilERT2; Fpnf/f mice (n = 12) and Fpnf/f mice (n = 8) with chronic DSS-induced colitis. Data are shown as mean ± SEM, statistical analysis was performed using two-tailed unpaired T-test. All experiments were repeated at least twice independently, with similar results.
Extended Data Fig. 4 ACSL4 is expressed in epithelial cells.
a, IF staining for ACSL4 (red), EpCAM (cyan), and Hoechst (blue) in IBD and normal tissue. b, IF staining for ACSL4 (red), EpCAM (cyan), and Hoechst (blue) in basal, acute DSS, and chronic DSS conditions. The experiments were repeated three times independently, with similar results. These samples are the same ones shown in Fig. 4 with more details and with a different antibody displayed.
Extended Data Fig. 5 ACSL4 expression is increased in fibroblasts in IBD tissue.
a, UMAP and violin plots for ACSL4 expression in GSE114374, b, GSE214695, c, SCP1884, and d, E-MTAB-8901.
Extended Data Fig. 6 Overexpression ACSL4 in fibroblasts worsens chronic DSS induced colitis.
a, Relative gene expression of ferroptosis-related genes (Chac1, Ptgs2, Fsp1 and Fsp27). b, Representative H&E colon tissue, c, inflammatory histology scores, d, colon length, and lipid ROS levels in e, epithelium and f, fibroblasts, as well as cell death (%) in g, epithelium and h, fibroblasts from LSL-Acsl4 mice and PdgfrαERT2; LSL-Acsl4 mice treated with or without liproxstatin-1 (10 mg/Kg, i.p., daily) (LSL-Acsl4 + vehicle (n = 7), LSL-Acsl4 + liproxstatin-1 (n = 7), PdgfrαERT2; LSL-Acsl4 + vehicle (n = 7) and PdgfrαERT2; LSL-Acsl4 + liproxstatin-1 (n = 7)). Data are shown as mean ± SEM, statistical analysis was performed using 2-way ANOVA. All experiments were repeated at least twice independently, with similar results.
Extended Data Fig. 7 ACSL4 in fibroblast sensitizes epithelial ferroptosis through secreted PUFA.
a, Cell death (%) of NCM-460 cell treated with CM from ACSL4-Con and ACSL4-OE fibroblast (n = 3 for each group). b, Cell death (%) of NCM-460 cell treated with Control (0.5% DMSO), aqueous phase and lipid phase metabolite extraction from ACSL4-OE fibroblast CM, with and without liproxstatin-1 (1 μM) (n = 3 for each group). c, Cell death (%) of NCM-460 cell treated with RSL3 (0.5 μM), Control (BSA 5 μM), AA (20 μM), EPA (20 μM) and DHA (20 μM) with and without liproxstatin-1 (1 μM) (n = 3 for each group). Data are shown as mean ± SEM, statistical analysis was performed using two-tailed unpaired T-test (for two column comparison) or 2-way ANOVA (for groups comparison). All experiments were repeated at least twice independently, with similar results.
Extended Data Fig. 8 ACSL4 inhibitors protected DSS induced chronic colitis.
a, Representative H&E colon tissue, b, inflammatory histology scores, c, colon length, d, lipid ROS levels in epithelium and fibroblast, and e, cell death (%) in epithelium and fibroblast from WT mice treated with Control diet (n = 8) and Pioglitazone diet (n = 8). Data are shown as mean ± SEM, statistical analysis was performed using two-tailed unpaired T-test (for two column comparison) or 2-way ANOVA (for groups comparison). All experiments were repeated at least twice independently, with similar results.
Extended Data Fig. 9 No sex differences were observed in the key experiment.
a, histology scores, b, colon length and relative lipid ROS levels in c, epithelial cells and d, fibroblasts of LSL-Acsl4 and PdgfrαERT2, LSL-Acsl4 mice treated with acute DSS colitis ± liproxstatin-1 (10 mg/Kg, i.p. injection, daily) (n = 5 (male, LSL-Acsl4, vehicle), n = 6 (female, LSL-Acsl4, vehicle); n = 5 (male, PdgfrαERT2; LSL-Acsl4, vehicle), n = 7 (female, PdgfrαERT2; LSL-Acsl4, vehicle); n = 5 (male, LSL-Acsl4, liproxstatin-1), n = 4 (female, LSL-Acsl4, liproxstatin-1); n = 5 (male, PdgfrαERT2; LSL-Acsl4, liproxstatin-1), n = 6 (female, PdgfrαERT2; LSL-Acsl4, liproxstatin-1).). Data are shown as mean ± SEM, statistical analysis was performed using two-tailed unpaired T-test (for two column comparison) or 2-way ANOVA (for groups comparison). All experiments were repeated at least twice independently, with similar results.
Supplementary information
Supplementary Information (download PDF )
Supplementary Fig. 1.
Supplementary Table 1 (download XLSX )
Supplementary Tables 1–6.
Supplementary Data 1 (download XLSX )
Lipidomics dataset.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data in an Excel file.
Source Data Fig. 2 (download XLSX )
Statistical source data in an Excel file.
Source Data Fig. 3 (download XLSX )
Statistical source data in an Excel file.
Source Data Fig. 4 (download XLSX )
Statistical source data in an Excel file.
Source Data Fig. 5 (download XLSX )
Statistical source data in an Excel file.
Source Data Fig. 6 (download XLSX )
Statistical source data in an Excel file.
Source Data Fig. 7 (download XLSX )
Statistical source data in an Excel file.
Source Data Extended Data Fig./Table 2 (download XLSX )
Statistical source data in an Excel file.
Source Data Extended Data Fig./Table 3 (download XLSX )
Statistical source data in an Excel file.
Source Data Extended Data Fig./Table 6 (download XLSX )
Statistical source data in an Excel file.
Source Data Extended Data Fig./Table 7 (download XLSX )
Statistical source data in an Excel file.
Source Data Extended Data Fig./Table 8 (download XLSX )
Statistical source data in an Excel file.
Source Data Extended Data Fig./Table 9 (download XLSX )
Statistical source data in an Excel file.
Source Data Figures and Extended Data Figures (download ZIP )
Supporting blot files.
Source Data Figures and Extended Data Figures (download ZIP )
Supporting source data image files.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Huang, W., Zhang, Y., Das, N.K. et al. Fibroblast lipid metabolism through ACSL4 regulates epithelial sensitivity to ferroptosis in IBD. Nat Metab 7, 1358–1374 (2025). https://doi.org/10.1038/s42255-025-01313-x
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s42255-025-01313-x
This article is cited by
-
Inflammatory bowel diseases: pathological mechanisms and therapeutic perspectives
Molecular Biomedicine (2026)
-
Cancer-associated fibroblasts at the crossroads of tumor progression and therapy resistance: from heterogeneity to precision reprogramming
Journal of the Egyptian National Cancer Institute (2025)
-
When gut fibroblasts feed epithelial cells to death
Nature Metabolism (2025)
-
Selenium Induces Ferroptosis in Colorectal Cancer Cells via Direct Interaction with Nrf2 and Gpx4
Biological Trace Element Research (2025)