Abstract
Human exposure to environmental microplastics and nanoplastics via oral ingestion is a topic of public health and food safety concern. In this Review, the physicochemical properties of drug-delivery nanoparticles were considered to evaluate the biological plausibility of environmental nanoplastics overcoming biological-selective barriers. Physicochemical characteristics probably have a key role in the interaction of nanoplastics with the mucus layer and the intestinal mucosal epithelium. Substantial gaps in experimental designs and analytical methodologies pose a major challenge to fully understanding nanoplastic uptake and should be prioritized for future research.
Similar content being viewed by others
Main
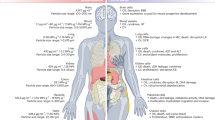
Human exposure to plastics through oral ingestion is a significant food safety and public health concern1,2,3. The World Health Organization has recently reviewed the current evidence on the presence of microplastics and nanoplastics in food, focusing on potential human health implications4. Similarly, the European Food Safety Authority Expert Panel has raised concerns regarding their presence, particularly in seafood products5. Comprehensive data on the occurrence of nanoplastics and associated migrating substances—such as polyethylene glycol (PEG)6—in various foods have been provided by Vitali et al.7, including seafood (on average 30 items per individual, where ‘items’ refers to small pieces of plastic, without specifying the type of polymer or their size; 15 items per gram), fruits and vegetables, salt (average 400 items per kg), honey (average 600 items per kg), sugar, water (10,000 items per litre) and beverages (100 items per litre)7. Additionally, dairy products, alcoholic and non-alcoholic beverages have also demonstrated contamination, with reported concentrations ranging from 300 µg g−1 to 6,930 µg g−1 (refs. 8,9).
This food contamination is directly attributable to human-driven plastic pollution. Recent estimates suggest that each person produces approximately 35 kg of plastic per year, contributing to a global total of 275 million tonnes of plastic waste annually. Improper and sometimes illegal plastic waste management leads to its dissemination into environmental compartments and entry into the food chain10. Food represents a direct exposure route to microplastics and nanoplastics for humans; however, regulatory guidelines for exposure risk assessment and management are still lacking3. A recent review by Ramsperger et al.3 reported the presence of small plastic particles in the human colon and faeces (on average 28 microplastics per gram, predominantly fibrous), highlighting that these data were consistently obtained from post-mortem human samples or during surgical procedures. The behaviour and fate of nanoparticles within the human body have attracted growing scientific interest, particularly concerning how the intestine processes these nanoparticles. This reveals the existing knowledge gaps, limited evidence and ongoing contradictions regarding the fate of nanoplastics in the human body. This uncertainty is exacerbated by the lack of a universally accepted scientific definition of ‘nanoplastics’, which hampers the comparability of studies and contributes to significant methodological variability. Gigault et al.11 defined nanoplastics as particles unintentionally produced from the degradation and manufacture of plastic objects, exhibiting colloidal behaviour and sizes ranging from 1 nm to 1,000 nm. In 2019, Hartmann et al.12 emphasized the lack of a unified definition and pragmatically proposed that ‘nanoplastics’ should refer to particles sized between 1 nm and 100 nm. More recently, however, Mitrano et al.13 have pointed out that no consensus exists regarding the upper size limit or the specific physicochemical properties. Therefore, the term ‘nanoplastics’ remains scientifically controversial, as does their biological fate and behaviour. By contrast, some evidence regarding the pathways by which nanoparticles—although not specifically nanoplastics—can cross intestinal biological barriers was already provided in 2010 by Powell et al.14, identifying endocytosis by enterocytes, microfold (M) cells in Peyer’s patches and passive paracellular transport as the main mechanisms14. Regarding nanoparticles, several studies have focused on drug-delivery nanoparticles to investigate their post-oral ingestion behaviour.
This Review aims to highlight the optimal characteristics of drug-delivery nanoparticles that enable effective interaction with the intestinal mucus layer and epithelium. This collection of information is intended to serve as a basis for a deeper understanding of nanoplastic-related pathways. This Review seeks to apply the drug-delivery nanoparticle model to virgin and environmental nanoplastics, while clearly specifying its applicability limits.
Protein corona
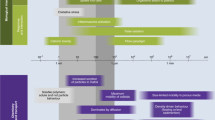
The use of nanoparticles for therapeutic purposes represents a valuable opportunity to optimize oral drug delivery15. Both in vitro and in vivo studies are being conducted to improve their application (Table 1). Oral administration of nanoparticles faces several challenges, including their resistance to the gastric environment16, poor penetration through the mucus layer (which has a thickness of 30–300 µm in the stomach and 150–400 µm in the small intestine)17, and the intestinal mucosa, which are selective biological barriers that limit their absorption into the bloodstream18,19. It has been established that the physicochemical properties of nanoparticles—specifically size, hydrophilicity and surface charge—have a crucial role in overcoming these barriers19,20. Interaction with biomolecules in the gastric environment leads to the formation of a protein corona, which influences the identity of the nanoparticles, their subsequent cellular interactions and immune recognition21,22,23,24,25. The mechanisms underlying corona formation in the gastric environment remain unclear; however, the composition of the protein corona (Fig. 1) is influenced by the nanoparticles’ physicochemical properties and the patient’s physiological conditions26.
The surface of the nanoplastic is covered by proteins, lipid components and other potential biomolecules, which together determine its biological properties, interactions with cells, and fate in the biological microenvironment.
In vitro studies on poly(lactic-co-glycolic acid) (PLGA) nanoparticles (200–440 nm in diameter), widely used in drug delivery, have demonstrated protein corona formation after exposure to simulated biological fluids, with the identification of specific adsorbed proteins27,28. Current nanoparticle design strategies aim to minimize protein corona formation using hydrophilic polymer coatings—such as PEG, polyvinyl alcohol and polyacrylates—or by adding cross-linkers or functional groups to modulate protein adsorption, enhance interaction selectivity and reduce immune recognition25,27,28,29. In vitro investigations (Table 1) have shown that the most effective nanoparticles for drug delivery typically have sizes below 250 nm, appropriate surface charges, hydrophilicity and surface functionalization that promote mucus diffusion and cellular uptake, modulation of the protein corona to limit opsonization and the ability to exploit multiple endocytic mechanisms15,19,20,30,31,32,33,34,35,36,37,38,39 (Table 3). Positively charged nanoparticles, functionalized with chitosan, promote electrostatic interactions with the intestinal mucus, enhancing muco-adhesion and activating endocytosis mediated by clathrin, caveolin and macropinocytosis. By contrast, neutral or slightly negatively charged nanoparticles, typically PEGylated, favour muco-penetration, reduce immune recognition and increase systemic residence time17,20,31,33,35,40.
Assuming effective mucus penetration, it is evident that nanoparticle size and shape are key determinants. Epithelial intracellular spaces present junctions sized between 0.3 nm and 1 nm, which can widen up to a maximum of 20 nm via specific receptors40,41. This indicates that nanoparticles larger than 20 nm cannot passively cross the intestinal mucosa and must rely on receptor-mediated endocytic mechanisms, which themselves have additional size limitations (Table 1). Regarding shape, although in vitro drug-delivery studies primarily use spherical nanoparticles15,19,20,30,31,32,33,34,35,36,37,38,39, evidence suggests that compact and elongated nanoparticles—such as rods or ellipsoids—may exhibit superior intestinal mucus penetration due to reduced interaction and higher mobility. Additionally, elongated, disc-shaped or elliptical particles may demonstrate higher endocytosis rates in vitro compared to spherical particles17,41.
Crossing the intestinal mucus layer—the first selective barrier when administered orally42—the fate of nanoparticles is determined in the complex interaction context between intestinal enterocytes of the intestinal epithelium and M cells (Fig. 2), which is probably dependent on the surface characteristics of the nanoparticles themselves.
Microplastics and nanoplastics have at least three possible routes from the intestinal lumen to the lamina propria: (1) endocytosis by enterocytes; (2) between tight junctions; and (3) through M cells. After this, they are probably phagocytosed by dendritic cells and macrophages.
Endocytic pathways
The internalization of nanoparticles by intestinal enterocytes can occur through various endocytic pathways. Clathrin-mediated endocytosis directs nanoparticles to lysosomes, where they may undergo degradation, whereas caveolin-mediated endocytosis and macropinocytosis allow non-degradative translocation towards the Golgi apparatus and endoplasmic reticulum41,43. Beyond classical cellular routes, recent studies indicate that chylomicrons—lipoproteins synthesized by enterocytes for lipid transport—may contribute to the systemic distribution of nanoparticles modified with hydrophobic surface coatings44,45,46. Conversely, uptake by phagocytic cells may provide a pathway to bypass intracellular degradation, as these cells are characterized by lower enzymatic activity compared to enterocytes41,43. M cells facilitate the translocation of nanoparticles from the intestinal lumen to dendritic cells and macrophages located in the lamina propria, enabling subsequent professional phagocytosis41,43,47. These findings suggest that the design of drug-delivery nanoparticles is carefully tailored to specific applications and can be engineered to either promote or avoid interactions with immune cells48,49. This concept may also be relevant to environmental nanoplastics. Although the formation of a protein corona on environmental nanoplastics remains uncharacterized, including the composition, relative abundance of the associated proteins and their capacity to traverse selective biological barriers, a plausible scenario can be envisioned. If the surface properties of these nanoplastics permit them to cross such barriers and reach immune cells, the protein corona could mediate interactions with phagocytic immune cells. Consequently, this mechanism may represent a potential pathway for nanoplastic internalization50. Additionally, the presence of inflammatory bowel diseases appears to significantly increase the transport and absorption of PEG–PLGA nanoparticles compared to healthy conditions, probably because of inflammatory cytokines that disrupt endocytic mechanisms, as well as lesions or breaches in the intestinal barrier at inflamed sites. The physicochemical surface properties of nanoparticles allow them to specifically target these gastrointestinal inflammatory lesions15,39,51.
Nanoplastic uptake
The cellular uptake of nanoplastics is supported by various in vitro and in vivo experimental studies (Table 2). Available experimental evidence primarily involves virgin nanoplastics, which are synthesized and sometimes surface-modified or functionalized for experimental purposes42,52,53,54,55,56,57,58,59,60,61,62,63,64,65,66,67. Both in vitro and in vivo experiments mainly use polystyrene nanoplastics (size range: 10 nm to 10 µm, most frequently 20–500 nm). In vitro studies generally confirm the cellular uptake of polystyrene nanoplastics; however, in most cases, the absorption mechanisms remain hypothetical and are not validated using specific endocytic pathway inhibitors. Conversely, some in vitro studies directly investigate the mechanisms underlying cellular internalization. For example, Kharaghani et al.52 used inhibitors of clathrin-mediated endocytosis and macropinocytosis to identify the endocytic mechanism responsible for the uptake of spherical gold-core polystyrene nanoplastics (25 nm) in epithelial cells within a small intestine triculture model, suggesting their involvement. Xu et al.61 used endocytic pathway inhibitors following exposure of polystyrene nanoplastics (100 nm, spherical) to Caco-2 cells, suggesting internalization via clathrin-mediated endocytosis. Ding et al.62 suggested that spherical polystyrene nanoplastics (100 nm) are internalized in GES-1 cells via caveolin-mediated endocytosis, with a higher uptake rate for amino-modified nanoplastics. Liu et al.65 proposed that spherical polystyrene nanoplastics (50 nm and 500 nm) are internalized in RBL-2H3 cells via clathrin-mediated endocytosis for 50-nm particles and via macropinocytosis for 500-nm particles, supported by confocal microscopy observations.
Across these studies, a common feature is the surface modification of nanoplastics with functional groups during synthesis52,61,62,65. It is important to note that, although many studies did not directly investigate uptake mechanisms, the authors nonetheless suggest the occurrence of cellular internalization. Specifically, the potential for nanoplastic uptake and accumulation in lysosomes56 and nuclei68 has been proposed. By contrast, Paul et al.42 reported the absence of nanoplastics (in this case, spherical polymethylmethacrylate of 25 nm) both inside and outside the cell membrane after exposure. In vivo studies have demonstrated the presence of nanoplastics in intestinal sections, sometimes distributed within the villi and lamina propria63, along with intracellular effects such as dysfunctional mitophagy, intestinal barrier weakening56 and mitochondrial damage59, confirmed via confocal microscopy, fluorescence microscopy and transmission electron microscopy.
However, in vivo studies in mice do not investigate the underlying cellular uptake mechanisms. An exception is the study by Das et al.69 in Daphnia magna, which suggested the absorption of polystyrene nanoplastics (54 nm, gold-core shell) functionalized with 4-mercaptobenzoic acid to facilitate Raman detection. Experimental evidence indicated uptake by intestinal epithelial cells via clathrin-mediated endocytosis and macropinocytosis, confirmed using endocytic pathway inhibitors. These mechanisms also appeared to enable nanoplastic translocation from the intestine to other organs. Das et al.69 highlighted that the polystyrene nanoplastics were synthesized using a cationic initiator (2,2′-azobis[2-methylpropionamidine] dihydrochloride or AIBA), which confers a positive surface charge, potentially enhancing interactions with intestinal epithelial cells. Similarly, an ex vivo study on Dicentrarchus labrax suggested the translocation of spherical polystyrene nanoplastics (50 nm) across the intestinal epithelium70. In this experimental design, nanoplastics were functionalized with amine groups to facilitate analytical detection. Although such functionalization was intended to enhance analytical sensitivity, it may also have influenced nanoplastic behaviour. Moreover, analytical techniques currently used present substantial variability and lack of standardization, potentially leading to overestimated conclusions regarding the translocation across biological barriers and subsequent accumulation. For example, it has been reported that when using virgin, often fluorescent nanoplastics, the fluorescence detected in zebrafish tissues may result from fluorophore leaching rather than actual barrier penetration and accumulation71.
An environmental study not based on laboratory exposure reported the presence of microplastic particles (300 µm–1 mm, film and fibrous forms) throughout the bodies of bivalves. Soft tissues were digested using an aqueous potassium hydroxide solution (10% at 40 °C for 24 h). Once isolated, microplastics were analysed by Fourier-transform infra-red spectroscopy. Although the presence of microplastics in bivalve bodies was confirmed72, no clear information emerged regarding their fate and behaviour within the organism. Regarding the potential role of chylomicrons in nanoplastic absorption, no specific experimental investigations are currently available. However, nanoparticles designed to structurally mimic lipids that are typically incorporated into chylomicrons were not found to be incorporated73.
Across the studies considered, it is evident that virgin nanoplastics are predominantly used. These materials cannot fully represent environmental nanoplastics. Commercial nanoplastics—mainly polystyrene—do not reflect environmental heterogeneity, as environmental nanoplastics undergo exposure to light, heat, salts, enzymes and natural organic substances that alter their surface charge, hydrophobicity and, consequently, their biological behaviour74. Environmental nanoplastics are considered hybrids of a polymeric core and adsorbed organic molecules, ions and contaminants, all of which can influence biological and cellular interactions. Furthermore, recent comparisons of laboratory-based nanoplastic aging protocols highlight a lack of standardization among aging methods, raising concerns about their environmental representativeness. For example, the use of unrealistic UV-C radiation, excessively high temperatures, insufficient biofilm replication and incomplete protein corona formation may limit the relevance of laboratory models75. Nanoplastics incubated in biologically active environments may acquire a specific coating associated with the host’s digestive tract, influencing subsequent biological interactions—a condition that is difficult to replicate in vitro50,76,77. The presence of an eco-corona, which may include lipid and protein components, could influence cellular uptake by mimicking surface functionalization50. In the context of nanoparticle-based drug delivery, intestinal inflammatory conditions have been shown to increase mucus layer permeability. Similarly, plastic particle ingestion in model organisms such as Danio rerio has been associated with increased intestinal permeability78. Increased intestinal permeability is also suggested in vitro due to the compromised integrity of the epithelial barrier52,79.
Lessons from nanoparticle drug delivery
The nanoparticle absorption model identifies the physicochemical characteristics that enable optimal translocation across biological-selective barriers. In our view, this model could be comparatively applied to virgin and environmental nanoplastics to address some of the current gaps in oral exposure risk assessment, based on the potential overlap—or divergence—of specific properties between nanoplastics and drug-delivery nanoparticles80. However, although the sharing of certain properties may be plausible, the inherent heterogeneity of nanoplastics compared to drug-delivery nanoparticles must be carefully considered81.
A critical comparison of the characteristics and properties of drug-delivery nanoparticles, virgin nanoplastics and environmental nanoplastics is proposed in Table 2.
The body of knowledge developed for drug-delivery nanoparticles highlights specific characteristics designed to enable functional interactions with the mucus layer and the intestinal mucosa. Establishing behavioural analogies between drug-delivery nanoparticles and virgin or environmental nanoplastics may prove complex, as the surface properties and structures of environmental nanoplastics are neither controlled nor controllable11,82,83. Nonetheless, in our view, the proposed model could help identify which characteristics of environmental nanoplastics and which uptake pathways might facilitate their translocation across biological barriers and subsequent absorption.
Firstly, the properties of virgin nanoplastics influence the formation of the protein corona differently compared to environmental nanoplastics. Virgin nanoplastics, characterized by uniform shape, specific coatings and functionalized surface features—thus being less complex than real-world particles—tend to adsorb different proteins and/or in different proportions compared to environmental nanoplastics. This may affect their ability to cross biological-selective barriers and undergo cellular uptake. Experimental studies using virgin nanoplastics have occasionally reported the presence of ‘key receptor’ proteins that may facilitate interactions with the mucus layer and subsequent uptake. By contrast, for environmental nanoplastics, due to their complex adsorption chemistries across different and sequential biologically active environments, the protein corona may incorporate a diverse set of proteins that could affect interactions with the mucus layer and the translocation across the epithelial mucosa84 (Table 4).
The presence of small environmental plastic particles—at both micro- and nano-scale—has been investigated in various living organisms. For instance, Zhu et al.85 reported the detection of microplastics in human tissues, particularly in the small intestine. Their method, which involved alkaline digestion using potassium hydroxide (10% at 50 °C for 72 h under agitation at 150 rpm) followed by Fourier-transform infra-red spectroscopy, confirmed the presence of microplastics in the small intestine. However, no further investigation into subsequent biological interactions was performed. More recently, Di Fiore et al.86 reviewed the presence of microplastics and nanoplastics in various invertebrates, marine vertebrates and mammals. It was suggested that microplastics detected within organisms may have been passing through organs and tissues considered primary exposure points. This supports the occurrence of micro- and nano-scale plastic particles in organisms; what remains to be elucidated is whether these particles can consistently reach internal accumulation sites within deeper biological tissues86. In this context, current limitations in analytical methodologies have a crucial role, especially regarding the lack of standardized quality-control and quality assurance protocols that would ensure the robustness of the findings80,86,87.
Studies included in this Review suggest the translocation across biological-selective barriers and cellular uptake, primarily based on microscopic techniques—such as confocal microscopy58,61,88, fluorescence microscopy63 and transmission electron microscopy64, sometimes combined with Raman spectroscopy71 and flow cytometry64. Analytical limitations remain a major challenge in this research field, especially for validating and confirming the robustness of experimental results87. For example, fluorescence microscopy can detect plastic particles down to 3 μm, but its application for nanoplastic detection remains controversial due to the small size of nanoplastics15. This technique lacks the resolution needed for precise localization of nanoplastics within tissues and, importantly, for distinguishing them from free fluorescent dyes used for identification. Combining fluorescence microscopy with other techniques may improve resolution, as proposed by Nguyen and Tufenkji89, who developed a highly stable nanoplastic labelling method for stimulated emission depletion microscopy, achieving a resolution an order of magnitude higher than that of conventional fluorescence microscopy. Confocal microscopy also presents dimensional limitations. It has been reported that its resolution is insufficient for the precise and unequivocal localization of particles smaller than 200 nm. Moreover, the challenge of differentiating background signals further complicates the accurate identification and distribution of nanoplastics in biological environments90. Domenech et al.88 reported that polystyrene nanoplastics—probably exhibiting surface modifications due to their monodisperse behaviour in water—were detected within epithelial monolayer cells in bicellular and tricellular in vitro models, as observed by confocal fluorescence microscopy88,91. Even in this case, a deeper evaluation is required to confirm the actual intracellular presence of nanoplastics, considering the concerns regarding the accuracy of the analytical techniques used. Therefore, to unequivocally assess the translocation across biological-selective barriers, it is essential to further investigate and optimize analytical methodologies in terms of robustness, precision and accuracy.
Current limitations and future perspectives
This Review aimed to provide a model to assess the plausibility of orally ingested nanoplastics overcoming biological-selective barriers, using evidence available from nanoparticles used in drug delivery. The proposed model suggests that the physicochemical characteristics of nanoparticles influence their ability to cross selective barriers. Specifically, small particle size (<250 nm), suitable surface charge and possible surface functionalization appear to enhance this phenomenon. The frequent use of virgin nanoplastics in experimental designs—often fluorescently labelled and/or surface-modified for analytical purposes—does not accurately reflect the heterogeneity of environmental nanoplastics, nor does it adequately describe their behaviour and fate within the human intestine. The situation is further complicated by the substantial limitations of current analytical methodologies, which remain inadequate for the precise and unequivocal localization of nanoplastics within biological tissues, especially for particles smaller than 200 nm. In a few experimental scenarios, combined exposure through both water and food has shown an additive accumulation of nanoplastics at the intestinal level, with more pronounced inflammatory effects compared to single exposure routes43. This suggests the need to evaluate the potential influence of the food matrix on nanoplastic ingestion and subsequent fate. Furthermore, experimental designs should carefully consider the various biologically active environments through which nanoplastics transit. Environmental conditions affect the colloidal stability and aggregation phenomena of nanoplastics, ultimately reducing endocytic uptake mechanisms92. Therefore, there is a clear need to optimize experimental designs—considering mainly environmental nanoplastics93,94 —and improve analytical techniques to fully elucidate the fate of environmental nanoplastics in the human body. This is particularly urgent given the increasing evidence of adverse health effects, including metabolic disorders, neurotoxicity and potential carcinogenicity95.
References
Liu, Q. et al. Microplastics and nanoplastics: emerging contaminants in food. J. Agric. Food Chem. 69, 10450–10468 (2021).
Zarus, G. M., Muianga, C., Hunter, C. M. & Pappas, R. S. A review of data for quantifying human exposures to micro and nanoplastics and potential health risks. Sci. Total Environ. 756, 144010 (2021).
Ramsperger, A. F. R. M. et al. Nano- and microplastics: a comprehensive review on their exposure routes, translocation, and fate in humans. NanoImpact 29, 100441 (2023).
World Health Organization. Dietary and Inhalation Exposure to Nano- and Microplastic Particles and Potential Implications for Human Health (WHO, 2022).
European Food Safety Authority Panel on Contaminants in the Food Chain (CONTAM). Presence of microplastics and nanoplastics in food, with particular focus on seafood. EFSA J. 14, e04501 (2016).
Landrigan, P. J. et al. The Minderoo-Monaco Commission on Plastics and Human Health. Ann. Glob. Health 89, 23 (2023).
Vitali, C., Peters, R. J. B., Janssen, H.-G. & Nielen, M. W. F. Microplastics and nanoplastics in food, water, and beverages; part I. occurrence. Trends Anal. Chem. 159, 116670 (2023).
Boctor, J. et al. Microplastics and nanoplastics: fate, transport, and governance from agricultural soil to food webs and humans. Environ. Sci. Eur. 37, 68 (2025).
Jeffries, C., Rauert, C. & Thomas, K. V. Quantifying nanoplastics and microplastics in food and beverages using pyrolysis-gas chromatography–mass spectrometry: challenges and implications. ACS Food Sci. Technol. 5, 1536–1545 (2025).
Nelis, J. L. D. et al. The measurement of food safety and security risks associated with micro- and nanoplastic pollution. Trends Anal. Chem. 161, 116993 (2023).
Gigault, J. et al. Current opinion: what is a nanoplastic?. Environ. Pollut. 235, 1030–1034 (2018).
Hartmann, N. B. et al. Are we speaking the same language? Recommendations for a definition and categorization framework for plastic debris. Environ. Sci. Technol. 53, 1039–1047 (2019).
Mitrano, D. M., Wick, P. & Nowack, B. Placing nanoplastics in the context of global plastic pollution. Nat. Nanotechnol. 16, 491–500 (2021).
Powell, J. J., Faria, N., Thomas-McKay, E. & Pele, L. C. Origin and fate of dietary nanoparticles and microparticles in the gastrointestinal tract. J. Autoimmun. 34, J226–J233 (2010).
Feng, S., Raimi-Abraham, B. T. & Vllasaliu, D. PEG-PLGA nanoparticles transport across in vitro intestinal epithelial models and show potential for oral delivery of antibodies in inflammatory bowel disease. J. Drug Deliv. Sci. Technol. 108, 106925 (2025).
Desai, P. P., Date, A. A. & Patravale, V. B. Overcoming poor oral bioavailability using nanoparticle formulations – opportunities and limitations. Drug Discov. Today Technol. 9, e87–e95 (2012).
Wang, Y. et al. The influence of nanoparticle properties on oral bioavailability of drugs. Int. J. Nanomedicine 15, 6295–6310 (2020).
Berardi, A. & Baldelli Bombelli, F. Oral delivery of nanoparticles - let’s not forget about the protein corona. Expert Opin. Drug Deliv. 16, 563–566 (2019).
Schreiner, J., Brettner, F. E. B., Gier, S., Vogel-Kindgen, S. & Windbergs, M. Unlocking the potential of microfold cells for enhanced permeation of nanocarriers in oral drug delivery. Eur. J. Pharm. Biopharm. 202, 114408 (2024).
Bai, Y. et al. Nanoparticles with surface features of dendritic oligopeptides as potential oral drug delivery systems. J. Mater. Chem. B 8, 2636–2649 (2020).
Mahmoudi, M. et al. Protein−nanoparticle interactions: opportunities and challenges. Chem. Rev. 111, 5610–5637 (2011).
Zhang, T., Zhu, G., Lu, B., Qian, Z. & Peng, Q. Protein corona formed in the gastrointestinal tract and its impacts on oral delivery of nanoparticles. Med. Res. Rev. 41, 1835–1850 (2021).
Zhou, P., Guo, M. & Cui, X. Effect of food on orally-ingested titanium dioxide and zinc oxide nanoparticle behaviors in simulated digestive tract. Chemosphere 268, 128843 (2021).
Xiao, Q. et al. The effects of protein corona on in vivo fate of nanocarriers. Adv. Drug Deliv. Rev. 186, 114356 (2022).
Yang, D. et al. Protein coronas derived from mucus act as both spear and shield to regulate transferrin functionalized nanoparticle transcellular transport in enterocytes. ACS Nano 18, 7455–7472 (2024).
Pino, P. D. et al. Protein corona formation around nanoparticles – from the past to the future. Mater. Horiz. 1, 301–313 (2014).
Semete, B. et al. Effects of protein binding on the biodistribution of PEGylated PLGA nanoparticles post oral administration. Int. J. Pharm. 424, 115–120 (2012).
Partikel, K., Korte, R., Mulac, D., Humpf, H.-U. & Langer, K. Serum type and concentration both affect the protein-corona composition of PLGA nanoparticles. Beilstein J. Nanotechnol. 10, 1002–1015 (2019).
Liu, H. et al. Modulating the protein corona on nanoparticles by finely tuning cross-linkers improves macrophage targeting in oral small interfering RNA delivery. ACS Nano 19, 16469–16487 (2025).
Song, Y. et al. Oral delivery system for low molecular weight protamine-dextran-poly(lactic-co-glycolic acid) carrying exenatide to overcome the mucus barrier and improve intestinal targeting efficiency. Nanomed. 14, 989–1009 (2019).
Zhou, J. et al. A nano-delivery system based on preventing degradation and promoting absorption to improve the oral bioavailability of insulin. Int. J. Biol. Macromol. 244, 125263 (2023).
Feng, Q. -P. et al. Oral administration co-delivery nanoparticles of docetaxel and bevacizumab for improving intestinal absorption and enhancing anticancer activity. Mater. Sci. Eng. C Mater. Biol. Appl. 124, 112039 (2021).
Song, Y. et al. Synthesis of CSK-DEX-PLGA nanoparticles for the oral delivery of exenatide to improve its mucus penetration and intestinal absorption. Mol. Pharm. 16, 518–532 (2019).
Liang, Q. et al. Development of rifapentine-loaded PLGA-based nanoparticles: in vitro characterisation and in vivo study in mice. Int. J. Nanomedicine 15, 7491–7507 (2020).
Akhtar, B., Muhammad, F., Aslam, B., Saleemi, M. K. & Sharif, A. Pharmacokinetic profile of chitosan modified poly lactic co-glycolic acid biodegradable nanoparticles following oral delivery of gentamicin in rabbits. Int. J. Biol. Macromol. 164, 1493–1500 (2020).
Ranganathan, A. et al. Poly (d, l-lactide-co-glycolide)-phospholipid nanocarrier for efficient delivery of macular pigment lutein: absorption pharmacokinetics in mice and antiproliferative effect in Hep G2 cells. Drug Deliv. Transl. Res. 9, 178–191 (2019).
Yu, M. et al. Rapid transport of deformation-tuned nanoparticles across biological hydrogels and cellular barriers. Nat. Commun. 9, 2607 (2018).
Anderski, J., Mahlert, L., Mulac, D. & Langer, K. Mucus-penetrating nanoparticles: promising drug delivery systems for the photodynamic therapy of intestinal cancer. Eur. J. Pharm. Biopharm. 129, 1–9 (2018).
Yagi, Y. et al. Oral administration of PLGA nanoparticles to deliver antisense oligonucleotides to inflammatory lesions in the gastrointestinal tract. Biol. Pharm. Bull. 47, 848–855 (2024).
Ejazi, S. A., Louisthelmy, R. & Maisel, K. Mechanisms of nanoparticle transport across intestinal tissue: an oral delivery perspective. ACS Nano 17, 13044–13061 (2023).
Wang, Y. et al. Intestinal nanoparticle delivery and cellular response: a review of the bidirectional nanoparticle-cell interplay in mucosa based on physiochemical properties. J. Nanobiotechnology 22, 669 (2024).
Paul, M. B., Böhmert, L., Hsiao, I.-L., Braeuning, A. & Sieg, H. Complex intestinal and hepatic in vitro barrier models reveal information on uptake and impact of micro-, submicro- and nanoplastics. Environ. Int. 179, 108172 (2023).
Wang, J. et al. Enhanced gut-to-liver oral drug delivery via ligand-modified nanoparticles by attenuating protein corona adsorption. ACS Nano 18, 35310–35324 (2024).
Yao, M., McClements, D. J., Zhao, F., Craig, R. W. & Xiao, H. Controlling the gastrointestinal fate of nutraceutical and pharmaceutical-enriched lipid nanoparticles: from mixed micelles to chylomicrons. NanoImpact 5, 13–21 (2017).
Beg, S., Alam, M. N., Ahmad, F. J. & Singh, B. Chylomicron mimicking nanocolloidal carriers of rosuvastatin calcium for lymphatic drug targeting and management of hyperlipidemia. Colloids Surf. B Biointerfaces 177, 541–549 (2019).
Elnady, R. E., Amin, M. M. & Zakaria, M. Y. A review on lipid-based nanocarriers mimicking chylomicron and their potential in drug delivery and targeting infectious and cancerous diseases. AAPS Open 9, 13 (2023).
Sahay, G., Alakhova, D. Y. & Kabanov, A. V. Endocytosis of nanomedicines. J. Control. Release 145, 182–195 (2010).
Zolnik, B. S., González-Fernández, Á, Sadrieh, N. & Dobrovolskaia, M. A. Minireview: nanoparticles and the immune system. Endocrinology 151, 458–465 (2010).
Dobrovolskaia, M. A., Shurin, M. & Shvedova, A. A. Current understanding of interactions between nanoparticles and the immune system. Toxicol. Appl. Pharmacol. 299, 78–89 (2016).
Ramsperger, A. F. R. M. et al. Environmental exposure enhances the internalization of microplastic particles into cells. Sci. Adv. 6, eabd1211 (2020).
Zhang, M. & Merlin, D. Nanoparticle-based oral drug delivery systems targeting the colon for treatment of ulcerative colitis. Inflamm. Bowel Dis. 24, 1401–1415 (2018).
Kharaghani, D. et al. Toxicity and absorption of polystyrene micro-nanoplastics in healthy and Crohn’s disease human duodenum-chip models. J. Hazard. Mater. 490, 137714 (2025).
Du, B. et al. Analysis of biodistribution and in vivo toxicity of varying sized polystyrene micro and nanoplastics in mice. Int. J. Nanomedicine 19, 7617–7630 (2024).
Liang, B. et al. Underestimated health risks: polystyrene micro- and nanoplastics jointly induce intestinal barrier dysfunction by ROS-mediated epithelial cell apoptosis. Part. Fibre Toxicol. 18, 20 (2021).
Hillery, A., Jani, P. & Florence, A. Comparative, quantitative study of lymphoid and non-lymphoid uptake of 60 nm polystyrene particles. J. Drug Target. 2, 151–156 (1994).
Zhang, Y., Jia, Z., Gao, X., Zhao, J. & Zhang, H. Polystyrene nanoparticles induced mammalian intestine damage caused by blockage of BNIP3/NIX-mediated mitophagy and gut microbiota alteration. Sci. Total Environ. 907, 168064 (2024).
Paul, M. B. et al. Beyond microplastics - investigation on health impacts of submicron and nanoplastic particles after oral uptake in vitro. Microplast. Nanoplast. 2, 16 (2022).
Nikolic, S. et al. Orally administered fluorescent nanosized polystyrene particles affect cell viability, hormonal and inflammatory profile, and behavior in treated mice. Environ. Pollut. 305, 119206 (2022).
Yasin, N. A. E. et al. Exposure to polystyrene nanoparticles induces liver damage in rat via induction of oxidative stress and hepatocyte apoptosis. Environ. Toxicol. Pharmacol. 94, 103911 (2022).
Schwarzfischer, M. et al. Ingested nano- and microsized polystyrene particles surpass the intestinal barrier and accumulate in the body. NanoImpact 25, 100374 (2022).
Xu, D., Ma, Y., Han, X. & Chen, Y. Systematic toxicity evaluation of polystyrene nanoplastics on mice and molecular mechanism investigation about their internalization into Caco-2 cells. J. Hazard. Mater. 417, 126092 (2021).
Ding, Y. et al. Tissue distribution of polystyrene nanoplastics in mice and their entry, transport, and cytotoxicity to GES-1 cells. Environ. Pollut. 280, 116974 (2021).
Zhang, Z. et al. Continuous oral exposure to micro- and nanoplastics induced gut microbiota dysbiosis, intestinal barrier and immune dysfunction in adult mice. Environ. Int. 182, 108353 (2023).
Li, Y. et al. In vitro study on the toxicity of nanoplastics with different charges to murine splenic lymphocytes. J. Hazard. Mater. 424, 127508 (2022).
Liu, L. et al. Cellular internalization and release of polystyrene microplastics and nanoplastics. Sci. Total Environ. 779, 146523 (2021).
Ruan, Y. et al. Correlation between cellular uptake and cytotoxicity of polystyrene micro/nanoplastics in HeLa cells: a size-dependent matter. PLoS ONE 18, e0289473 (2023).
Xu, D. et al. Differently surface-labeled polystyrene nanoplastics at an environmentally relevant concentration induced Crohn’s ileitis-like features via triggering intestinal epithelial cell necroptosis. Environ. Int. 176, 107968 (2023).
Cortés, C. et al. Nanoplastics as a potential environmental health factor: effects of polystyrene nanoparticles on human intestinal epithelial Caco-2 cells. Environ. Sci. Nano 7, 272–285 (2020).
Das, A., Terry, L. R., Sanders, S., Yang, L. & Guo, H. Confocal surface-enhanced raman imaging of the intestinal barrier crossing behavior of model nanoplastics in Daphnia Magna. Environ. Sci. Technol. 58, 11615–11624 (2024).
Vagner, M. et al. Experimental evidence that polystyrene nanoplastics cross the intestinal barrier of European seabass. Environ. Int. 166, 107340 (2022).
Catarino, A. I., Frutos, A. & Henry, T. B. Use of fluorescent-labelled nanoplastics (NPs) to demonstrate NP absorption is inconclusive without adequate controls. Sci. Total Environ. 670, 915–920 (2019).
Magesh, N. S. & Ajith, K. V. Microplastics contamination and risk assessment in bivalves of economic importance from Beypore estuary, Southern India. Environ. Res. 261, 119711 (2024).
Hussain, N. Recent advances in the understanding of uptake of microparticulates across the gastrointestinal lymphatics. Adv. Drug Deliv. Rev. 50, 107–142 (2001).
Lee, Y. et al. Potential lifetime effects caused by cellular uptake of nanoplastics: a review. Environ. Pollut. 329, 121668 (2023).
Alimi, O. S. et al. Weathering pathways and protocols for environmentally relevant microplastics and nanoplastics: what are we missing?. J. Hazard. Mater. 423, 126955 (2022).
Schür, C. et al. Incubation in wastewater reduces the multigenerational effects of microplastics in Daphnia magna. Environ. Sci. Technol. 55, 2491–2499 (2021).
Nasser, F. & Lynch, I. Secreted protein eco-corona mediates uptake and impacts of polystyrene nanoparticles on Daphnia magna. J. Proteomics 137, 45–51 (2016).
Agrawal, M. et al. Micro- and nano-plastics, intestinal inflammation, and inflammatory bowel disease: a review of the literature. Sci. Total Environ. 953, 176228 (2024).
He, Y. et al. Polystyrene nanoplastics deteriorate LPS-modulated duodenal permeability and inflammation in mice via ROS drived-NF-κB/NLRP3 pathway. Chemosphere 307, 135662 (2022).
Rauert, C. et al. Assessing the efficacy of pyrolysis–gas chromatography–mass spectrometry for nanoplastic and microplastic analysis in human blood. Environ. Sci. Technol. 59, 1984–1994 (2025).
Gigault, J. et al. Nanoplastics are neither microplastics nor engineered nanoparticles. Nat. Nanotechnol. 16, 501–507 (2021).
Nanoplastic should be better understood. Nat. Nanotechnol. 14, 299–299 (2019).
Reynaud, S., Aynard, A., Grassl, B. & Gigault, J. Nanoplastics: from model materials to colloidal fate. Curr. Opin. Colloid Interface Sci. 57, 101528 (2022).
Ducoli, S. et al. A different protein corona cloaks “true-to-life” nanoplastics with respect to synthetic polystyrene nanobeads. Environ. Sci. Nano 9, 1414–1426 (2022).
Zhu, L. et al. Tissue accumulation of microplastics and potential health risks in human. Sci. Total Environ. 915, 170004 (2024).
Di Fiore, C., Ishikawa, Y. & Wright, S. L. A review on methods for extracting and quantifying microplastic in biological tissues. J. Hazard. Mater. 464, 132991 (2024).
Seewoo, B. J. et al. How do plastics, including microplastics and plastic-associated chemicals, affect human health?. Nat. Med. 30, 3036–3037 (2024).
Domenech, J., Hernández, A., Rubio, L., Marcos, R. & Cortés, C. Interactions of polystyrene nanoplastics with in vitro models of the human intestinal barrier. Arch. Toxicol. 94, 2997–3012 (2020).
Nguyen, B. & Tufenkji, N. Single-particle resolution fluorescence microscopy of nanoplastics. Environ. Sci. Technol. 56, 6426–6435 (2022).
Lv, Z., Su, B., Xu, X., Li, W. & Cui, W. Deep learning enables label-free nanoparticle localization from bright-field microscopy images. Colloids Surf. A Physicochem. Eng. Asp. 709, 136061 (2025).
Domenech, J. et al. Long-term effects of polystyrene nanoplastics in human intestinal Caco-2 cells. Biomolecules 11, 1442 (2021).
Boughbina-Portolés, A. & Campíns-Falcó, P. Assessing the size transformation of nanoplastics in natural water matrices. Sci. Total Environ. 953, 176225 (2024).
Dusza, H. M. et al. Uptake, transport, and toxicity of pristine and weathered micro- and nanoplastics in human placenta cells. Environ. Health Perspect. 130, 097006 (2022).
Gong, L. et al. Effects of weathering and simulated gastric fluid exposure on cellular responses to polystyrene particles. Environ. Sci. Nano 12, 528–547 (2025).
Rahman, A., Sarkar, A., Yadav, O. P., Achari, G. & Slobodnik, J. Potential human health risks due to environmental exposure to nano- and microplastics and knowledge gaps: a scoping review. Sci. Total Environ. 757, 143872 (2021).
Acknowledgements
This work was supported by the Horizon Europe (EDIAQI project no. 101057497) and the BRIC INAIL (CELLOPHAN project no. 14/2022). EDIAQI project is funded by the European Union under G.A. no. 101057497. Views and opinions expressed are, however, those of the authors only and do not necessarily reflect those of the European Union. Neither the European Union nor the granting authority can be held responsible for them. This study was carried out under the grant BRIC.INAIL ID 14/2022 ‘Approccio integrato per la determinazione e la caratterizzazione di microplastiche aerodisperse in ambienti di lavoro’; project title: ‘Caraterizzazione delle Emissioni in Luoghi di LavOro di microPlasticHe Aerodisperse e Nanoplastiche—CELLOPHAN’.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Health thanks Raffaele Marfella, Bhedita Seewoo and the other, anonymous, reviewer(s) for their contribution to the peer review of this work. Primary Handling Editor: Ben Johnson, in collaboration with the Nature Health team.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Di Fiore, C., Avino, P. Microplastics and nanoplastics in the human diet. Nat. Health 1, 48–57 (2026). https://doi.org/10.1038/s44360-025-00025-6
Received:
Accepted:
Published:
Version of record:
Issue date:
DOI: https://doi.org/10.1038/s44360-025-00025-6