Abstract
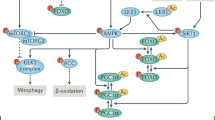
The nuclear envelope (NE) is essential for cellular homeostasis, yet its integrity declines with age, accelerating functional deterioration. Here we report a mitochondria-to-NE signalling pathway that safeguards NE integrity through redox-dependent lipid metabolism. In Caenorhabditis elegans, reducing mitochondrial ETC activity preserves NE morphology during ageing. This effect requires developmental mitochondrial superoxide, which downregulates SBP-1 (SREBP orthologue) and suppresses unsaturated fatty acid biosynthesis. The resulting reduction in unsaturated fatty acid levels limits lipid peroxidation, thereby preserving NE structure. Interventions targeting lipid peroxidation preserve NE integrity, extend lifespan in worms and ameliorate senescence-associated phenotypes in human fibroblasts and monkey cells mimicking Hutchinson–Gilford progeria syndrome disease. Our findings reveal a previously unrecognized role for mitochondrial superoxide as a protective developmental signal that programs long-term NE integrity. This work establishes lipid peroxidation control as a conserved strategy to delay nuclear ageing and highlights redox–lipid cross-talk as a therapeutic axis for healthy ageing.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$32.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to the full article PDF.
USD 39.95
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Data availability
The details of the chemical reagents, kits and software are provided in Supplementary Tables 5. All relevant data are available. The raw sequencing data for RNA-seq generated in this study have been deposited in the Genome Sequence Archive in the National Genomics Data Center under accession number CRA009340. Further information and requests for resources and reagents should be directed to and will be fulfilled by corresponding author. Source data are provided with this paper.
Code availability
Code is available on GitHub (https://github.com/hirscheylab/ddh/) for a package we generated that includes detailed documentation, which explains the model’s architecture, data preprocessing steps and how to run the code.
References
Mekhail, K. & Moazed, D. The nuclear envelope in genome organization, expression and stability. Nat. Rev. Mol. Cell Biol. 11, 317–328 (2010).
Strambio-De-Castillia, C., Niepel, M. & Rout, M. P. The nuclear pore complex: bridging nuclear transport and gene regulation. Nat. Rev. Mol. Cell Biol. 11, 490–501 (2010).
Uhler, C. & Shivashankar, G. V. Regulation of genome organization and gene expression by nuclear mechanotransduction. Nat. Rev. Mol. Cell Biol. 18, 717–727 (2017).
Hachiya, N. et al. Nuclear envelope and nuclear pore complexes in neurodegenerative diseases-new perspectives for therapeutic interventions. Mol. Neurobiol. 58, 983–995 (2021).
Chow, K. H., Factor, R. E. & Ullman, K. S. The nuclear envelope environment and its cancer connections. Nat. Rev. Cancer 12, 196–209 (2012).
Prissette, M. et al. Disruption of nuclear envelope integrity as a possible initiating event in tauopathies. Cell Rep. 40, 111249 (2022).
Pathak, R. U., Soujanya, M. & Mishra, R. K. Deterioration of nuclear morphology and architecture: a hallmark of senescence and aging. Ageing Res. Rev. 67, 101264 (2021).
Prokocimer, M. et al. Nuclear lamins: key regulators of nuclear structure and activities. J. Cell. Mol. Med. 13, 1059–1085 (2009).
Jacob, J. T. et al. Keratin 17 regulates nuclear morphology and chromatin organization. J. Cell Sci. 133, jcs254094 (2020).
Maheshwari, R., Rahman, M. M., Joseph-Strauss, D. & Cohen-Fix, O. An RNAi screen for genes that affect nuclear morphology in Caenorhabditis elegans reveals the involvement of unexpected processes. G3 11, jkab264 (2021).
Kristiani, L. & Kim, Y. The interplay between oxidative stress and the nuclear lamina contributes to laminopathies and age-related diseases. Cells 12, 1234 (2023).
Ruan, L. et al. Mitochondria-associated proteostasis. Annu. Rev. Biophys. 49, 41–67 (2020).
Ng, M. Y. W., Wai, T. & Simonsen, A. Quality control of the mitochondrion. Dev. Cell 56, 881–905 (2021).
Bock, F. J. & Tait, S. W. G. Mitochondria as multifaceted regulators of cell death. Nat. Rev. Mol. Cell Biol. 21, 85–100 (2020).
Ristow, M. & Schmeisser, K. Mitohormesis: promoting health and lifespan by increased levels of reactive oxygen species (ROS). Dose Response 12, 288–341 (2014).
Barcena, C., Mayoral, P. & Quiros, P. M. Mitohormesis, an antiaging paradigm. Int. Rev. Cell Mol. Biol. 340, 35–77 (2018).
Ristow, M. & Zarse, K. How increased oxidative stress promotes longevity and metabolic health: the concept of mitochondrial hormesis (mitohormesis). Exp. Gerontol. 45, 410–418 (2010).
Haithcock, E. et al. Age-related changes of nuclear architecture in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 102, 16690–16695 (2005).
Morales-Martinez, A., Dobrzynska, A. & Askjaer, P. Inner nuclear membrane protein LEM-2 is required for correct nuclear separation and morphology in C. elegans. J. Cell Sci. 128, 1090–1096 (2015).
Dillin, A. et al. Rates of behavior and aging specified by mitochondrial function during development. Science 298, 2398–2401 (2002).
Rea, S. L., Ventura, N. & Johnson, T. E. Relationship between mitochondrial electron transport chain dysfunction, development, and life extension in Caenorhabditis elegans. PLoS Biol. 5, e259 (2007).
Hansen, M., Hsu, A. L., Dillin, A. & Kenyon, C. New genes tied to endocrine, metabolic, and dietary regulation of lifespan from a Caenorhabditis elegans genomic RNAi screen. PLoS Genet. 1, 119–128 (2005).
Durieux, J. & Dillin, A. Mitochondria and aging: dilution is the solution. Cell Metab. 6, 427–429 (2007).
Senoo-Matsuda, N., Hartman, P. S., Akatsuka, A., Yoshimura, S. & Ishii, N. A complex II defect affects mitochondrial structure, leading to ced-3- and ced-4-dependent apoptosis and aging. J. Biol. Chem. 278, 22031–22036 (2003).
Yang, W. & Hekimi, S. Two modes of mitochondrial dysfunction lead independently to lifespan extension in Caenorhabditis elegans. Aging Cell 9, 433–447 (2010).
Bennett, C. F. et al. Activation of the mitochondrial unfolded protein response does not predict longevity in Caenorhabditis elegans. Nat. Commun. 5, 3483 (2014).
Chaudhari, S. N. & Kipreos, E. T. Increased mitochondrial fusion allows the survival of older animals in diverse C. elegans longevity pathways. Nat. Commun. 8, 182 (2017).
Houtkooper, R. H. et al. Mitonuclear protein imbalance as a conserved longevity mechanism. Nature 497, 451–457 (2013).
Kenyon, C., Chang, J., Gensch, E., Rudner, A. & Tabtiang, R. A C. elegans mutant that lives twice as long as wild type. Nature 366, 461–464 (1993).
Arantes-Oliveira, N., Apfeld, J., Dillin, A. & Kenyon, C. Regulation of life-span by germ-line stem cells in Caenorhabditis elegans. Science 295, 502–505 (2002).
Hsin, H. & Kenyon, C. Signals from the reproductive system regulate the lifespan of C. elegans. Nature 399, 362–366 (1999).
Lakowski, B. & Hekimi, S. The genetics of caloric restriction in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 95, 13091–13096 (1998).
Perez-Jimenez, M. M., Rodriguez-Palero, M. J., Rodenas, E., Askjaer, P. & Munoz, M. J. Age-dependent changes of nuclear morphology are uncoupled from longevity in Caenorhabditis elegans IGF/insulin receptor daf-2 mutants. Biogerontology 15, 279–288 (2014).
Shpilka, T. & Haynes, C. M. The mitochondrial UPR: mechanisms, physiological functions and implications in ageing. Nat. Rev. Mol. Cell Biol. 19, 109–120 (2018).
Zhu, D., Li, X. & Tian, Y. Mitochondrial-to-nuclear communication in aging: an epigenetic perspective. Trends Biochem. Sci. 47, 645–659 (2022).
Zhang, H. et al. Inter-tissue communication of mitochondrial stress and metabolic health. Life Metab. 2, load001 (2023).
Nargund, A. M., Pellegrino, M. W., Fiorese, C. J., Baker, B. M. & Haynes, C. M. Mitochondrial import efficiency of ATFS-1 regulates mitochondrial UPR activation. Science 337, 587–590 (2012).
Merkwirth, C. et al. Two conserved histone demethylases regulate mitochondrial stress-induced longevity. Cell 165, 1209–1223 (2016).
Tian, Y. et al. Mitochondrial stress induces chromatin reorganization to promote longevity and UPR(mt). Cell 165, 1197–1208 (2016).
Zhu, D. et al. NuRD mediates mitochondrial stress-induced longevity via chromatin remodeling in response to acetyl-CoA level. Sci. Adv. 6, eabb2529 (2020).
Shao, L.-W. et al. Histone deacetylase HDA-1 modulates mitochondrial stress response and longevity. Nat. Commun. 11, 4639 (2020).
Haynes, C. M., Petrova, K., Benedetti, C., Yang, Y. & Ron, D. ClpP mediates activation of a mitochondrial unfolded protein response in C. elegans. Dev. Cell 13, 467–480 (2007).
Li, T. Y. et al. The transcriptional coactivator CBP/p300 is an evolutionarily conserved node that promotes longevity in response to mitochondrial stress. Nat. Aging 1, 165–178 (2021).
Zhou, J. et al. Mitochondrial stress orchestrates chromatin remodeling and longevity via phosphoregulation of the NuRD component LIN-40. Sci. China Life Sci. 68, 3340–3352 (2025).
Zhao, R. Z., Jiang, S., Zhang, L. & Yu, Z. B. Mitochondrial electron transport chain, ROS generation and uncoupling (Review). Int. J. Mol. Med. 44, 3–15 (2019).
Yang, W. & Hekimi, S. A mitochondrial superoxide signal triggers increased longevity in Caenorhabditis elegans. PLoS Biol. 8, e1000556 (2010).
Zhitkovich, A. N-acetylcysteine: antioxidant, aldehyde scavenger, and more. Chem. Res. Toxicol. 32, 1318–1319 (2019).
Ramachandran, P. V. et al. Lysosomal signaling promotes longevity by adjusting mitochondrial activity. Dev. Cell 48, 685–696 (2019).
Kowaltowski, A. J., de Souza-Pinto, N. C., Castilho, R. F. & Vercesi, A. E. Mitochondria and reactive oxygen species. Free Radic. Biol. Med. 47, 333–343 (2009).
Murphy, M. P. How mitochondria produce reactive oxygen species. Biochem. J 417, 1–13 (2009).
Brand, M. D. The sites and topology of mitochondrial superoxide production. Exp. Gerontol. 45, 466–472 (2010).
Li, N. et al. Mitochondrial complex I inhibitor rotenone induces apoptosis through enhancing mitochondrial reactive oxygen species production. J. Biol. Chem. 278, 8516–8525 (2003).
Castello, P. R., Drechsel, D. A. & Patel, M. Mitochondria are a major source of paraquat-induced reactive oxygen species production in the brain. J. Biol. Chem. 282, 14186–14193 (2007).
Zarse, K. et al. Impaired insulin/IGF1 signaling extends life span by promoting mitochondrial L-proline catabolism to induce a transient ROS signal. Cell Metab. 15, 451–465 (2012).
Barayeu, U. et al. Hydropersulfides inhibit lipid peroxidation and ferroptosis by scavenging radicals. Nat. Chem. Biol. 19, 28–37 (2023).
Urban, N. et al. Non-linear impact of glutathione depletion on C. elegans life span and stress resistance. Redox Biol. 11, 502–515 (2017).
Jenkins, N. L. et al. Changes in ferrous iron and glutathione promote ferroptosis and frailty in aging Caenorhabditis elegans. Elife 9, e56580 (2020).
Van Raamsdonk, J. M. & Hekimi, S. Deletion of the mitochondrial superoxide dismutase sod-2 extends lifespan in Caenorhabditis elegans. PLoS Genet. 5, e1000361 (2009).
An, J. H. & Blackwell, T. K. SKN-1 links C. elegans mesendodermal specification to a conserved oxidative stress response. Genes Dev. 17, 1882–1893 (2003).
Chavez, V., Mohri-Shiomi, A., Maadani, A., Vega, L. A. & Garsin, D. A. Oxidative stress enzymes are required for DAF-16-mediated immunity due to generation of reactive oxygen species by Caenorhabditis elegans. Genetics 176, 1567–1577 (2007).
Senchuk, M. M. et al. Activation of DAF-16/FOXO by reactive oxygen species contributes to longevity in long-lived mitochondrial mutants in Caenorhabditis elegans. PLoS Genet. 14, e1007268 (2018).
Hwang, A. B. & Lee, S. J. Regulation of life span by mitochondrial respiration: the HIF-1 and ROS connection. Aging 3, 304–310 (2011).
Lee, S. J., Hwang, A. B. & Kenyon, C. Inhibition of respiration extends C. elegans life span via reactive oxygen species that increase HIF-1 activity. Curr. Biol. 20, 2131–2136 (2010).
Hwang, A. B. et al. Feedback regulation via AMPK and HIF-1 mediates ROS-dependent longevity in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 111, E4458–E4467 (2014).
Watts, J. L. Using Caenorhabditis elegans to uncover conserved functions of omega-3 and omega-6 fatty acids. J. Clin. Med. 5, 19 (2016).
Entchev, E. V. et al. LET-767 is required for the production of branched chain and long chain fatty acids in Caenorhabditis elegans. J. Biol. Chem. 283, 17550–17560 (2008).
Watts, J. L. & Browse, J. Genetic dissection of polyunsaturated fatty acid synthesis in Caenorhabditis elegans. Proc. Natl Acad. Sci. USA 99, 5854–5859 (2002).
Kniazeva, M. et al. Suppression of the ELO-2 FA elongation activity results in alterations of the fatty acid composition and multiple physiological defects, including abnormal ultradian rhythms, in Caenorhabditis elegans. Genetics 163, 159–169 (2003).
Yang, F. et al. An ARC/mediator subunit required for SREBP control of cholesterol and lipid homeostasis. Nature 442, 700–704 (2006).
Van Gilst, M. R., Hadjivassiliou, H., Jolly, A. & Yamamoto, K. R. Nuclear hormone receptor NHR-49 controls fat consumption and fatty acid composition in C. elegans. PLoS Biol. 3, e53 (2005).
Brock, T. J., Browse, J. & Watts, J. L. Genetic regulation of unsaturated fatty acid composition in C. elegans. PLoS Genet. 2, e108 (2006).
Taubert, S., Van Gilst, M. R., Hansen, M. & Yamamoto, K. R. A Mediator subunit, MDT-15, integrates regulation of fatty acid metabolism by NHR-49-dependent and -independent pathways in C. elegans. Genes Dev. 20, 1137–1149 (2006).
Agmon, E., Solon, J., Bassereau, P. & Stockwell, B. R. Modeling the effects of lipid peroxidation during ferroptosis on membrane properties. Sci Rep. 8, 5155 (2018).
Mas-Bargues, C., Escriva, C., Dromant, M., Borras, C. & Vina, J. Lipid peroxidation as measured by chromatographic determination of malondialdehyde. Human plasma reference values in health and disease. Arch. Biochem. Biophys. 709, 108941 (2021).
Ayala, A., Munoz, M. F. & Arguelles, S. Lipid peroxidation: production, metabolism, and signaling mechanisms of malondialdehyde and 4-hydroxy-2-nonenal. Oxid. Med. Cell Longev. 2014, 360438 (2014).
Drummen, G. P., van Liebergen, L. C., Op den Kamp, J. A. & Post, J. A. C11-BODIPY(581/591), an oxidation-sensitive fluorescent lipid peroxidation probe: (micro)spectroscopic characterization and validation of methodology. Free Radic. Biol. Med. 33, 473–490 (2002).
Sakamoto, T., Maebayashi, K., Nakagawa, Y. & Imai, H. Deletion of the four phospholipid hydroperoxide glutathione peroxidase genes accelerates aging in Caenorhabditis elegans. Genes Cells 19, 778–792 (2014).
Sakamoto, T., Maebayashi, K., Tsunoda, Y. & Imai, H. Inhibition of lipid peroxidation during the reproductive period extends the lifespan of Caenorhabditis elegans. J. Clin. Biochem. Nutr. 66, 116–123 (2020).
Niki, E. Role of vitamin E as a lipid-soluble peroxyl radical scavenger: in vitro and in vivo evidence. Free Radic. Biol. Med. 66, 3–12 (2014).
Gaschler, M. M. et al. Determination of the subcellular localization and mechanism of action of ferrostatins in suppressing ferroptosis. ACS Chem. Biol. 13, 1013–1020 (2018).
von Krusenstiern, A. N. et al. Identification of essential sites of lipid peroxidation in ferroptosis. Nat. Chem. Biol. 19, 719–730 (2023).
Hobson, C. M. et al. Correlating nuclear morphology and external force with combined atomic force microscopy and light sheet imaging separates roles of chromatin and lamin A/C in nuclear mechanics. Mol. Biol. Cell 31, 1788–1801 (2020).
Jevtic, P., Edens, L. J., Vukovic, L. D. & Levy, D. L. Sizing and shaping the nucleus: mechanisms and significance. Curr. Opin. Cell Biol. 28, 16–27 (2014).
Beck, M. & Hurt, E. The nuclear pore complex: understanding its function through structural insight. Nat. Rev. Mol. Cell Biol. 18, 73–89 (2017).
D’Angelo, M. A., Raices, M., Panowski, S. H. & Hetzer, M. W. Age-dependent deterioration of nuclear pore complexes causes a loss of nuclear integrity in postmitotic cells. Cell 136, 284–295 (2009).
Wu, L. et al. An ancient, unified mechanism for metformin growth inhibition in C. elegans and cancer. Cell 167, 1705–1718 (2016).
Gruenbaum, Y., Margalit, A., Goldman, R. D., Shumaker, D. K. & Wilson, K. L. The nuclear lamina comes of age. Nat. Rev. Mol. Cell Biol. 6, 21–31 (2005).
Kristiani, L., Kim, M. & Kim, Y. Role of the nuclear lamina in age-associated nuclear reorganization and inflammation. Cells 9, 718 (2020).
Almendáriz-Palacios, C. et al. The nuclear lamina: protein accumulation and disease. Biomedicines 8, 188 (2020).
Eriksson, M. et al. Recurrent de novo point mutations in lamin A cause Hutchinson–Gilford progeria syndrome. Nature 423, 293–298 (2003).
De Sandre-Giovannoli, A. et al. Lamin A truncation in Hutchinson–Gilford progeria. Science 300, 2055 (2003).
Lopez-Otin, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. Hallmarks of aging: an expanding universe. Cell 186, 243–278 (2022).
Martins, F., Sousa, J., Pereira, C. D., da Cruz, E. S. O. A. B. & Rebelo, S. Nuclear envelope dysfunction and its contribution to the aging process. Aging Cell 19, e13143 (2020).
Burrows, A. E., Smogorzewska, A. & Elledge, S. J. Polybromo-associated BRG1-associated factor components BRD7 and BAF180 are critical regulators of p53 required for induction of replicative senescence. Proc. Natl Acad. Sci. USA 107, 14280–14285 (2010).
Beausejour, C. M. et al. Reversal of human cellular senescence: roles of the p53 and p16 pathways. EMBO J. 22, 4212–4222 (2003).
Karoutas, A. & Akhtar, A. Functional mechanisms and abnormalities of the nuclear lamina. Nat. Cell Biol. 23, 116–126 (2021).
Burke, B. & Stewart, C. L. Life at the edge: the nuclear envelope and human disease. Nat. Rev. Mol. Cell Biol. 3, 575–585 (2002).
Wang, F. et al. Generation of a Hutchinson-Gilford progeria syndrome monkey model by base editing. Protein Cell 11, 809–824 (2020).
Nishizawa, H. et al. Lipid peroxidation and the subsequent cell death transmitting from ferroptotic cells to neighboring cells. Cell Death Dis. 12, 332 (2021).
Scaffidi, P. & Misteli, T. Lamin A-dependent nuclear defects in human aging. Science 312, 1059–1063 (2006).
Lee, B. Y. et al. Senescence-associated beta-galactosidase is lysosomal beta-galactosidase. Aging Cell 5, 187–195 (2006).
Heckenbach, I. et al. Nuclear morphology is a deep learning biomarker of cellular senescence. Nat. Aging 2, 742–755 (2022).
Krizhevsky, A., Sutskever, I. & Hinton, G. E. ImageNet classification with deep convolutional neural networks. Commun. ACM 60, 84–90 (2017).
Shelhamer, E., Long, J. & Darrell, T. Fully convolutional networks for semantic segmentation. IEEE Trans. Pattern Anal. Mach. Intell. 39, 640–651 (2017).
Shadel, G. S. & Horvath, T. L. Mitochondrial ROS signaling in organismal homeostasis. Cell 163, 560–569 (2015).
Hamanaka, R. B. & Chandel, N. S. Mitochondrial reactive oxygen species regulate cellular signaling and dictate biological outcomes. Trends Biochem. Sci 35, 505–513 (2010).
Yun, J. & Finkel, T. Mitohormesis. Cell Metab. 19, 757–766 (2014).
Xu, S. & Chisholm, A. D. C. elegans epidermal wounding induces a mitochondrial ROS burst that promotes wound repair. Dev. Cell 31, 48–60 (2014).
Tormos, K. V. et al. Mitochondrial complex III ROS regulate adipocyte differentiation. Cell Metab. 14, 537–544 (2011).
West, A. P., Shadel, G. S. & Ghosh, S. Mitochondria in innate immune responses. Nat. Rev. Immunol. 11, 389–402 (2011).
Johnson, D., Allman, E. & Nehrke, K. Regulation of acid-base transporters by reactive oxygen species following mitochondrial fragmentation. Am. J. Physiol. Cell Physiol. 302, C1045–C1054 (2012).
Hamanaka, R. B. et al. Mitochondrial reactive oxygen species promote epidermal differentiation and hair follicle development. Sci. Signal 6, ra8 (2013).
Yu, G., Wang, L. G., Han, Y. & He, Q. Y. clusterProfiler: an R package for comparing biological themes among gene clusters. OMICS 16, 284–287 (2012).
Zhang, Q. et al. The mitochondrial unfolded protein response is mediated cell-non-autonomously by retromer-dependent Wnt signaling. Cell 174, 870–883 (2018).
Acknowledgements
We thank the laboratory members of Y.T. for insightful discussions and Y. Liu for help in strain maintenance. We are grateful to D. C. Xu from the Shanghai Institute of Organic Chemistry and Y. Y. Niu from Kunming University of Science and Technology for sharing monkey HGPS (p.Cys1824Thr LMNA) fibroblast cells; and to T. Li and L. K. Wang from the Institute of Biophysics for sharing human BJ fibroblast cells and the assistance with cell culture. We thank the Center for Biological Imaging (CBI), Institute of Biophysics, Chinese Academy of Science for our electron microscopy, and we are grateful to L. Wang and C. Peng for their help in making EM samples. Y.T. is supported by the National Key R&D Program of China (2022YFA1303000), the National Natural Science Foundation of China (32225025, 32321004, 92254305), the CAS Project for Young Scientists in Basic Research (YSBR-076) and the New Cornerstone Science Foundation through the XPLORER PRIZE. X.W. is supported by the National Natural Science Foundation of China (32371214). Q.Z. is supported by the National Natural Science Foundation of China (32471212). C.Z. is supported by the CAS project (CAS-WX2022GC-0104). We acknowledge the Japanese National BioResource Project and the Caenorhabditis Genetics Center, funded by the National Institutes of Health-Officer of Research Infrastructure Programs (P40 OD010440) for providing the C. elegans strains used in this work.
Author information
Authors and Affiliations
Contributions
Y.T., P.C. and L.Z. conceived the study and designed the experiments. P.C., L.Z., J.Z. and X.H. performed the genetic crosses, RNAi experiments and lifespan analysis, as well as imaging and data analysis. P.C., L.Z., X.W. and Q.Z. performed the RNA-seq experiment analysis. P.C., L.Z., Y.L., Q.Z. and F.Z. performed lipidomic data analysis. C.Z, Z.L. and Q.Y. assisted with the AI-based imaging quantification platform. Y.T., P.C. and L.Z. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
Y.T., P.C., L.Z. and X.W. have applied a patent (202511963787.6 China) for the use of lipid peroxidation inhibitors in the treatment of premature ageing syndrome and delay ageing. Z.L., Q.Y., C.Z., Y.T. and P.C. have applied a patent (202310952096.0, China) for a cellular senescence state prediction method based on nuclear membrane morphology. The other authors declare no competing interests.
Peer review
Peer review information
Nature Metabolism thanks the anonymous reviewers for their contribution to the peer review of this work. Primary Handling Editor: Christoph Schmitt, in collaboration with the Nature Metabolism team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Developmental inhibition of cco-1 RNAi is crucial for NE protection during aging.
a, Quantification of the intestinal nuclei area in NPP-1::GFP animals grown on EV or cco-1 RNAi bacteria during aging. b, Relative fold changes of cco-1 expression were quantified by qPCR to assess RNAi knockdown efficiency. c, Oxygen consumption rate (OCR) was measured in N2 animals grown on EV or cco-1 RNAi bacteria at the L4 stage. d, Representative images of hypodermal NE morphology in EMR-1::mCherry animals grown on EV or cco-1 RNAi bacteria during aging. e, Quantification of the nuclei based is on their morphology, as depicted in (d). f, Quantification of the nuclei is based on their circularity ratio, as depicted in (d). g, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on cco-1 RNAi bacteria during development or during post-development (start from late L4 or young adult stage) with age. h, Quantification of the nuclei is based on their morphology, as depicted in (g). i, Quantification of the nuclei is based on their circularity ratio, as depicted in (g). 100 nuclei from more than 20 animals were examined for each group (e,h); the exact number of NE in each class was provided as source data; 3 independent experiments were analyzed in each condition (b,e,h). 5 independent experiments were analyzed in each condition (c). n = 50 nuclei were analyzed in each condition (a), n = 20 nuclei were analyzed in each condition (f,i). Scale bars, 10 μm (d,g). Data are mean ± s.e.m. P values were calculated using two-sided unpaired t-test (b,c), Chi-square test (e,h), One-way ANOVA (a,f,i). Source numerical data are available in the source data.
Extended Data Fig. 2 Changes in NE morphology across different ETC perturbations and long-lived mutants.
a, Representative images of intestinal NE morphology in wild-type or mev-1(kn1) animals grown on OP50 bacteria. b, Quantification of the intestinal nuclei is based on their morphology, as depicted in (a). c, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (a). d, Representative images of intestinal NE morphology in isp-1 RNAi, isp-1(qm150), nuo-6 RNAi, or nuo-6(qm200) animals with age. e, Quantification of the intestinal nuclei is based on their morphology, as depicted in (d). f, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (d). g, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV, tomm-22, cts-1, or spg-7 RNAi bacteria. h, Quantification of the intestinal nuclei is based on their morphology, as depicted in (g). i, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (g). j, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV or mrps-5 RNAi bacteria. k, Quantification of the intestinal nuclei is based on their morphology, as depicted in (j). l, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (j). m, Representative images of intestinal NE morphology in wild-type, daf-2(e1370), glp-1(e2141), eat-2(ad1116) animals with age. n, Quantification of the intestinal nuclei is based on their morphology, as depicted in (m). o, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (m). 100 nuclei from more than 20 animals were examined for each group (b,e,h,k,n); the exact number of NE in each class was provided as source data; 3 independent experiments were analyzed in each condition (b,e,h,k,n). n = 20 nuclei were analyzed in each condition (c,f,i,l,o). Scale bars, 10 μm (a,d,g,j,m). Data are mean ± s.e.m. P values were derived from two-sided Pearson’s chi-square tests (b,e,h,k,n), One-way ANOVA (c,f,i,l,o). Source numerical data are available in the source data.
Extended Data Fig. 3 Mitochondrial ROS induction underlies NE protection.
a, Representative images of intestinal NE morphology in wild-type, daf-16(mu86), hif-1(ok2564) or skn-1(zu169) animals grown on cco-1 RNAi bacteria. b, Quantification of the intestinal nuclei is based on their morphology, as depicted in (a). c, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (a). d, Representative images of intestinal NE morphology in wild-type, lgg-1(bp500), or atg-3(bp412) animals grown on EV or cco-1 RNAi bacteria. e, Quantification of the nuclei is based on their morphology, as depicted in (d). f, Quantification of the nuclei is based on their circularity ratio, as depicted in (d). g, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV bacteria with 0/0.1/0.2 mM paraquat (PQ) supplementation. h, Quantification of the nuclei is based on their morphology, as depicted in (g). i, Quantification of the nuclei is based on their circularity ratio, as depicted in (g). j, Representative images of the subcellular location of SOD-2 or SOD-3. TOMM-20 is a mitochondrial outer membrane protein. 100 nuclei from more than 20 animals were examined for each group (b,e,h); the exact number of NE in each class was provided as source data; 3 independent experiments were analyzed in each condition (b,e,h). n = 20 nuclei were analyzed in each condition (c,f,i). Scale bars, 10 μm (a,d,g,j). Data are mean ± s.e.m. P values were derived from two-sided Pearson’s chi-square tests (b,e,h), One-way ANOVA (c,f,i). Source numerical data are available in the source data.
Extended Data Fig. 4 Developmental mitochondrial superoxide accumulation correlates with NE protection across genetic backgrounds.
a, Representative images of intestine stained with MitoSOX Red dye in N2 animals grown on EV, nuo-2, mev-1, cyc-1, cco-2, or atp-4 RNAi bacteria at L4 stage. b, Quantification of MitoSOX red fluorescence in animals as depicted in (a). c, Representative images of intestine stained with MitoSOX Red dye in N2, isp-1(qm150) or nuo-6(qm200) animals grown on OP50 at early or late L4 stage. d, Quantification of MitoSOX red fluorescence in animals as depicted in (c). e, Lifespan analysis of N2, isp-1(qm150), or nuo-6(qm200) animals grown on EV bacteria. f, Lifespan analysis of N2 animals grown on EV, isp-1 RNAi, or nuo-6 RNAi bacteria. g, Representative images of intestine stained with MitoSOX Red dye in N2 animals grown on EV or mev-1 RNAi, N2 or mev-1(kn1) animals grown on OP50 at L4 stage. h, Quantification of MitoSOX red fluorescence in animals as depicted in (g). i, Representative images of intestine stained with MitoSOX Red dye in N2, daf-2(e1370), glp-1(e2141), eat-2 (ad1116), isp-1 RNAi, or isp-1(qm150) animals at L4 stage. j, Quantification of MitoSOX red fluorescence in animals as depicted in (i). At least 20 animals were quantified in each condition (b,d,h,j). Scale bars, 20 μm (a,c,g,i). Data are mean ± s.e.m. P values were calculated using One-way ANOVA (b,d,h,j). Lifespan data were analyzed with Kaplan Meier (e,f). Source numerical data are available in the source data.
Extended Data Fig. 5 Glutathione (GSH) mediates the disruptive effect of NAC on NE protection during decreased ETC activity.
a, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV or cco-1 RNAi bacteria with or without NAC (8 mM), DEM (0.5 mM) or NAC + DEM supplementation. b, Quantification of the intestinal nuclei is based on their morphology, as depicted in (a). c, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (a). d, Relative GSH level of L4 animals grown on EV or cco-1 RNAi bacteria with or without NAC (8 mM). e, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV or cco-1 RNAi bacteria with or without GSH (10 mM) supplementation. GSH supplementation from L1 to L4. f, Quantification of the intestinal nuclei is based on their morphology, as depicted in (e). g, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (e). 100 nuclei from more than 20 animals were examined for each group (b,f); the exact number of NE in each class was provided as source data; 3 independent experiments were analyzed in each condition (b,d,f). n = 20 nuclei were analyzed in each condition (c,g). Scale bars, 10 μm (a,e). Data are mean ± s.e.m. P values were derived from two-sided Pearson’s chi-square tests (b,f), One-way ANOVA (c,d,g). Source numerical data are available in the source data.
Extended Data Fig. 6 Canonical oxidative stress pathways are dispensable for NE protection when ETC activity decreases.
a, Representative images of intestinal NE morphology in wild-type, daf-16(mu86), hif-1(ok2564) or skn-1(zu169) animals grown on cco-1 RNAi bacteria. b, Quantification of the intestinal nuclei based on their morphology as depicted in (a). c, Quantification of the intestinal nuclei based on their circularity ratio as depicted in (a). d, Venn diagram of differentially expressed genes (DEGs) in N2 animals grown on EV or cco-1 RNAi bacteria with or without NAC (8 mM). Genes with (|log2FoldChange| ≥ 0.5, adjusted P ≤ 0.05) were selected as DEGs. e, Venn diagram of differentially abundant fatty acids in N2 animals grown on EV or cco-1 RNAi bacteria with or without NAC (8 mM). Fatty acids with the value of variable importance in the projection (VIP) > 0.5, P ≤ 0.05 (Student’s t test) were selected. f, RT-qPCR analyses of sbp-1 in N2 animals grown on EV, cco-1, isp-1,or nuo-6 RNAi bacteria, or in N2, isp-1(qm150), or nuo-6(qm200) mutants grown on OP50 bacteria. g, RT-qPCR analyses of sbp-1 in N2 animals grown on EV, mev-1 RNAi bacteria. h, RT-qPCR analyses of sbp-1 in N2, daf2-(e1370), eat-2(ad1116), or glp-1(e2141) mutants grown on OP50 bacteria. 100 nuclei from more than 20 animals were examined for each group (b); the exact number of NE in each class was provided as source data; 3 independent experiments were analyzed in each condition (b,f,g,h). n = 20 nuclei were analyzed in each condition (c). Scale bars, 10 μm (a). Data are mean ± s.e.m. P values were derived from two-sided Pearson’s chi-square tests (b), One-way ANOVA (c,f,h), two-sided unpaired t-test (g). Source numerical data are available in the source data.
Extended Data Fig. 7 Genetic and dietary reduction of UFAs preserves NE morphology.
a, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV, let-767, elo-1, or elo-2 RNAi bacteria. b, Quantification of the intestinal nuclei is based on their morphology, as depicted in (a). c, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (a). d, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV, cco-1 RNAi, or sod-2(gk257);sod-3(tm760) animals with or without AA (C20:4, 2 mM) supplementation. e, Quantification of the intestinal nuclei is based on their morphology, as depicted in (d). f, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (d). g, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV or cco-1 RNAi bacteria with or without oleic acid (OA, C18:1, 5 mM) supplementation. h, Quantification of the intestinal nuclei is based on their morphology, as depicted in (g). i, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (g). j, Representative images of intestinal NE morphology in NPP-1::GFP animals grown on EV or cco-1 RNAi bacteria with or without stearic acid (SA, C18:0, 5 mM) supplementation. k, Quantification of the intestinal nuclei is based on their morphology, as depicted in (j). l, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (j). 100 nuclei from more than 20 animals were examined for each group (b,e,h,k); the exact number of NE in each class was provided as source data; 3 independent experiments were analyzed in each condition (b,e,h,k). n = 20 nuclei were analyzed in each condition (c,f,i,l). Scale bars, 10 μm (a,d,g,j). Data are mean ± s.e.m. P values were derived from two-sided Pearson’s chi-square tests (b,e,h,k), One-way ANOVA (c,f,i,l). Source numerical data are available in the source data.
Extended Data Fig. 8 Inhibition of lipid peroxidation protects NE morphology and function with age.
a, Representative images of animals stained with lipid peroxidation probe BODIPYTM 581/591 C11 grown on EV or cco-1 RNAi bacteria at Day1 stage. b, Quantification of oxidized lipid ratio based on the fluorescence of BODIPYTM 581/591 C11 staining as depicted in (a). c, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (Fig. 5c). d, Quantification of the nuclei is based on their circularity ratio, as depicted in (Fig. 5e). e, Representative images of intestinal NE morphology in NPP-1::GFP animals in DMSO or FINO2 (2 mM). f, Quantification of the intestinal nuclei is based on their morphology, as depicted in (e). g, Quantification of the intestinal nuclei is based on their circularity ratio, as depicted in (e). h, Quantification of the nuclei is based on their circularity ratio, as depicted in (Fig. 5h). i, Representative fluorescent micrographs of intestinal nuclear permeability in wild-type, cco-1 RNAi, or fer-2 (5 mM) supplementation animals injected with 70 kD dextran in the cytosol and corresponding fluorescence intensity distribution curve. j, Quantification of the ratio of leaky nuclei as depicted in (i). k, Representative fluorescent micrographs of intestinal nuclear permeability in wild-type, cco-1 RNAi, or fer-2 (5 mM) supplementation animals injected with 500 kD dextran in the cytosol with age and corresponding fluorescence intensity distribution curve. l, Quantification of the ratio of leaky nuclei as depicted in (k). n = 21 animals were analyzed in each condition(b). 100 nuclei from more than 20 animals were examined for each group (f), the exact number of NE in each class was provided as source data; n = 20 nuclei were analyzed in each condition (c,d,g,h). n = 3 independent biological repeats (f,j,l). Scale bars, 10 μm (e,i,k). 20 μm (a). Data are mean ± s.e.m. P values were derived from two-sided unpaired t-test (b), two-sided Pearson’s chi-square tests (f), One-way ANOVA (c,d,g,h,j,i). Source numerical data are available in the source data.
Extended Data Fig. 9 Iron chelation and anti-lipid peroxidation interventions preserve NE integrity and extend lifespan.
a, Representative images of intestinal NE morphology in lmn-1 RNAi animals grown on OP50 bacteria with or without VE (5 mM) or fer-2 (2 mM) supplementation. b, Quantification of the intestinal nuclei is based on their morphology, as depicted in (a). c, Lifespan analysis of N2 animals grown on EV or cco-1 RNAi bacteria with or without NAC (8 mM) supplementation. d, Lifespan analysis of N2 animals grown on OP50 E.coli bacteria with or without fer-2 (2 mM or 5 mM) supplementation. e, Lifespan analysis of N2 animals grown with or without FINO2 (2 mM) supplementation. f, Lifespan analysis of N2 animals grown on EV bacteria with or without EPA (C22:5, 2 or 5 mM) supplementation. g, Lifespan analysis of N2 animals grown on EV or cco-1 RNAi bacteria with or without Fer-2 or FINO2 (Fer-2 5 mM, FINO2 2 mM) supplementation. h, Lifespan analysis of N2 animals grown on EV or cco-1 RNAi bacteria with or without 2,2’-bipyridyl (bpy, 10 μM) supplementation. i, Lifespan analysis of N2 animals grown on EV or cco-1 RNAi bacteria with or without 2 2’-bipyridyl (bpy, 50 μM) supplementation. 100 nuclei from more than 20 animals were examined for each group (a); the exact number of NE in each class was provided as source data. 3 independent experiments were analyzed in each condition (b). Scale bars, 10 μm (a). Data are mean ± s.e.m. P values were derived from two-sided Pearson’s chi-square tests (b); lifespan data were analyzed with Kaplan-Meier (c,d,e,f,g,h,i). Source numerical data are available in the source data.
Extended Data Fig. 10 Anti-lipid peroxidation interventions preserve NE morphology and delay cell senescence in normal and HGPS cells.
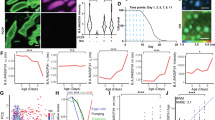
a-c, Analysis workflow of the roundness (a), smoothness (b), and invagination of nuclear envelope (c) of the nuclei in BJ fibroblasts. d-f, Quantification of the roundness (d), smoothness (e), and invagination (f) of the nuclei in BJ fibroblasts. 20 nuclei were analyzed in (b). 50 nuclei were examined for each group (d); Scale bars, 10 μm (a,b,c). Data are mean ± s.e.m. P values were calculated using One-way ANOVA (d,e,f). Source numerical data are available in the source data.
Supplementary information
Supplementary Information (download PDF )
Supplementary Figs. 1–5; Key Resources table.
Supplementary Table 1 (download XLSX )
RNA-seq data.
Supplementary Table 2 (download XLSX )
Targeted-lipidomic data.
Supplementary Table 3 (download XLSX )
qPCR primer list.
Supplementary Table 4 (download XLSX )
Strain list.
Supplementary Data 1 (download XLSX )
Statistical source data.
Supplementary Data 2 (download XLSX )
Statistical source data.
Supplementary Data 3 (download XLSX )
Statistical source data.
Supplementary Data 4 (download XLSX )
Statistical source data.
Supplementary Data 5 (download XLSX )
Statistical source data.
Source data
Source Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 1 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 2 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 3 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 4 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 5 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 6 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 7 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 8 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 9 (download XLSX )
Statistical source data.
Source Data Extended Data Fig. 10 (download XLSX )
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, P.X., Zhang, L., Wu, X. et al. Mitochondrial superoxide regulates nuclear envelope integrity and ageing via redox-mediated lipid metabolism. Nat Metab 8, 371–388 (2026). https://doi.org/10.1038/s42255-026-01452-9
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s42255-026-01452-9